Abstract
Per- and polyfluoroalkyl substances (PFAS) are a class of fluorinated chemicals used widely in consumer and industrial products. Their human toxicity and ecosystem impacts have received extensive public, scientific and regulatory attention. Regulatory PFAS guidance is rapidly evolving, with the inclusion of a wider range of PFAS included in advisories and a continued decrease in what is deemed safe PFAS concentrations. In this study we collated PFAS concentration data for over 45,000 surface and groundwater samples from around the world to assess the global extent of PFAS contamination and their potential future environmental burden. Here we show that a substantial fraction of sampled waters exceeds PFAS drinking water guidance values, with the extent of exceedance depending on the jurisdiction and PFAS source. Additionally, current monitoring practices probably underestimate PFAS in the environment given the limited suite of PFAS that are typically quantified but deemed of regulatory concern. An improved understanding of the range of PFAS embodied in consumer and industrial products is required to assess the environmental burden and develop mitigation measures. While PFAS is the focus of this study, it also highlights society’s need to better understand the use, fate and impacts of anthropogenic chemicals.
Similar content being viewed by others
Main
Per- and polyfluoroalkyl substances (PFAS) constitute a class of over 14,0001 chemicals extensively used in industrial applications and consumer products because of their distinct water and oil repellent properties and high heat tolerance. PFAS are defined as fluorinated substances that contain at least one fully fluorinated methyl or methylene carbon atom2. This includes fluoropolymers (for example, Teflon), some fluorinated insecticides (for example, Fludioxonil) and pharmaceuticals (for example, Bicalutamide)3. PFAS are referred to as ‘forever chemicals’4 because of their persistence in the environment. Perfluorooctanesulfonic acid (PFOS) and perfluorooctanoic acid (PFOA), two of the highest-profile PFAS, were added to the Stockholm Convention for the protection of human health and the environment from persistent organic pollutants (POPs)5 in 2009 and 2019, respectively, limiting their use and production. This also coincided with a shift from ‘legacy PFAS’ towards novel PFAS6 (Extended Data Table 1).
Regulators worldwide have proposed or regulated varying concentrations for PFAS in drinking water. One of the most restrictive recommendations for drinking water is Health Canada’s, with the sum of all PFAS being less than 30 ng l−1 (ref. 7), whereas the European Union recommends the sum off all PFAS being less than 500 ng l−1 or the sum of 20 select PFAS being less than 100 ng l−1 (ref. 8). It is noted, however, that currently Health Canada only requires quantification of either at least 18 PFAS or using US Environmental Protection Agency (EPA) methods 533 and/or 537.19. The US EPA has proposed drinking water concentration limits of 4 ng l−1 for PFOS and PFOA in their National Primary Drinking Water Regulation and limits on perfluorononanoic acid (PFNA), perfluorobutanesulfonic acid (PFBS), perfluorohexanesulfonic acid (PFHxS) and hexafluoropropylene oxide dimer acid (GenX) through the hazard index (HI)10.
Toxicity concerns increase with fluorinated chain length (FCL), because long-chain PFAS (FCL > 6) usually take longer to be excreted from the body due to their lower water solubility, higher affinity for serum proteins and enterohepatic recirculation, which increase their elimination time from plasma and tissue11,12,13. All perfluoroalkyl carboxylic acids (PFCA) with a FCL ≥ 7 are currently candidates for potential inclusion on the Stockholm Convention for the protection of human health and the environment from POPs5.
Certain PFAS degrade to terminal perfluoroalkyl carboxylic acids (PFCAs) and perfluoroalkyl sulfonic acids (PFSAs) and are referred to as precursors14 (Supplementary Table 1 and Supplementary Fig. 1). Precursors are used extensively in the manufacture of consumer products such as cosmetics, surface treated paper, waterproof textiles, insecticides, food packaging and firefighting foams15. Whereas there are too many PFAS precursors to list individually, they are generally separated into three major groupings: fluorotelomers, sulfonamides and polyfluorinated alkyl phosphate esters (PAPs).
Whereas studies have estimated PFAS production globally16,17 and quantified PFAS in commercial and industrial products, their fate is still unknown. Numerous studies have investigated PFAS extent in environmental compartments, including one that suggests that four select PFAS exceed the planetary boundary18. Studies have also assessed or compared aqueous phase PFAS concentrations in select regions19,20. Whereas it is widely acknowledged that PFAS are globally pervasive, the extent of PFAS in global surface (SW) and groundwater (GW) is unknown, as is the extent to which PFAS concentrations exceed proposed or implemented PFAS drinking water guidelines.
