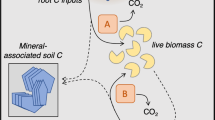
Abstract
Soil organic matter is the dominant carbon pool in terrestrial ecosystems, and its management is of increasing policy relevance. Soil microbes are the main drivers of soil organic carbon sequestration, especially through accumulation of their necromass. However, since the direct characterization of microbial necromass in soil is challenging, its composition and formation remain unresolved. Here we provide evidence that microbial death pathways (the distinct processes of microbial dying) in soil affect necromass composition and its subsequent fate. Importantly, the composition of derived microbial necromass does not equal that of microbial biomass. From biomass to necromass, distinct chemical transformations lead to increases in cell wall/cytoplasm ratios while nutrient contents and easily degradable compounds are depleted. The exact changes depend on environmental conditions and the relevance of different microbial death pathways, for example, predation, starvation or anthropogenic stresses. This has far-reaching consequences for mechanisms underpinning biogeochemical processes: (1) the quantity and persistence of microbial necromass is governed by microbial death pathways, not only the initial biomass composition; (2) efficient recycling of nutrients within microbial biomass presents a possible pathway of organic carbon sequestration that minimizes nitrogen losses; (3) human-induced disturbances affect the causes of microbial death and consequently necromass composition. Thus, new research focusing on microbial death pathways holds great potential to improve management strategies for soil organic carbon storage. Not only microbial growth but also death drive the soil microbial carbon pump.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Bradford, M. A. et al. Soil carbon science for policy and practice. Nat. Sustain. 2, 1070–1072 (2019).
Lehmann, J. & Kleber, M. The contentious nature of soil organic matter. Nature 528, 60–68 (2015).
Liang, C., Schimel, J. P. & Jastrow, J. D. The importance of anabolism in microbial control over soil carbon storage. Nat. Microbiol. 2, 17105 (2017).
Liang, C., Amelung, W., Lehmann, J. & Kästner, M. Quantitative assessment of microbial necromass contribution to soil organic matter. Glob. Change Biol. 25, 3578–3590 (2019).
Wang, B. R., An, S. S., Liang, C., Liu, Y. & Kuzyakov, Y. Microbial necromass as the source of soil organic carbon in global ecosystems. Soil Biol. Biochem. 162, 108422 (2021).
Kästner, M. & Miltner, A. in The Future of Soil Carbon (eds Garcia, C. et al.) Ch. 5 (Academic Press, 2018).
Buckeridge, K. M. et al. Sticky dead microbes: rapid abiotic retention of microbial necromass in soil. Soil Biol. Biochem. 149, 107929 (2020).
Kallenbach, C. M., Grandy, A. S., Frey, S. D. & Diefendorf, A. F. Microbial physiology and necromass regulate agricultural soil carbon accumulation. Soil Biol. Biochem. 91, 279–290 (2015).
Kallenbach, C. M., Frey, S. D. & Grandy, A. S. Direct evidence for microbial-derived soil organic matter formation and its ecophysiological controls. Nat. Commun. 7, 13630 (2016).
Emerson, J. B. et al. Schrödinger’s microbes: tools for distinguishing the living from the dead in microbial ecosystems. Microbiome 5, 86 (2017).
Zhang, Y. et al. Simulating measurable ecosystem carbon and nitrogen dynamics with the mechanistically defined MEMS 2.0 model. Biogeosciences 18, 3147–3171 (2021).
Ackermann, M., Stearns Stephen, C. & Jenal, U. Senescence in a bacterium with asymmetric division. Science 300, 1920–1920 (2003).
Aguilaniu, H., Gustafsson, L., Rigoulet, M. & Nyström, T. Asymmetric inheritance of oxidatively damaged proteins during cytokinesis. Science 299, 1751–1753 (2003).
Maheshwari, R. & Navaraj, A. Senescence in fungi: the view from Neurospora. FEMS Microbiol. Lett. 280, 135–143 (2008).
See, C. R. et al. Hyphae move matter and microbes to mineral microsites: integrating the hyphosphere into conceptual models of soil organic matter stabilization. Glob. Change Biol. 28, 2527–2540 (2022).
Pusztahelyi, T. et al. Comparative studies of differential expression of chitinolytic enzymes encoded by chiA, chiB, chiC and nagA genes in Aspergillus nidulans. Folia Microbiologica 51, 547–554 (2006).
