Abstract
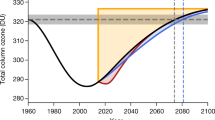
The recovery of stratospheric ozone from past depletion is underway owing to the 1987 Montreal Protocol and its subsequent amendments, which have been effective in phasing out the production and consumption of the major ozone-depleting substances (ODSs). However, there is uncertainty about the future rate of recovery. This uncertainty relates partly to unexpected emissions of controlled anthropogenic ODSs such as CCl3F and slower-than-expected declines in atmospheric CCl4. A further uncertainty surrounds emissions of uncontrolled short-lived anthropogenic ODSs (such as CH2Cl2 and CHCl3), which observations show have been increasing in the atmosphere through 2017, as well as potential emission increases in natural ODSs (such as CH3Cl and CH3Br) induced by climate change, changes in atmospheric concentrations of greenhouse gases N2O and CH4, and stratospheric geoengineering. These challenges could delay the return of stratospheric ozone levels to historical values, (for example, the abundance in 1980), by up to decades, depending on the future evolution of the emissions and other influencing factors. To mitigate the threats to future ozone recovery, it is crucial to ensure that the Montreal Protocol and its amendments continue to be implemented effectively in order to have firm control on future levels of ODSs. This action needs to be supported by an expansion of the geographic coverage of atmospheric observations of ODSs, by enhancing the ability of source attribution modelling, and by improving understanding of the interactions between climate change and ozone recovery.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Molina, M. J. & Rowland, F. S. Stratospheric sink for chlorofluoromethanes: chlorine atomic-catalysed destruction of ozone. Nature 249, 810–812 (1974).
Handbook for the Montreal Protocol on Substances that Deplete the Ozone Layer 12th edn (UNEP, 2018); http://ozone.unep.org/sites/default/files/MP_handbook-english-2018.pdf
Prinn, R. G. et al. History of chemically and radiatively important atmospheric gases from the Advanced Global Atmospheric Gases Experiment (AGAGE). Earth Syst. Sci. Data 10, 985–1018 (2018).
Engel, A. et al. Scientific Assessment of Ozone Depletion: 2018 Report No. 58, Ch. 1 (Global Ozone Research and Monitoring Project, WMO, 2018).
Kuttippurath, J., Kumar, P., Nair, P. J. & Pandey, P. C. Emergence of ozone recovery evidenced by reduction in the occurrence of Antarctic ozone loss saturation. npj Clim. Atmos. Sci. 1, 42 (2018).
Solomon, S. et al. Emergence of healing in the Antarctic ozone layer. Science 353, 269–274 (2016).
Shepherd, T. G. et al. Reconciliation of halogen-induced ozone loss with the total-column ozone record. Nat. Geosci. 7, 443–449 (2014).
Scientific Assessment of Ozone Depletion: 2018 Report No. 58 (Global Ozone Research and Monitoring Project, WMO, 2018).
Montzka, S. A. et al. An unexpected and persistent increase in global emissions of ozone-depleting CFC-11. Nature 557, 413–417 (2018).
Vollmer, M. K. et al. Atmospheric histories and emissions of chlorofluorocarbons CFC-13 (CClF3), ΣCFC-114 (C2Cl2F4), and CFC-115 (C2ClF5). Atmos. Chem. Phys. 18, 979–1002 (2018).
Laube, J. C. et al. Newly detected ozone-depleting substances in the atmosphere. Nat. Geosci. 7, 266–269 (2014).
Hossaini, R. et al. The increasing threat to stratospheric ozone from dichloromethane. Nat. Commun. 8, 15962 (2017).
Fang, X. et al. Rapid increase in ozone-depleting chloroform emissions from China. Nat. Geosci. 12, 89–93 (2019).
Chipperfield, M. P. et al. Detecting recovery of the stratospheric ozone layer. Nature 549, 211–218 (2017).
Dhomse, S. S. et al. Estimates of ozone return dates from Chemistry-Climate Model Initiative simulations. Atmos. Chem. Phys. 18, 8409–8438 (2018).
Rigby, M. et al. Increase in CFC-11 emissions from eastern China based on atmospheric observations. Nature 569, 546–550 (2019).
Carpenter, L. J. et al. Scientific Assessment of Ozone Depletion: 2018 Report No. 58, Ch. 6 (Global Ozone Research and Monitoring Project, WMO, 2018).
Scientific Assessment of Ozone Depletion: 2014 Report No. 55 (Global Ozone Research and Monitoring Project, WMO, 2014); https://www.wmo.int/pages/prog/arep/gaw/ozone_2014/full_report_TOC.html
Sherry, D., McCulloch, A., Liang, Q., Reimann, S. & Newman, P. A. Current sources of carbon tetrachloride (CCl4) in our atmosphere. Environ. Res. Lett. 13, 024004 (2018).