Here we investigate the extent and distribution of PFAS surface and groundwater contamination globally. We assess PFAS concentrations with respect to current and proposed PFAS drinking water regulations or advisories. Finally, we investigate the source of PFAS contamination, including the distribution of PFAS used in various consumer products, providing insights into the global pervasiveness of PFAS and the ability to predict the future environmental burden of PFAS.
Extent of global PFAS water contamination
To assess the global extent and importance of PFAS in the environment, an extensive global dataset was developed from 273 environmental studies since 2004, which include data for over 12,000 SW and 33,900 GW samples. As PFAS are not naturally occurring21, any PFAS found in the environment was introduced from a range of consumer and industrial products.
PFAS are pervasive in SW and GW worldwide (Fig. 1). Note that, while the mapped data suggest Australia, China, Europe and North America are PFAS hotspots relative to the world (Fig. 1a), when comparing against the number of samples collected (Fig. 1b), it implies that these are high-sampling zones, potentially skewing the representation of actual distribution. If research were undertaken in more locations worldwide at sites with high aqueous film forming foam (AFFF) usage, such as major airports, comparable PFAS contamination levels would probably be found. Additionally, high PFAS contamination in Fig. 1a is not limited to areas near manufacturing sites but also high-use areas. For example, Australia has no PFAS manufacturing facilities22,23 but has highly contaminated PFAS sites from firefighting activities. Furthermore, sampled locations could have higher PFAS concentrations compared to unsampled areas, as research efforts tend to concentrate on locations where PFAS presence is likely. Given this, the occurrence of surface and groundwater with large PFAS concentrations estimated in this study may be high.
a, Sum of concentration of 20 PFAS subject to EU guidance in surface water, groundwater and drinking water samples. Those above the EU drinking water limit of 100 ng l−1 (marked red on scale bar) are circled in red (for known contamination sources (for example, AFFF or non-AFFF)) or black (unknown sources). b, Number of PFAS samples available on a 5° longitude/latitude grid worldwide.
Threshold regulatory PFAS concentration limits are used to benchmark the PFAS global extent in SW and GW (Extended Data Table 2). PFAS sources were divided into three categories: known non-AFFF (for example, production facilities using or producing PFAS, landfills), known AFFF (for example, firefighting training area) or unknown. A higher proportion of samples exceeded threshold limits when associated with a known source of PFAS contamination compared with an unknown source (Fig. 2 and Extended Data Figs. 1–6). For GW samples with known AFFF contamination, 71, 72 and 63% exceeded the proposed US EPA HI (n = 6,312) or their proposed PFOS (n = 6,442) and PFOA (n = 6,447) drinking water regulation, respectively. However, when there was no known source, the incidence of exceedance of these criteria was still elevated (31, 50 and 40% for the US EPA HI (n = 14,905), PFOS (n = 15,351) and PFOA (n = 15,499) drinking water regulation, respectively). Given that guidance on PFAS threshold concentrations vary globally, the proportion of samples that are deemed of concern also varies. Groundwater with no known contamination source exceeded Health Canada’s criteria in 69% of samples whereas only 6% of these samples exceeded the EU’s sum of all PFAS criteria (500 ng l−1) (n = 16,151). If the alternate EU sum of 20 PFAS criteria is considered, 16% of groundwater samples with no known contamination source were in exceedance (n = 16,143). Regardless of the regulatory threshold considered, a large fraction of groundwater samples would be considered unacceptable for drinking water consumption. For known AFFF source SW samples, the proportion exceeding regulatory thresholds is similar to GW samples. However, when there was no known PFAS source, or a known non-AFFF source, the incidence of SW samples exceeding regulatory thresholds was lower. This is expected as residence times in surface waters are lower than for groundwater. For this analysis, samples that were below detection limits (BDL) were randomly assigned a concentration between zero and the detection limit. To assess potential bias, particularly for low-threshold criteria jurisdictions (for example, PFOA < 4 ng l−1 US EPA), this analysis was repeated with PFAS concentrations with BDL set to zero (Extended Data Table 3 and Supplementary Figs. 2–8). Whereas assumptions made dealing with detection limits impact results, both approaches conclude that an important fraction of samples exceeds regulatory threshold levels. As method-detection limits continually decrease, the extent of exceedances will be better informed.
For samples where PFAS concentrations were below detection limits, a PFAS concentration was randomly assigned between zero and the detection limit.
Where does PFAS come from
To assess PFAS sources to the environment, consumer and industrial products containing PFAS were divided into those used for AFFF and non-AFFF. AFFF applications typically result in high concentration point sources of PFAS, as do industrial manufacturing sites that synthesize or use PFAS. The latter are considered known (non-AFFF) sources in this study.
