Abstract
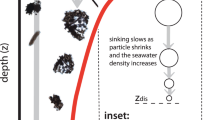
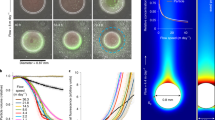
In ocean waters, anaerobic microbial respiration should be confined to the anoxic waters found in coastal regions and tropical oxygen minimum zones, where it is energetically favourable. However, recent molecular and geochemical evidence has pointed to a much broader distribution of denitrifying and sulfate-reducing microbes. Anaerobic metabolisms are thought to thrive in microenvironments that develop inside sinking organic aggregates, but the global distribution and geochemical significance of these microenvironments is poorly understood. Here, we develop a new size-resolved particle model to predict anaerobic respiration from aggregate properties and seawater chemistry. Constrained by observations of the size spectrum of sinking particles, the model predicts that denitrification and sulfate reduction can be sustained throughout vast, hypoxic expanses of the ocean, and could explain the trace metal enrichment observed in particles due to sulfide precipitation. Globally, the expansion of the anaerobic niche due to particle microenvironments doubles the rate of water column denitrification compared with estimates based on anoxic zones alone, and changes the sensitivity of the marine nitrogen cycle to deoxygenation in a warming climate.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Thamdrup, B., Dalsgaard, T. & Peter Revsbech, N. Widespread functional anoxia in the oxygen minimum zone of the eastern South Pacific. Deep Sea Res. Pt I 65, 36–45 (2012).
Froelich, P. N. et al. Early oxidation of organic matter in pelagic sediments of the eastern equatorial Atlantic: suboxic diagenesis. Geochim. Cosmochim. Acta 43, 1075–1090 (1979).
Garcia, H. E. et al. World Ocean Atlas 2009, Volume 4: Nutrients (Phosphate, Nitrate, and Silicate) NOAA Atlas NESDIS 71 (U.S. Government Printing Office, Washington, DC, 2010).
Ulloa, O., Canfield, D. E., DeLong, E. F., Letelier, R. M. & Stewart, F. J. Microbial oceanography of anoxic oxygen minimum zones. Proc. Natl Acad. Sci. USA 109, 15996–16003 (2012).
Smith, M. W., Allen, L. Z., Allen, A. E., Herfort, L. & Simon, H. M. Contrasting genomic properties of free-living and particle-attached microbial assemblages within a coastal ecosystem. Front. Microbiol. 4, 120 (2013).
Ganesh, S., Parris, D. J., DeLong, E. F. & Stewart, F. J. Metagenomic analysis of size-fractionated picoplankton in a marine oxygen minimum zone. ISME J. 8, 187–211 (2014).
Michotey, V. & Bonin, P. Evidence for anaerobic bacterial processes in the water column: denitrification and dissimilatory nitrate ammonification in the northwestern Mediterranean Sea. Mar. Ecol. Progress. Ser. 160, 47–56 (1997).
Wolgast, D., Carlucci, A. & Bauer, J. Nitrate respiration associated with detrital aggregates in aerobic bottom waters of the abyssal NE Pacific. Deep Sea Res. Pt II 45, 881–892 (1998).
Kalvelage, T. et al. Oxygen sensitivity of anammox and coupled N-cycle processes in oxygen minimum zones. PLoS ONE 6, e29299 (2011).
Fuchs, B. M., Woebken, D., Zubkov, M. V., Burkill, P. & Amann, R. Molecular identification of picoplankton populations in contrasting waters of the Arabian Sea. Aquat. Microb. Ecol. 39, 145–157 (2005).
Canfield, D. E. et al. A cryptic sulfur cycle in oxygen-minimum–zone waters off the Chilean coast. Science 330, 1375–1378 (2010).
Carolan, M. & Beman, J. M. Transcriptomic evidence for microbial sulfur cycling in the eastern tropical North Pacific oxygen minimum zone. Front. Microbiol. 6, 334 (2015).