Bartoszewska, M. & Kiel, J. A. The role of macroautophagy in development of filamentous fungi. Antioxid. Redox Signal. 14, 2271–2287 (2011).
Josefsen, L. et al. Autophagy provides nutrients for nonassimilating fungal structures and is necessary for plant colonization but not for infection in the necrotrophic plant pathogen Fusarium graminearum. Autophagy 8, 326–337 (2012).
Heaton, L. L., Jones, N. S. & Fricker, M. D. Energetic constraints on fungal growth. Am. Nat. 187, E27–E40 (2016).
Taiz, L. & Zeiger, E. Plant Physiology 4th edn (Spektrum Akademischer Verlag, 2008).
Bowman, E. J. & Bowman, B. J. in Cellular and Molecular Biology of Filamentous Fungi (eds Borkovich, K. & Ebbole, D.) 179–190 (ASM Press, 2010).
Voigt, O. & Pöggeler, S. Self-eating to grow and kill: autophagy in filamentous ascomycetes. Appl. Microbiol. Biotechnol. 97, 9277–9290 (2013).
Grimmett, I. J., Shipp, K. N., Macneil, A. & Barlocher, F. Does the growth rate hypothesis apply to aquatic hyphomycetes? Fungal Ecol. 6, 493–500 (2013).
Camenzind, T., Philipp Grenz, K., Lehmann, J. & Rillig, M. C. Soil fungal mycelia have unexpectedly flexible stoichiometric C:N and C:P ratios. Ecol. Lett. 24, 208–218 (2021).
Mason-Jones, K., Robinson, S. L., Veen, G. F., Manzoni, S. & van der Putten, W. H. Microbial storage and its implications for soil ecology. ISME J. 16, 617–629 (2022).
Gow, N. A. R., Latge, J. P. & Munro, C. A. The fungal cell wall: structure, biosynthesis, and function. Microbiol. Spectr. 5, FUNK-0035–2016 (2017).
Steiner, U. K. Senescence in bacteria and its underlying mechanisms. Front. Cell Dev. Biol. 9, 668915 (2021).
Allocati, N., Masulli, M., Di Ilio, C. & De Laurenzi, V. Die for the community: an overview of programmed cell death in bacteria. Cell Death Dis. 6, e1609 (2015).
Peeters, S. H. & de Jonge, M. I. For the greater good: programmed cell death in bacterial communities. Microbiol. Res. 207, 161–169 (2018).
Wang, J. & Bayles, K. W. Programmed cell death in plants: lessons from bacteria? Trends Plant Sci. 18, 133–139 (2013).
Nagamalleswari, E., Rao, S., Vasu, K. & Nagaraja, V. Restriction endonuclease triggered bacterial apoptosis as a mechanism for long time survival. Nucleic Acids Res. 45, 8423–8434 (2017).
Kysela, D. T., Brown, P. J. B., Huang, K. C. & Brun, Y. V. Biological consequences and advantages of asymmetric bacterial growth. Annu. Rev. Microbiol. 67, 417–435 (2013).
Bayles, K. W. Bacterial programmed cell death: making sense of a paradox. Nat. Rev. Microbiol. 12, 63–69 (2014).
Flemming, H.-C. & Wuertz, S. Bacteria and archaea on Earth and their abundance in biofilms. Nat. Rev. Microbiol. 17, 247–260 (2019).
Coleman, D. C. & Wall, D. H. in Soil Microbiology, Ecology and Biochemistry 4th edn (ed. Paul, E. A.) Ch. 5 (Academic Press, 2015).
Hungate, B. A. et al. The functional significance of bacterial predators. mBio 12, e00466-21 (2021).
Kuzyakov, Y. & Mason-Jones, K. Viruses in soil: nano-scale undead drivers of microbial life, biogeochemical turnover and ecosystem functions. Soil Biol. Biochem. 127, 305–317 (2018).
Williamson, K. E., Fuhrmann, J. J., Wommack, K. E. & Radosevich, M. Viruses in soil ecosystems: an unknown quantity within an unexplored territory. Annu. Rev. Virol. 4, 201–219 (2017).
Sokol, N. W. et al. Life and death in the soil microbiome: how ecological processes influence biogeochemistry. Nat. Rev. Microbiol. 20, 415–430 (2022).
Bonkowski, M. & Clarholm, M. J. A. P. Stimulation of plant growth through interactions of bacteria and protozoa: testing the auxiliary microbial loop hypothesis. Acta Protozool. 51, 237–247 (2012).