Non-AFFF consumer and industrial products
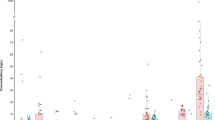
PFAS in 943 non-AFFF consumer products in 15 categories were characterized from 38 literature studies since 2010. In these studies, 113 PFAS were quantified, although at most 60 PFAS were analysed in any given study24. Comparison of PFAS classes in consumer products is challenging as the same suite of PFAS are not quantified in each study. For example, at least two PFCAs or PFSAs were measured in 89% and 69% of all non-AFFF product samples, respectively, whereas only 49%, 35%, 20%, 12% and 15% of studies quantified at least two fluorotelomers, sulfonamides, PAPs, novel or other PFAS, respectively. When measured, however, fluorotelomers and traditional PFCAs represented the dominant PFAS subclass in most of the product categories investigated (for example, coatings, cosmetics and textiles) (Fig. 3). Fluorotelomers represented a median of 72% of the total measured PFAS by mass in consumer products, whereas PFCAs represented 25%. PAPs and sulfonamides were also relevant when measured with a median of 14% and 7%, respectively. Interestingly, PFSAs were typically much lower, accounting for a median of 4% of the total quantified PFAS mass.
Box dimensions show the span between quartiles 1 and 3 (interquartile range, IQR). Outliers are defined as values greater than 1.5× the IQR. Whiskers extend from these quartiles to the largest (quartile 3) or smallest (quartile 1) non-outlier value (that is, <1.5× the IQR). Y-axis units are ng ml−1 or µg kg−1 equivalent to ppb.
Different jurisdictions worldwide provide guidance, or regulate, differing ranges of PFAS, with no standard approach to quantify PFAS. For example, the US EPA has three methods to measure PFAS in aqueous samples, methods 533, 537.1 and 8327, with an additional non-drinking aqueous method (1633) in development. EPA method 537 and its revisions have been the most used since 2009, quantifying 14 PFAS. In 2018, this method was revised as 537.1 to include four additional PFAS. All other EPA methods were developed in 2019 or later and quantify a total of 32 PFAS, including seven PFSA, 11 PFCA, three fluorotelomers, three sulfonamides and eight novel PFAS (Extended Data Table 2). In this study, EPA draft method 1633 is used as a benchmark as EPA methods are commonly used globally and method 1633 is the most comprehensive. In doing so, this provides a preliminary assessment of the extent to which the most comprehensive EPA method captures PFAS mass and the extent of unaccounted PFAS.
If only the PFAS listed in draft method 1633 were used to quantify PFAS in consumer products within this dataset, the total embodied PFAS would be substantially underestimated (Fig. 4) and the PFAS distribution would completely change. For example, the median concentration of PFAS regulated in the United States (sum of PFBS, PFHxS, PFOS, PFOA, PFNA and GenX) in textiles (n = 227) and coatings (n = 167) is two and three orders of magnitude smaller than the median of all PFAS quantified. Across all products, EPA method 1633 suggests a median distribution of 73% PFCA (n = 781), 11% PFSA (n = 750), 16% fluorotelomers (n = 353), 10% sulfonamides (n = 242) and 0.1% novel PFAS (n = 27), with phosphate-based PFAS not being quantified with this method. This results in the proportion of PFCAs, PFSAs and sulfonamides being overestimated by a factor of 2.8, 2.8 and 4.2, respectively, whereas fluorotelomers would be underestimated by a factor of 25. A median of 4% of the PFAS mass in consumer products is currently subject to the Stockholm Convention (n = 976), increasing to 18% with the inclusion of candidate PFAS (PFCAs with FCL ≥ 7) (n = 976). The average amount of long-chain PFAS within this dataset, including PFCAs, is 66% (n = 976), indicating that long-chain PFAS are dominant in consumer products.
PFAS concentrations are in ng ml−1 or µg kg−1 equivalent to ppb. Box dimensions show the span between quartiles 1 and 3 (IQR). Outliers are defined as values greater than 1.5× the IQR. Whiskers extend from these quartiles to the largest (quartile 3) or smallest (quartile 1) non-outlier value (that is, <1.5× the IQR).
As previously mentioned, fluorotelomers represent the largest contributor to PFAS mass in consumer products. Fluorotelomers are comprised of numerous subgroups including fluorotelomer sulfonates (FTS), fluorotelomer alcohols (FTOH), fluorotelomer iodides, fluorotelomer acrylates, fluorotelomer methacrylates, fluorotelomer mercaptoalkyl phosphate diester, fluorotelomer unsaturated carboxylic acids (FTUCA) and fluorotelomer carboxylic acids (FTCA). FTS represent a median 2% (n = 338) of the total PFAS in consumer products when two or more PFAS classes are quantified and are the only fluorotelomers quantified using the US EPA methods. FTOH require a different analytical method to most other PFAS and were not often analysed. However, when two or more PFAS in this subclass were quantified, they represented an important proportion (median of 58% (n = 365)) of the total PFAS in consumer products.
