Abstract
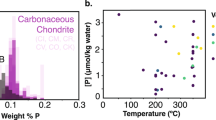
The redox chemistry of anoxic continental margin settings evolved from widespread sulfide-containing (euxinic) conditions to a global ferruginous (iron-containing) state in the early Neoproterozoic era (from ~1 to 0.8 billion years ago). Ocean redox chemistry exerts a strong control on the biogeochemical cycling of phosphorus, a limiting nutrient, and hence on primary production, but the response of the phosphorus cycle to this major ocean redox transition has not been investigated. Here, we use a geochemical speciation technique to investigate the phase partitioning of phosphorus in an open marine, early Neoproterozoic succession from the Huainan Basin, North China. We find that effective removal of bioavailable phosphorus in association with iron minerals in a globally ferruginous ocean resulted in oligotrophic (nutrient limited) conditions, and hence a probable global decrease in primary production, organic carbon burial and, subsequently, oxygen production. Nevertheless, phosphorus availability and organic carbon burial were sufficient to maintain an oxidizing atmosphere. These data imply substantial nutrient-driven variability in atmospheric oxygen levels through the Proterozoic, rather than the stable levels commonly invoked.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated and analysed for the current study are attached, and are available from data repository https://doi.org/10.5285/72c9a48f-4813-4507-9137-a97d7e6bd2d9.
References
Tyrrell, T. The relative influences of nitrogen and phosphorus on oceanic primary production. Nature 400, 525–531 (1999).
Planavsky, N. J. et al. Low mid-Proterozoic atmospheric oxygen levels and the delayed rise of animals. Science 346, 635–638 (2014).
Kump, L. R. The rise of atmospheric oxygen. Nature 451, 277–278 (2008).
Zhang, S. et al. Sufficient oxygen for animal respiration 1,400 million years ago. Proc. Natl Acad. Sci. USA 113, 1731–1736 (2016).
Lyons, T. W., Reinhard, C. T. & Planavsky, N. J. The rise of oxygen in Earth’s early ocean and atmosphere. Nature 506, 307–315 (2014).
Reinhard, C. T., Planavsky, N. J., Olson, S. L., Lyons, T. W. & Erwin, D. H. Earth’s oxygen cycle and the evolution of animal life. Proc. Natl Acad. Sci. USA 113, 8933–8938 (2016).
Zhang, K. et al. Oxygenation of the Mesoproterozoic ocean and the evolution of complex eukaryotes. Nat. Geosci. 11, 345–350 (2018).
Guilbaud, R., Poulton, S. W., Butterfield, N. J., Zhu, M. & Shields-Zhou, G. A. A global transition to ferruginous conditions in the early Neoproterozoic oceans. Nat. Geosci. 8, 466–470 (2015).
Sperling, E. A. et al. Statistical analysis of iron geochemical data suggests limited late Proterozoic oxygenation. Nature 523, 451–454 (2015).
Bjerrum, C. J. & Canfield, D. E. Ocean productivity before about 1.9 Gyr ago limited by phosphorus adsorption onto iron oxides. Nature 417, 159 (2002).
Planavsky, N. J. et al. The evolution of the marine phosphate reservoir. Nature 467, 1088–1090 (2010).
Reinhard, C. T. et al. Evolution of the global phosphorus cycle. Nature 541, 386–389 (2017).
Jones, C., Nomosatryo, S., Crowe, S. A., Bjerrum, C. J. & Canfield, D. E. Iron oxides, divalent cations, silica, and the early Earth phosphorus crisis. Geology 43, 135–138 (2015).
Konhauser, K. O., Lalonde, S. V., Amskold, L. & Holland, H. D. Was there really an Archean phosphate crisis? Science 315, 1234–1234 (2007).
Poulton, S. W. Biogeochemistry: early phosphorus redigested. Nat. Geosci. 10, 75–76 (2017).
Rivas-Lamelo, S. et al. Magnetotactic bacteria as a new model for P sequestration in the ferruginous Lake Pavin. Geochem. Perspect. Lett. 5, 35–41 (2017).
Konhauser, K. O. et al. Decoupling photochemical Fe (II) oxidation from shallow-water BIF deposition. EarthPlanet. Sci. Lett. 258, 87–100 (2007).