Potapov, A. M., Pollierer, M. M., Salmon, S., Šustr, V. & Chen, T.-W. Multidimensional trophic niche revealed by complementary approaches: gut content, digestive enzymes, fatty acids and stable isotopes in Collembola. J. Anim. Ecol. 90, 1919–1933 (2021).
Esteban, G. F. & Fenchel, T. M. in Ecology of Protozoa: The Biology of Free-living Phagotrophic Protists (eds Esteban, G. F. & Fenchel, T. M.) 33–54 (Springer, 2020).
Koksharova, O. A. Bacteria and phenoptosis. Biochemistry 78, 963–970 (2013).
Tilman, D. Resource Competition and Community Structure (Princeton Univ. Press, 1982).
Boddy, L. Interspecific combative interactions between wood-decaying basidiomycetes. FEMS Microbiol. Ecol. 31, 185–194 (2000).
Hibbing, M. E., Fuqua, C., Parsek, M. R. & Peterson, S. B. Bacterial competition: surviving and thriving in the microbial jungle. Nat. Rev. Microbiol. 8, 15–25 (2010).
Müller, S. et al. Predation by Myxococcus xanthus induces Bacillus subtilis to form spore-filled megastructures. Appl. Environ. Microbiol. 81, 203–210 (2015).
Laskowska, E. & Kuczynska-Wisnik, D. New insight into the mechanisms protecting bacteria during desiccation. Curr. Genet. 66, 313–318 (2020).
Rillig, M. C., Ryo, M. & Lehmann, A. Classifying human influences on terrestrial ecosystems. Glob. Change Biol. 27, 2273–2278 (2021).
Dörr, T., Moynihan, P. J. & Mayer, C. Bacterial cell wall structure and dynamics. Front. Microbiol. 10, 02051 (2019).
Corredor, B., Lang, B. & Russell, D. Effects of nitrogen fertilization on soil fauna in a global meta-analysis. Preprint at Res. Sq. https://doi.org/10.21203/rs.3.rs-1438491/v1 (2022).
Blankinship, J. C., Niklaus, P. A. & Hungate, B. A. A meta-analysis of responses of soil biota to global change. Oecologia 165, 553–565 (2011).
Manzoni, S., Chakrawal, A., Spohn, M. & Lindahl, B. D. Modeling microbial adaptations to nutrient limitation during litter decomposition. Front. For. Glob. Change 4, 686945 (2021).
Frank, D. et al. Effects of climate extremes on the terrestrial carbon cycle: concepts, processes and potential future impacts. Glob. Change Biol. 21, 2861–2880 (2015).
Gunina, A. & Kuzyakov, Y. From energy to (soil organic) matter. Glob. Change Biol. 28, 2169–2182 (2022).
Fernandez, C. W. & Koide, R. T. Initial melanin and nitrogen concentrations control the decomposition of ectomycorrhizal fungal litter. Soil Biol. Biochem. 77, 150–157 (2014).
Kästner, M., Miltner, A., Thiele-Bruhn, S. & Liang, C. Microbial necromass in soils—linking microbes to soil processes and carbon turnover. Front. Environ. Sci. 9, 756378 (2021).
Buckeridge, K. M., Creamer, C. & Whitaker, J. Deconstructing the microbial necromass continuum to inform soil carbon sequestration. Funct. Ecol. 36, 1396–1410 (2022).
Lehmann, J. et al. Persistence of soil organic carbon caused by functional complexity. Nat. Geosci. 13, 529–534 (2020).
Blazewicz, S. J. et al. Taxon-specific microbial growth and mortality patterns reveal distinct temporal population responses to rewetting in a California grassland soil. ISME J. 14, 1520–1532 (2020).
Kallenbach, C. M., Wallenstein, M. D., Schipanksi, M. E. & Grandy, A. S. Managing agroecosystems for soil microbial carbon use efficiency: ecological unknowns, potential outcomes, and a path forward. Front. Microbiol. 10, 1146 (2019).
Liang, C. Soil microbial carbon pump: mechanism and appraisal. Soil Ecol. Lett. 2, 241–254 (2020).
Sinsabaugh, R. L., Manzoni, S., Moorhead, D. L. & Richter, A. Carbon use efficiency of microbial communities: stoichiometry, methodology and modelling. Ecol. Lett. 16, 930–939 (2013).
van Groenigen, J. W. et al. Sequestering soil organic carbon: a nitrogen dilemma. Environ. Sci. Technol. 51, 4738–4739 (2017).