Although most PFAS in consumer products may not be currently regulated, many will transform to regulated PFAS in the environment (Supplementary Tables 1 and 2). Studies that have used the total oxidizable precursor (TOP) assay found a notable increase in PFCAs following oxidation. This suggests that traditional EPA-based methods do not adequately capture PFAS embodied in consumer products and their potential environmental burden24,25,26,27.
AFFF
Eleven literature studies characterize PFAS in 148 AFFF samples from different suppliers and synthesis methods sold since 1980. These studies quantified 69 PFAS with a maximum of 40 PFAS being measured in any given study28. PFAS for AFFF applications have been synthesized by two synthesis processes: electrochemical fluorination and telomerization21. These processes result in a range of products with electrochemical fluorination-producing PFOS and telomerization-producing fluorotelomers21,29. Depending on the manufacturer and year produced, AFFF has different formulations (Supplementary Table 3). PFOS represents a median 51% of the PFAS in historic 3M AFFF (n = 14), with other PFSAs and sulfonamides also forming important contributions. All other PFAS in historic 3M AFFF had low concentrations, when measured. Fluorotelomers and PFCAs, were the dominant PFAS in Angus AFFF (n = 28), with a median of 64% and 36%, respectively. Several other AFFF have been investigated, however, the supplier’s name was not provided or PFCA and PFSA concentrations were not quantified. In these samples, fluorotelomers represented the dominant PFAS (median = 93%, n = 83). Of these fluorotelomers, important subclasses include FTS (median = 73% of total PFAS, n = 69) and FTOH (median = 10% of total PFAS, n = 38). Comparison of PFAS quantified using EPA method 1633 to the sum of all PFAS quantified suggests that exclusively reporting PFAS quantified using EPA method 1633 underrepresents total PFAS in AFFF by a median factor of 2.8. A median 60% of the PFAS mass in historic 3M AFFF is subject to the Stockholm Convention whereas Angus AFFF has no PFAS subject to the Stockholm Convention. For non-3M AFFF (n = 134), including candidate PFAS, 0.6% of the PFAS mass would be subject to the Stockholm Convention, increasing to 1% if long-chain PFAS are considered. This analysis of AFFF formulations suggests that known PFAS in AFFF presents a large environmental burden, with an important fraction either currently subject to regulatory oversight, or likely in future. However, an undetected fraction of PFAS in AFFF probably exists30. It is important to note that many of these studies quantify a limited number of PFAS, similar to non-AFFF product studies. Therefore, it is challenging to predict the AFFF environment burden because not all PFAS are quantified. Furthermore, when the TOP assay is applied to AFFF samples, considerable increases in total PFAS mass has been reported31,32, as noted in non-AFFF consumer product studies.
Finding the missing piece in FTOH and other under measured PFAS
Across the 33,940 groundwater samples, 57 distinct PFAS were quantified. On average, 16 distinct PFAS (maximum of 38 PFAS) were quantified and an average of 15 PFAS within the suite of proposed US EPA method 1633. PFCAs, PFSAs and sulfonamides were routinely quantified (at least two PFCAs, PFSAs and sulfonamides were quantified in 91%, 89% and 54% of studies, respectively). Whereas at least two fluorotelomers were quantified in 26% of the groundwater studies, this was almost exclusively FTS, with FTCA and FTUCA quantified to a lesser extent and no studies quantifying FTOH. This is despite the fact that FTOH are an important PFAS present in consumer products, when quantified. It is important to note that existing EPA aqueous methods (EPA methods 533, 537, 1633) are liquid chromatography with tandem mass sprectrometry (LC-MS/MS) based. Analysis of FTOH requires gas chromatography tandem mass spectrometry (GC-MS/MS), with no US EPA GC/MS/MS methods for aqueous PFAS in existence. With regards to surface water, PFCAs, PFSAs and fluorotelomers were quantified to a similar extent as groundwater samples, with FTS representing the dominant fluorotelomers quantified. Unlike groundwater studies, four of the surface water studies quantified FTOH33,34,35,36, with only two also quantifying PFCAs, PFSAs or both, facilitating an assessment of the relative importance of FTOH. In the 16 urban river samples in China34 and eight river samples in Bangladesh33, FTOH represented a median of 53% of the total PFAS (range of 46 to 62%) and 2% (ranging from 0.9 to 34%), respectively. It is difficult to draw definitive conclusions from two studies with relatively few samples, however, coupled with the FTOH prevalence in consumer products, it suggests that FTOH could be an important class of unquantified PFAS. Because only a limited suite of PFAS are typically quantified, any estimate of PFAS environmental burden is likely to be an underestimate, and a broader suite of PFAS needs to be quantified.