Swan, B. K. et al. Potential for chemolithoautotrophy among ubiquitous bacteria lineages in the dark ocean. Science 333, 1296–1300 (2011).
Alldredge, A. L. & Cohen, Y. Can microscale chemical patches persist in the sea? Microelectrode study of marine snow, fecal pellets. Science 235, 689–691 (1987).
Ploug, H. & Buchholz, B. Anoxic aggregates an ephemeral phenomenon in the ocean. Aquat. Microb. Ecol. 13, 285–294 (1997).
Klawonn, I., Bonaglia, S., Brüchert, V. & Ploug, H. Aerobic and anaerobic nitrogen transformation processes in N2-fixing cyanobacterial aggregates. ISME J. 9, 1456–1466 (2015).
Ganesh, S. et al. Size-fraction partitioning of community gene transcription and nitrogen metabolism in a marine oxygen minimum zone. ISME J. 9, 2682–2696 (2015).
Shanks, A. L. & Reeder, M. L. Reducing microzones and sulfide production in marine snow. Mar. Ecol. Prog. Ser. 96, 43–47 (1993).
Waeles, M., Maguer, J.-F., Baurand, F. & Riso, R. D. Off Congo waters (Angola Basin, Atlantic Ocean): a hot spot for cadmium-phosphate fractionation. Limnol. Oceanogr. 58, 1481–1490 (2013).
Janssen, D. J. et al. Undocumented water column sink for cadmium in open ocean oxygen-deficient zones. Proc. Natl Acad. Sci. USA 111, 6888–6893 (2014).
Simon, M., Grossart, H.-P., Schweitzer, B. & Ploug, H. Microbial ecology of organic aggregates in aquatic ecosystems. Aquat. Microb. Ecol. 28, 175–211 (2002).
Burd, A. B. & Jackson, G. A. Particle aggregation. Annu. Rev. Mar. Sci. 1, 65–90 (2009).
Lam, P. J. & Marchal, O. Insights into particle cycling from thorium and particle data. Annu. Rev. Mar. Sci. 7, 159–184 (2015).
McDonnell, A., Boyd, P. & Buesseler, K. Effects of sinking velocities and microbial respiration rates on the attenuation of particulate carbon fluxes through the mesopelagic zone. Glob. Biogeochem. Cycles 29, 175–193 (2015).
Smayda, T. J. Normal and accelerated sinking of phytoplankton in the sea. Mar. Geol. 11, 105–122 (1971).
Jin, Q. & Bethke, C. M. Predicting the rate of microbial respiration in geochemical environments. Geochim. Cosmochim. Acta 69, 1133–1143 (2005).
Guidi, L. et al. Relationship between particle size distribution and flux in the mesopelagic zone. Deep Sea Res. Pt I 55, 1364–1374 (2008).
Durkin, C. A., Estapa, M. L. & Buesseler, K. O. Observations of carbon export by small sinking particles in the upper mesopelagic. Mar. Chem. 175, 72–81 (2015).
Ploug, H., Iversen, M. & Fischer, G. Ballast, sinking velocity and apparent diffusivity in marine snow and zooplankton fecal pellets: implications for substrate turnover by attached bacteria. Limnol. Oceanogr. 53, 1878–1886 (2008).
Picheral, M. et al. The Underwater Vision Profiler 5: an advanced instrument for high spatial resolution studies of particle size spectra and zooplankton. Limnol. Oceanogr. Methods 8, 462–473 (2010).
Weber, T., Cram, J. A., Leung, S. W., DeVries, T. & Deutsch, C. Deep ocean nutrients imply large latitudinal variation in particle transfer efficiency. Proc. Natl Acad. Sci. USA 113, 8606–8611 (2016).
Kalvelage, T. et al. Aerobic microbial respiration in oceanic oxygen minimum zones. PLoS ONE 10, e0133526 (2015).
Johnston, D. et al. Placing an upper limit on cryptic marine sulphur cycling. Nature 513, 530–533 (2014).