Zegeye, A. et al. Green rust formation controls nutrient availability in a ferruginous water column. Geology 40, 599–602 (2012).
Cosmidis, J. et al. Biomineralization of iron-phosphates in the water column of Lake Pavin (Massif Central, France). Geochim. Cosmochim. Acta 126, 78–96 (2014).
Froelich, P. N. et al. Early oxidation of organic matter in pelagic sediments of the eastern equatorial Atlantic: suboxic diagenesis. Geochim. Cosmochim. Acta 43, 1075–1090 (1979).
Ingall, E. & Jahnke, R. Influence of water-column anoxia on the elemental fractionation of carbon and phosphorus during sediment diagenesis. Mar. Geol. 139, 219–229 (1997).
Krom, M., Kress, N., Brenner, S. & Gordon, L. Phosphorus limitation of primary productivity in the eastern Mediterranean Sea. Limnol. Oceanogr. 36, 424–432 (1991).
Slomp, C. P., Thomson, J. & de Lange, G. J. Controls on phosphorus regeneration and burial during formation of eastern Mediterranean sapropels. Mar. Geol. 203, 141–159 (2004).
Dellwig, O. et al. A new particulate Mn–Fe–P-shuttle at the redoxcline of anoxic basins. Geochim. Cosmochim. Acta 74, 7100–7115 (2010).
Thompson, J. et al. Development of a modified SEDEX phosphorus speciation method for ancient rocks and modern iron-rich sediments. Chem. Geol. 524, 383–393 (2019).
Egger, M., Jilbert, T., Behrends, T., Rivard, C. & Slomp, C. P. Vivianite is a major sink for phosphorus in methanogenic coastal surface sediments. Geochim. Cosmochim. Acta 169, 217–235 (2015).
Slomp, C. P. et al. Coupled dynamics of iron and phosphorus in sediments of an oligotrophic coastal basin and the impact of anaerobic oxidation of methane. PLoS ONE 8, e62386 (2013).
Xiong, Y. et al. Phosphorus cycling in Lake Cadagno, Switzerland: a low sulfate euxinic ocean analogue. Geochim. Cosmochim. Acta 251, 116–135 (2019).
Van Cappellen, P. & Ingall, E. D. Benthic phosphorus regeneration, net primary production, and ocean anoxia: a model of the coupled marine biogeochemical cycles of carbon and phosphorus. Paleoceanography 9, 677–692 (1994).
Canfield, D. E., Raiswell, R. & Bottrell, S. H. The reactivity of sedimentary iron minerals toward sulfide. Am. J. Sci. 292, 659–683 (1992).
Dos Santos Afonso, M. & Stumm, W. Reductive dissolution of iron(III) (hydr)oxides by hydrogen sulfide. Langmuir 8, 1671–1675 (1992).
Ruttenberg, K. C. & Berner, R. A. Authigenic apatite formation and burial in sediments from non-upwelling, continental margin environments. Geochim. Cosmochim. Acta 57, 991–1007 (1993).
Planavsky, N. J. et al. Widespread iron-rich conditions in the mid-Proterozoic ocean. Nature 477, 448–451 (2011).
Poulton, S. W., Fralick, P. W. & Canfield, D. E. Spatial variability in oceanic redox structure 1.8 billion years ago. Nat. Geosci. 3, 486–490 (2010).
Poulton, S. W. & Canfield, D. E. Ferruginous conditions: a dominant feature of the ocean through Earth’s history. Elements 7, 107–112 (2011).
Tang, Q. et al. Organic-walled microfossils from the early Neoproterozoic Liulaobei Formation in the Huainan region of North China and their biostratigraphic significance. Precambrian Res. 236, 157–181 (2013).
März, C. et al. Redox sensitivity of P cycling during marine black shale formation: dynamics of sulfidic and anoxic, non-sulfidic bottom waters. Geochim. Cosmochim. Acta 72, 3703–3717 (2008).
Turekian, K. K. & Wedepohl, K. H. Distribution of the elements in some major units of the Earth’s crust. Geol. Soc. Am. Bull. 72, 175–192 (1961).
Teng, Y.-C., Primeau, F. W., Moore, J. K., Lomas, M. W. & Martiny, A. C. Global-scale variations of the ratios of carbon to phosphorus in exported marine organic matter. Nat. Geosci. 7, 895–898 (2014).