Wastewater treatment plants (WWTPs) and landfills are focal point receptors of anthropogenic activity. Hence, representing an opportunity for quantification of the diverse PFAS suite that has or may be dispersed into the environment. Unfortunately, studies investigating WWTP influent and landfill leachate provide limited insights. Whereas landfill leachate studies quantify more PFAS than surface and groundwater studies, they have focused on the same range of PFAS (PFCAs, PFSAs, FTS and select sulfonamides) with no studies directly measuring FTOH37. However, studies have reported atmospheric FTOH emissions at landfill sites and WWTPs38. One Chinese study reported FTOH represented 8% of the PFAS WWTP influent mass39. FTOH could enter the wastewater system through various sources, including laundering of textiles40.
Studies using the TOP assay to WWTP effluent report a considerable PFAS fraction that go undetected using EPA methods41,42. Similarly, studies that oxidized landfill leachate reported minimum to moderate changes in PFAS concentrations, suggesting that unknown PFAS transformed biotically or abiotically in landfill cells43,44. Whereas limited studies have applied the TOP assay to surface and groundwater, some report considerable increases in PFAS concentrations, although the increases are not consistent in the literature41,45,46. A major drawback of the TOP assay is that not all PFAS undergo oxidation to PFCAs or PFSA, particularly the perfluoroether class which transform into unmonitored terminal PFAS47. Furthermore, there is no standardized TOP assay method, and results from the variants available can differ greatly, with too harsh conditions leading to mineralization of terminal target PFAS48. These findings suggest that TOP assay results may underrepresent future PFASʼ environmental burden. Given the relatively limited suite of PFAS that have been quantified in surface and groundwater, it is not possible to reliably discuss the extent to which current PFAS methods adequately capture the range of PFAS and mass in these systems.
Overall, this study suggests that a large fraction of surface and groundwaters globally exceed PFAS international advisories and regulations and that future PFAS environmental burden is likely underestimated. Because PFAS definition continues to evolve, the extent of underestimation will be a function of PFAS definition. Additional work is needed to develop analytical techniques to quantify PFAS in environmental matrices, conduct a more systematic sampling regime of water sources globally and quantify human and ecological impacts of the broad range of PFAS in the environment.
Methods
This study reviewed and collated 48,985 samples from 367 published papers and government websites to build a comprehensive database to determine PFAS global distribution in surface and groundwater (Supplementary Table 4). This study is therefore limited to PFAS tested in previous studies, the analytical instruments and methods used and the locations that were sampled. The data were collated, compared and analysed and statistically validated using Python scripts and MS Excel.
PFAS is reported in ng l−1 for aqueous concentrations. When investigating PFAS concentrations in products, all data were converted to parts per billion (ppb) using appropriate area to mass conversions as the data include PFAS from an array of sources in different compartments and measured with different instruments and sample-preparation techniques.
The data available were converted into an Excel file using an online open-source portable document format converter when required. All data was then saved as a comma-separated values or Microsoft Excel spreadsheet document before analysis with Python. To check the data, an initial screening was done using a Python script, followed by manual checks. When analytes were reported as below detection limits (BDL) or not detected, a random value between 0 and the detection limit was assigned using a loop in Python and the detection limit provided in each study. Even though there are specific statistical methods for handling censored data, they assume a specific data distribution not applicable in this case and as there are less than 60% of samples below the detection limit, substitution was suitable49. Randomizing the substitution reduces clustering of data around a specific value and biasing of results. To represent data on a map, the latitude and longitude of the sampling location was used. Where no location was specified other than the country, a random major city in that country was assigned to capture the sample’s location.
A list of the PFAS analytes, their major PFAS class and fluorinated chain length are included in Supplementary Table 1. The PFAS classes considered include those that form as terminal products, that is, perfluorocarboxylates (PFCA), perfluorosulfonates (PFSA) and precursors to these terminal products. Precursors included are fluorotelomers, sulfonamides and polyfluorinated alkyl phosphate esters (PAPs). Within the fluorotelomer PFAS class subclasses include: alcohols (FTOH), sulfonates (FTS), iodides, n:2 saturated/unsaturated carboxylates (FTCA/FTUCA), acrylates and betaines. Finally, novel PFAS (which predominantly encapsulate the ether PFAS sub-group) were considered.
Data availability
Sources of data used to compile the database are provided in Supplementary Table 4. The data analysed and used to generate the figures and tables in this study are available in the following Zenodo data repository: https://doi.org/10.5281/zenodo.10616840. Source data are provided with this paper.
Code availability
Python scripts used to summarize data will be provided upon request.