Lunt, M. F. et al. Continued emissions of the ozone-depleting substance carbon tetrachloride from Eastern Asia. Geophys. Res. Lett. 45, 11423–11430 (2018).
Hossaini, R. et al. Growth in stratospheric chlorine from short-lived chemicals not controlled by the Montreal Protocol. Geophys. Res. Lett. 42, 4573–4580 (2015).
Thompson, D. W. J. et al. Signatures of the Antarctic ozone hole in Southern Hemisphere surface climate change. Nat. Geosci. 4, 741–749 (2011).
Eyring, V. et al. Multi-model assessment of stratospheric ozone return dates and ozone recovery in CCMVal-2 models. Atmos. Chem. Phys. 10, 9451–9472 (2010).
Hegglin, M. I. & Shepherd, T. G. Large climate-induced changes in ultraviolet index and stratosphere-to-troposphere ozone flux. Nat. Geosci. 2, 687–691 (2009).
Butchart, N. & Scaife, A. A. Removal of chlorofluorocarbons by increased mass exchange between the stratosphere and troposphere in a changing climate. Nature 410, 799–802 (2001).
Williams, M. B., Aydin, M., Tatum, C. & Saltzman, E. S. A 2000 year atmospheric history of methyl chloride from a South Pole ice core: evidence for climate-controlled variability. Geophys. Res. Lett. 34, L07811 (2007).
Pilinis, C., King, D. B. & Saltzman, E. S. The oceans: a source or a sink of methyl bromide? Geophys. Res. Lett. 23, 817–820 (1996).
Tegtmeier, S. et al. Oceanic bromoform emissions weighted by their ozone depletion potential. Atmos. Chem. Phys. 15, 13647–13663 (2015).
Liang, Q., Strahan, S. E. & Fleming, E. L. Concerns for ozone recovery. Science 358, 1257–1258 (2017).
Global Mitigation of Non-CO 2 Greenhouse Gases: 2010–2030 (EPA, 2013); https://www.epa.gov/sites/production/files/2016-06/documents/mac_report_2013.pdf
Ravishankara, A. R., Daniel, J. S. & Portmann, R. W. Nitrous oxide (N2O): the dominant ozone-depleting substance emitted in the 21st century. Science 326, 123–125 (2009).
Kanter, D. et al. A post-Kyoto partner: considering the stratospheric ozone regime as a tool to manage nitrous oxide. Proc. Natl Acad. Sci. USA 110, 4451–4457 (2013).
Meinshausen, M. et al. The RCP greenhouse gas concentrations and their extensions from 1765 to 2300. Clim. Change 109, 213–241 (2011).
Rasch, P. J. et al. An overview of geoengineering of climate using stratospheric sulphate aerosols. Philos. Trans. R. Soc. Lond. A 366, 4007–4037 (2008).
Tilmes, S., Muller, R. & Salawitch, R. The sensitivity of polar ozone depletion to proposed geoengineering schemes. Science 320, 1201–1204 (2008).
Keith, D. W., Weisenstein, D. K., Dykema, J. A. & Keutsch, F. N. Stratospheric solar geoengineering without ozone loss. Proc. Natl Acad. Sci. USA 113, 14910–14914 (2016).
Acknowledgements
X.F. and R.G.P. were supported by NASA grant numbers NAG5-12669, NNX07AE89G, NNX11AF17G and NNX16AC98G to MIT. S.P. was supported by the National Strategic Project-Fine particle of the NRF funded by the MSIT, ME and MOHW (grant no. NRF-2017M3D8A1092225). We thank the station personnel at AGAGE stations for continuously measuring atmospheric N2O, CH2Cl2, CHCl3 and other referenced species, and R. H. Wang at the Georgia Institute of Technology for producing global monthly mean data of these species from the measurements from individual AGAGE stations. We thank Z. Dai from Harvard University for useful discussions on stratospheric geoengineering.
Author information
Authors and Affiliations
Contributions
X.F. and R.G.P. were responsible for the overall project design. All authors wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fang, X., Pyle, J.A., Chipperfield, M.P. et al. Challenges for the recovery of the ozone layer. Nat. Geosci. 12, 592–596 (2019). https://doi.org/10.1038/s41561-019-0422-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-019-0422-7
This article is cited by
-
Ozone Layer Depletion in Children’s Books Available in Greece: examining accuracy in the representation of causes of ozone layer depletion in texts
Children's Literature in Education (2023)
-
Changes in tropospheric air quality related to the protection of stratospheric ozone in a changing climate
Photochemical & Photobiological Sciences (2023)
-
Application of copper(II)-based chemicals induces CH3Br and CH3Cl emissions from soil and seawater
Nature Communications (2022)
-
Apportionment of long-term trends in different sections of total ozone column over tropical region
Environmental Monitoring and Assessment (2022)
-
An asymmetric analysis of the impacts of energy use on carbon dioxide emissions in the G7 countries
Environmental Science and Pollution Research (2021)