Martin, J. H., Knauer, G. A., Karl, D. M. & Broenkow, W. W. VERTEX: carbon cycling in the northeast Pacific. Deep Sea Res. Pt A 34, 267–285 (1987).
Ohnemus, D. C. et al. Elevated trace metal content of prokaryotic communities associated with marine oxygen deficient zones. Limnol. Oceanogr. 62, 3–25 (2017).
Lee, J.-M., Heller, M. I. & Lam, P. J. Size distribution of particulate trace elements in the US GEOTRACES Eastern Pacific Zonal Transect (GP16). Mar. Chem. (in the press).
Guidi, L. et al. Plankton networks driving carbon export in the oligotrophic ocean. Nature 532, 465–470 (2016).
DeVries, T., Deutsch, C., Primeau, F., Chang, B. & Devol, A. Global rates of water-column denitrification derived from nitrogen gas measurements. Nat. Geosci. 5, 547–550 (2012).
Deutsch, C., Gruber, N., Key, R. M., Sarmiento, J. L. & Ganachaud, A. Denitrification and N2 fixation in the Pacific Ocean. Glob. Biogeochem. Cycles 15, 483–506 (2001).
Bianchi, D., Dunne, J. P., Sarmiento, J. L. & Galbraith, E. D. Data-based estimates of suboxia,denitrification, and N2O production in the ocean and their sensitivities to dissolved O2. Glob. Biogeochem. Cycles 26, GB2009 (2012).
Elderfield, H. & Rickaby, R. Oceanic Cd/P ratio and nutrient utilization in the glacial Southern Ocean. Nature 405, 305–310 (2000).
Franck, V. M., Bruland, K. W., Hutchins, D. A. & Brzezinski, M. A. Iron and zinc effects on silicic acid and nitrate uptake kinetics in three high-nutrient, low-chlorophyll (HNLC) regions. Mar. Ecol. Progress. Ser. 252, 15–33 (2003).
Brandes, J. A. & Devol, A. H. A global marine‐fixed nitrogen isotopic budget: implications for Holocene nitrogen cycling. Glob. Biogeochem. Cycles 16, 1120 (2002).
Deutsch, C., Sigman, D. M., Thunell, R. C., Meckler, A. N. & Haug, G. H. Isotopic constraints on glacial/interglacial changes in the oceanic nitrogen budget. Glob. Biogeochem. Cycles 18, GB4012 (2004).
DeVries, T., Deutsch, C., Rafter, P. & Primeau, F. Marine denitrification rates determined from a global 3-D inverse model. Biogeosciences 10, 2481–2496 (2013).
Großkopf, T. et al. Doubling of marine dinitrogen-fixation rates based on direct measurements. Nature 488, 361–364 (2012).
Halm, H. et al. Heterotrophic organisms dominate nitrogen fixation in the South Pacific Gyre. ISME J. 6, 1238–1249 (2012).
Bopp, L. et al. Multiple stressors of ocean ecosystems in the 21st century: projections with CMIP5 models. Biogeosciences 10, 6225–6245 (2013).
Deutsch, C. et al. Centennial changes in North Pacific anoxia linked to tropical trade winds. Science 345, 665–668 (2014).
DeVries, T., Liang, J.-H. & Deutsch, C. A mechanistic particle flux model applied to the oceanic phosphorus cycle. Biogeosciences 11, 5381–5398 (2014).
Stemmann, L. et al. Volume distribution for particles between 3.5 to 2000 µm in the upper 200m region of the South Pacific Gyre. Biogeosciences 5, 299–310 (2008).
Jokulsdottir, T. & Archer, D. A stochastic, Lagrangian model of sinking biogenic aggregates in the ocean (SLAMS 1.0): model formulation, validation and sensitivity. Geosci. Model Dev. 9, 1455–1476 (2016).
Passow, U. & Carlson, C. A. The biological pump in a high CO2 world. Mar. Ecol. Progress. Ser. 470, 249–271 (2012).