References
PFAS Structures in DSSTox (US EPA, 2022); https://comptox.epa.gov/dashboard/chemical-lists/PFASSTRUCTV5
OECD Reconciling Terminology of the Universe of Per- and Polyfluoroalkyl Substances Recommendations and Practical Guidance Series on Risk Management No. 61 (OECD Publishing, 2021).
Spaan, K. M., Seilitz, F., Plassmann, M. M., de Wit, C. A. & Benskin, J. P. Pharmaceuticals account for a significant proportion of the extractable organic fluorine in municipal wastewater treatment plant sludge. Environ. Sci. Technol. Lett. 10, 328–336 (2023).
Cousins, I. T. et al. The high persistence of PFAS is sufficient for their management as a chemical class. Environ. Sci. Processes Impacts 22, 2307–2312 (2020).
PFASs Listed Under the Stockholm Convention (UN Environment Programme, 2019); https://chm.pops.int/Implementation/IndustrialPOPs/PFAS/Overview/tabid/5221/Default.aspx
Panieri, E., Baralic, K., Djukic-Cosic, D., Buha Djordjevic, A. & Saso, L. PFAS molecules: a major concern for the human health and the environment. Toxics 10, 44 (2022).
Per- and Polyfluoroalkyl Substances (PFAS) in Drinking Water (Health Canada, 2023); https://www.canada.ca/en/health-canada/services/environmental-workplace-health/reports-publications/water-quality/water-talk-per-polyfluoroalkyl-substances-drinking-water.html
Official Journal of the European Union (European Parliament & Council of the European Union, 2020).
Draft Objective for Per- and Polyfluoroalkyl Substances in Canadian Drinking Water: Rationale (Health Canada, 2023); https://www.canada.ca/en/health-canada/programs/consultation-draft-objective-per-polyfluoroalkyl-substances-canadian-drinking-water/rationale.html
Per- and Polyfluoroalkyl Substances (PFAS) Proposed PFAS National Primary Drinking Water Regulation (US EPA, 2023); https://www.epa.gov/sdwa/and-polyfluoroalkyl-substances-pfas
Chambers, W. S., Hopkins, J. G. & Richards, S. M. A review of per- and polyfluorinated alkyl substance impairment of reproduction. Front. Toxicol. 3, 732436 (2021).
Fenton, S. E. et al. Per- and polyfluoroalkyl substance toxicity and human health review: current state of knowledge and strategies for informing future research. Environ. Toxicol. Chem. 40, 606–630 (2021).
Huang, M. C. et al. Toxicokinetics of perfluorobutane sulfonate (PFBS), perfluorohexane-1-sulphonic acid (PFHxS), and perfluorooctane sulfonic acid (PFOS) in male and female Hsd:Sprague Dawley SD rats after intravenous and gavage administration. Toxicol. Rep. 6, 645–655 (2019).
History and Use of Per- and Polyfluoroalkyl Substances (PFAS) (ITRC, 2017); https://pfas-1.itrcweb.org/fact_sheets_page/PFAS_Fact_Sheet_History_and_Use_April2020.pdf
Hees, P. Analysis of the Unknown Pool of PFAS: Total Oxidizable Precursors (TOP), PFOS Precursor (PreFOS) and Telomer Degradation (Eurofins, 2017); https://www.eurofins.se/media/1568225/top_precursor_short_facts_170613.pdf
Wang, Z., Cousins, I. T., Scheringer, M., Buck, R. C. & Hungerbühler, K. Global emission inventories for C4-C14 perfluoroalkyl carboxylic acid (PFCA) homologues from 1951 to 2030, part I: production and emissions from quantifiable sources. Environ. Int. 70, 62–75 (2014).
Prevedouros, K., Cousins, I. T., Buck, R. C. & Korzeniowski, S. H. Sources, fate and transport of perfluorocarboxylates. Environ. Sci. Technol. 40, 32–44 (2006).
Cousins, I. T., Johansson, J. H., Salter, M. E., Sha, B. & Scheringer, M. Outside the safe operating space of a new planetary boundary for per- and polyfluoroalkyl substances (PFAS). Environ. Sci. Technol. 56, 11172–11179 (2022).
Hu, X. C. et al. Detection of poly- and perfluoroalkyl substances (PFASs) in U.S. drinking water linked to industrial sites, military fire training areas, and wastewater treatment plants. Environ. Sci. Technol. Lett. 3, 344–350 (2016).
Calore, F. et al. Legacy and novel PFASs in wastewater, natural water, and drinking water: occurrence in western countries vs China. Emerging Contam. 9, 100228 (2023).
Buck, R. C. et al. Perfluoroalkyl and polyfluoroalkyl substances in the environment: terminology, classification, and origins. Integr. Environ. Assess. Manage. 7, 513–541 (2011).