Ingall, E. D., Bustin, R. & Van Cappellen, P. Influence of water column anoxia on the burial and preservation of carbon and phosphorus in marine shales. Geochim. Cosmochim. Acta 57, 303–316 (1993).
Schrag, D. P., Higgins, J. A., Macdonald, F. A. & Johnston, D. T. Authigenic carbonate and the history of the global carbon cycle. Science 339, 540–543 (2013).
Creveling, J. R. et al. Phosphorus sources for phosphatic Cambrian carbonates. Geol. Soc. Am. Bull. 126, 145–163 (2014).
Kipp, M. A. & Stüeken, E. E. Biomass recycling and Earth’s early phosphorus cycle. Sci. Adv. 3, eaao4795 (2017).
Sperling, E. A. & Stockey, R. G. The temporal and environmental context of early animal evolution: considering all the ingredients of an “Explosion”. Integr. Comp. Biol. 58, 605–622 (2018).
Daines, S. J., Mills, B. J. & Lenton, T. M. Atmospheric oxygen regulation at low Proterozoic levels by incomplete oxidative weathering of sedimentary organic carbon. Nat. Commun. 8, 14379 (2017).
Wedepohl, K. H. The composition of the continental crust. Geochim. Cosmochim. Acta 59, 1217–1232 (1995).
Mackenzie, F., Lerman, A. & Andersson, A. Past and present of sediment and carbon biogeochemical cycling models. Biogeosci. Discuss. 1, 27–85 (2004).
Compton, J. et al. in Marine Authigenesis: From Global to Microbial (ed. Glenn, C. R.) 21–33 (Society for Sedimentary Geology, 2000).
Stolper, D. A. & Keller, C. B. A record of deep-ocean dissolved O2 from the oxidation state of iron in submarine basalts. Nature 553, 323–327 (2018).
Lenton, T. M. & Daines, S. J. Biogeochemical transformations in the history of the ocean. Annu. Rev. Mar. Sci. 9, 31–58 (2017).
Ruttenberg, K. C. Development of a sequential extraction method for different forms of phosphorus in marine sediments. Limnol. Oceanogr. 37, 1460–1482 (1992).
Acknowledgements
This work was supported by NERC (NE/I005978/1) and NSFC (41661134048) through the Co-evolution of Life and the Planet programme, through the Biosphere Evolution, Transitions and Resilience (NE/P013651) programme, and the Strategic Priority Research Program (B) of the Chinese Academy of Sciences (XDB18000000). S.W.P. and T.M.L. were supported by Royal Society Wolfson Research Merit Awards and S.W.P. by a Leverhulme Trust Fellowship.
Author information
Authors and Affiliations
Contributions
R.G. and S.W.P. devised the research. R.G., S.W.P., G.A.S., Y.Z. and M.Z. collected samples. J.T. and K.F.H. analysed the Mesoproterozoic samples. R.G. analysed the Neoproterozoic samples and interpreted the data. R.G. wrote the manuscript with significant contributions from all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Peer review information Primary Handling Editors: James Super; Xujia Jiang.
Supplementary information
Supplementary Information
Supplementary discussion, Tables 2 and 3, and Figs. 1–5.
Supplementary Table 1
Geochemical analyses
Rights and permissions
About this article
Cite this article
Guilbaud, R., Poulton, S.W., Thompson, J. et al. Phosphorus-limited conditions in the early Neoproterozoic ocean maintained low levels of atmospheric oxygen. Nat. Geosci. 13, 296–301 (2020). https://doi.org/10.1038/s41561-020-0548-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-020-0548-7
This article is cited by
-
A phosphate-rich marine reservoir in the redox stratified Ediacaran ocean
Communications Earth & Environment (2024)
-
Dynamic redox and nutrient cycling response to climate forcing in the Mesoproterozoic ocean
Nature Communications (2023)
-
Recurrent photic zone euxinia limited ocean oxygenation and animal evolution during the Ediacaran
Nature Communications (2023)
-
Earth’s Great Oxidation Event facilitated by the rise of sedimentary phosphorus recycling
Nature Geoscience (2022)
-
A nutrient control on expanded anoxia and global cooling during the Late Ordovician mass extinction
Communications Earth & Environment (2022)