Greenlon, A. et al. Quantitative stable-isotope probing (qSIP) with metagenomics links microbial physiology and activity to soil moisture in Mediterranean-climate grassland ecosystems (in the press).
Mafla-Endara, P. M. et al. Microfluidic chips provide visual access to in situ soil ecology. Commun. Biol. 4, 889 (2021).
Schaible, G. A., Kohtz, A. J., Cliff, J. & Hatzenpichler, R. Correlative SIP-FISH-Raman-SEM-NanoSIMS links identity, morphology, biochemistry, and physiology of environmental microbes. ISME Commun. 2, 52 (2022).
See, C. R. et al. Distinct carbon fractions drive a generalisable two-pool model of fungal necromass decomposition. Funct. Ecol. 35, 796–806 (2021).
Wang, C. et al. Stabilization of microbial residues in soil organic matter after two years of decomposition. Soil Biol. Biochem. 141, 107687 (2020).
Veresoglou, S. D., Halley, J. M. & Rillig, M. C. Extinction risk of soil biota. Nat. Commun. 6, 8862 (2015).
Potapov, A. M. et al. Feeding habits and multifunctional classification of soil-associated consumers from protists to vertebrates. Biol. Rev. 97, 1057–1117 (2022).
Trap, J., Bonkowski, M., Plassard, C., Villenave, C. & Blanchart, E. Ecological importance of soil bacterivores for ecosystem functions. Plant Soil 398, 1–24 (2016).
Dooley, S. R. & Treseder, K. K. The effect of fire on microbial biomass: a meta-analysis of field studies. Biogeochemistry 109, 49–61 (2012).
Muñoz-Leoz, B., Ruiz-Romera, E., Antigüedad, I. & Garbisu, C. Tebuconazole application decreases soil microbial biomass and activity. Soil Biol. Biochem. 43, 2176–2183 (2011).
Meyer, M., Diehl, D., Schaumann, G. E. & Muñoz, K. Agricultural mulching and fungicides—impacts on fungal biomass, mycotoxin occurrence, and soil organic matter decomposition. Environ. Sci. Pollut. Res. 28, 36535–36550 (2021).
Thiery, S. & Kaimer, C. The predation strategy of Myxococcus xanthus. Front. Microbiol. 11, 2 (2020).
Laloux, G. Shedding light on the cell biology of the predatory bacterium Bdellovibrio bacteriovorus. Front. Microbiol. 10, 3136 (2020).
Acknowledgements
T.C. acknowledges funding by the Deutsche Forschungsgemeinschaft (grant number 465123751, SPP2322 SoilSystems). K.M.-J. acknowledges the Dutch Research Council (NWO) for funding of the Veni project VI.Veni.202.086. We thank S. Maaß and M. Maraun for valuable input on the feeding habits of fauna in soil.
Author information
Authors and Affiliations
Contributions
T.C. and J.L. generated the initial conceptual ideas. T.C. led the writing and literature search. K.M.-J., I.M., M.C.R., J.L. and T.C. developed the final concepts. All authors contributed to writing and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Geoscience thanks Joshua Schimel, Cynthia Kallenbach and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Xujia Jiang, in collaboration with the Nature Geoscience team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Camenzind, T., Mason-Jones, K., Mansour, I. et al. Formation of necromass-derived soil organic carbon determined by microbial death pathways. Nat. Geosci. 16, 115–122 (2023). https://doi.org/10.1038/s41561-022-01100-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-022-01100-3
This article is cited by
-
Patterns and driving factors of soil nutrient stoichiometry under three land use types in the alpine region of Tibet, China
Journal of Soils and Sediments (2024)
-
Bottom-up perspective – The role of roots and rhizosphere in climate change adaptation and mitigation in agroecosystems
Plant and Soil (2024)
-
Effects of microplastics on soil carbon pool and terrestrial plant performance
Carbon Research (2024)
-
Revisiting soil fungal biomarkers and conversion factors: Interspecific variability in phospholipid fatty acids, ergosterol and rDNA copy numbers
Soil Ecology Letters (2024)
-
Fungal residues were more sensitive to nitrogen addition than bacterial residues in a meadow grassland soil
Soil Ecology Letters (2024)