Per- and Poly-fluoroalkyl Substances (PFAS) Fact Sheet (Australian Government, 2016); https://www.health.gov.au/resources/publications/enhealth-guidance-per-and-polyfluoroalkyl-substances-pfas?language=en
PFAS Contamination FAQs (Australian Government, 2023); https://www.pfas.gov.au/about-pfas/faq
Robel, A. E. et al. Closing the mass balance on fluorine on papers and textiles. Environ. Sci. Technol. 51, 9022–9032 (2017).
Mumtaz, M. et al. Screening of textile finishing agents available on the Chinese market: an important source of per- and polyfluoroalkyl substances to the environment. Front. Environ. Sci. Eng. 13, 67 (2019).
Zhu, H. & Kannan, K. Total oxidizable precursor assay in the determination of perfluoroalkyl acids in textiles collected from the United States. Environ. Pollut. 265, 114940 (2020).
Chinthakindi, S., Zhu, H. & Kannan, K. An exploratory analysis of poly- and per-fluoroalkyl substances in pet food packaging from the United States. Environ. Technol. Innov. 21, 101247 (2021).
Favreau, P. et al. Multianalyte profiling of per- and polyfluoroalkyl substances (PFASs) in liquid commercial products. Chemosphere 171, 491–501 (2017).
Anderson, R. H., Long, G. C., Porter, R. C. & Anderson, J. K. Occurrence of select perfluoroalkyl substances at US Air Force aqueous film-forming foam release sites other than fire-training areas: field-validation of critical fate and transport properties. Chemosphere 150, 678–685 (2016).
Liu, M. et al. Hunting the missing fluorine in aqueous film-forming foams containing per- and polyfluoroalkyl substances. J. Hazard. Mater. 464, 133006 (2024).
Dauchy, X., Boiteux, V., Bach, C., Rosin, C. & Munoz, J.-F. Per- and polyfluoroalkyl substances in firefighting foam concentrates and water samples collected near sites impacted by the use of these foams. Chemosphere 183, 53–61 (2017).
Place, B. J. & Field, J. A. Identification of novel fluorochemicals in aqueous film-forming foams used by the US military. Environ. Sci. Technol. 46, 7120–7127 (2012).
Morales-McDevitt, M. E. et al. Poly- and perfluorinated alkyl substances in air and water from Dhaka, Bangladesh. Environ. Toxicol. Chem. 41, 334–342 (2022).
Si, Y. et al. Occurrence and ecological risk assessment of perfluoroalkyl substances (PFASs) in water and sediment from an urban river in South China. Arch. Environ. Contam. Toxicol. 81, 133–141 (2021).
Mahmoud, M. A. M., Kärrman, A., Oono, S., Harada, K. H. & Koizumi, A. Polyfluorinated telomers in precipitation and surface water in an urban area of Japan. Chemosphere 74, 467–472 (2009).
Xie, Z. et al. Neutral poly- and perfluoroalkyl substances in air and seawater of the North Sea. Environ. Sci. Pollut. Res. 20, 7988–8000 (2013).
Coffin, E. S., Reeves, D. M. & Cassidy, D. P. PFAS in municipal solid waste landfills: sources, leachate composition, chemical transformations, and future challenges. Curr. Opin. Environ. Sci. Health 31, 100418 (2023).
Ahrens, L. et al. Wastewater treatment plant and landfills as sources of polyfluoroalkyl compounds to the atmosphere. Environ. Sci. Technol. 45, 8098–8105 (2011).
Annunziato Kate, M. et al. Chemical characterization of a legacy aqueous film-forming foam sample and developmental toxicity in zebrafish (Danio rerio). Environ. Health Perspect. 128, 097006 (2020).
van der Veen, I. et al. Fate of per- and polyfluoroalkyl substances from durable water-repellent clothing during use. Environ. Sci. Technol. 56, 5886–5897 (2022).
Ye, F. et al. Spatial distribution and importance of potential perfluoroalkyl acid precursors in urban rivers and sewage treatment plant effluent—case study of Tama River, Japan. Water Res. 67, 77–85 (2014).
Tavasoli, E., Luek, J. L., Malley, J. P. & Mouser, P. J. Distribution and fate of per- and polyfluoroalkyl substances (PFAS) in wastewater treatment facilities. Environ. Sci. Processes Impacts 23, 903–913 (2021).
Rehnstam, S., Czeschka, M.-B. & Ahrens, L. Suspect screening and total oxidizable precursor (TOP) assay as tools for characterization of per- and polyfluoroalkyl substance (PFAS)-contaminated groundwater and treated landfill leachate. Chemosphere 334, 138925 (2023).
Chen, Y. et al. Concentrations of perfluoroalkyl and polyfluoroalkyl substances before and after full-scale landfill leachate treatment. Waste Manage. 153, 110–120 (2022).
Martin, D. et al. Zwitterionic, cationic, and anionic perfluoroalkyl and polyfluoroalkyl substances integrated into total oxidizable precursor assay of contaminated groundwater. Talanta 195, 533–542 (2019).
Neuwald, I. J. et al. Ultra-short-chain PFASs in the sources of German drinking water: prevalent, overlooked, difficult to remove, and unregulated. Environ. Sci. Technol. 56, 6380–6390 (2022).
Zhang, C., Hopkins, Z. R., McCord, J., Strynar, M. J. & Knappe, D. R. U. Fate of per- and polyfluoroalkyl ether acids in the total oxidizable precursor assay and implications for the analysis of impacted water. Environ. Sci. Technol. Lett. 6, 662–668 (2019).
Hutchinson, S., Rieck, T. & Wu, X. Advanced PFAS precursor digestion methods for biosolids. Environ. Chem. 17, 558–567 (2020).
Helsel, D. R. Fabricating data: how substituting values for nondetects can ruin results, and what can be done about it. Chemosphere 65, 2434–2439 (2006).
Acknowledgements
The Australian Government Research Training Program (RTP) Scholarship is acknowledged for the provision of candidature funding for D.A.G. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. All other authors received no specific funding for this work.
Funding
Open access funding provided through UNSW Library.
Author information
Authors and Affiliations
Contributions
Conceptualization: conceived by D.M.O. and refined by all authors. Methodology: D.A.G., D.G., A.M.J., T.C.G.K. and D.M.O. Data collection: D.A.G., D.G. and J.H. Data analysis: D.A.G., D.G., A.M.J., T.C.G.K. and D.M.O. Validation: D.A.G., D.G. and A.M.J. Supervision: A.M.J., M.J.L. and D.M.O. Writing: D.A.G., A.M.J. and D.M.O. with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Geoscience thanks Mark Strynar and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Xujia Jiang, in collaboration with the Nature Geoscience team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Cumulative distribution of surface water samples from an unknown source that exceed a given PFAS concentration.
Circles indicate relevant PFAS drinking water guidance values. For samples where PFAS concentrations were below detection limits a PFAS concentration was randomly assigned between zero the detection limit.
Extended Data Fig. 2 Cumulative distribution of surface water samples from a known non AFFF source that exceed a given PFAS concentration.
Circles indicate relevant PFAS drinking water guidance values. For samples where PFAS concentrations were below detection limits a PFAS concentration was randomly assigned between zero the detection limit.
Extended Data Fig. 3 Cumulative distribution of surface water samples from a known AFFF source that exceed a given PFAS concentration.
Circles indicate relevant PFAS drinking water guidance values. For samples where PFAS concentrations were below detection limits a PFAS concentration was randomly assigned between zero the detection limit.
Extended Data Fig. 4 Cumulative distribution of groundwater samples from an unknown source that exceed a given PFAS concentration.
Circles indicate relevant PFAS drinking water guidance values. For samples where PFAS concentrations were below detection limits a PFAS concentration was randomly assigned between zero the detection limit.
Extended Data Fig. 5 Cumulative distribution of groundwater samples from a known non AFFF source that exceed a given PFAS concentration.
Circles indicate relevant PFAS drinking water guidance values. For samples where PFAS concentrations were below detection limits a PFAS concentration was randomly assigned between zero the detection limit.
Extended Data Fig. 6 Cumulative distribution of groundwater samples from a known AFFF source that exceed a given PFAS concentration.
Circles indicate relevant PFAS drinking water guidance values. For samples where PFAS concentrations were below detection limits a PFAS concentration was randomly assigned between zero the detection limit.
Supplementary information
Supplementary Information
Supplementary Tables 1–4 and Figs. 1–9.
Supplementary Data 1
Source data for Supplementary Figs. 1–9.
Source data
Source Data
Statistical source data for Figs. 1–4.
Source Data Extended Data
Statistical source data for Extended Data Figs. 1–6.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ackerman Grunfeld, D., Gilbert, D., Hou, J. et al. Underestimated burden of per- and polyfluoroalkyl substances in global surface waters and groundwaters. Nat. Geosci. 17, 340–346 (2024). https://doi.org/10.1038/s41561-024-01402-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-024-01402-8
This article is cited by
-
The link between ancient microbial fluoride resistance mechanisms and bioengineering organofluorine degradation or synthesis
Nature Communications (2024)
-
Pervasive fluorinated chemicals
Nature Geoscience (2024)
-
Concentrations of Per-and Polyfluoroalkyl Substances (PFAS) and Pancreatic Cancer: A Case–Control Study in New York
Exposure and Health (2024)