Louca, S. et al. Integrating biogeochemistry with multiomic sequence information in a model oxygen minimum zone. Proc. Natl Acad. Sci. USA 113, E5925–E5933 (2016).
Ploug, H., Hietanen, S. & Kuparinen, J. Diffusion and advection within and around sinking, porous diatom aggregates. Limnol. Oceanogr. 47, 1129–1136 (2002).
Litchman, E., Klausmeier, C. A., Schofield, O. M. & Falkowski, P. G. The role of functional traits and trade-offs in structuring phytoplankton communities: scaling from cellular to ecosystem level. Ecol. Lett. 10, 1170–1181 (2007).
Mullin, M., Sloan, P. & Eppley, R. Relationship between carbon content, cell volume, and area in phytoplankton. Limnol. Oceanogr. 11, 307–311 (1966).
Alldredge, A. The carbon, nitrogen and mass content of marine snow as a function of aggregate size. Deep Sea Res. Pt I 45, 529–541 (1998).
Ploug, H. & Grossart, H.-P. Bacterial growth and grazing on diatom aggregates: respiratory carbon turnover as a function of aggregate size and sinking velocity. Limnol. Oceanogr. 45, 1467–1475 (2000).
Iversen, M. H., Nowald, N., Ploug, H., Jackson, G. A. & Fischer, G. High resolution profiles of vertical particulate organic matter export off Cape Blanc, Mauritania: degradation processes and ballasting effects. Deep Sea Res. Pt I 57, 771–784 (2010).
Lee, Z. et al. Euphotic zone depth: its derivation and implication to ocean‐color remote sensing. J. Geophys. Res. Oceans 112, C03009 (2007).
Garcia, H. et al. World Ocean Atlas 2009, Volume 3: Dissolved Oxygen, Apparent Oxygen Utilization, and Oxygen Saturation NOAA Atlas NESDIS 70 (U.S. Government Printing Office, Washington, DC, 2010).
Behrenfeld, M. J. & Falkowski, P. G. Photosynthetic rates derived from satellite-based chlorophyll concentration. Limnol. Oceanogr. 42, 1–20 (1997).
Carr, M.-E. et al. A comparison of global estimates of marine primary production from ocean color. Deep Sea Res. Pt II 53, 741–770 (2006).
Behrenfeld, M. J., Boss, E., Siegel, D. A. & Shea, D. M. Carbon-based ocean productivity and phytoplankton physiology from space. Glob. Biogeochem. Cycles 19, GB1006 (2005).
Dunne, J. P., Armstrong, R. A., Gnanadesikan, A. & Sarmiento, J. L. Empirical and mechanistic models for the particle export ratio. Glob. Biogeochem. Cycles 19, GB4026 (2005).
Laws, E. A., Falkowski, P. G., Smith, W. O., Ducklow, H. & McCarthy, J. J. Temperature effects on export production in the open ocean. Glob. Biogeochem. Cycles 14, 1231–1246 (2000).
Laws, E. A., D’Sa, E. & Naik, P. Simple equations to estimate ratios of new or export production to total production from satellite‐derived estimates of sea surface temperature and primary production. Limnol. Oceanogr. Methods 9, 593–601 (2011).
de Baar, H. J., Saager, P. M., Nolting, R. F. & van der Meer, J. Cadmium versus phosphate in the world ocean. Mar. Chem. 46, 261–281 (1994).
Wyatt, N. et al. Biogeochemical cycling of dissolved zinc along the GEOTRACES South Atlantic transect GA10 at 40S. Glob. Biogeochem. Cycles 28, 44–56 (2014).
Acknowledgements
D.B. was supported by NSF grant OCE-1635632; T.S.W. was supported by NSF grant OCE-1635414; C.D. was supported by the Gordon and Betty Moore Foundation (GBMF 3775); and R.K. was supported by the German Science Foundation through the Collaborative Research Center 754 ‘Climate-Biogeochemistry Interactions in the Tropical Ocean’. We thank D. Janssen and S. John for providing trace metal data from the Mauritanian region. We further acknowledge J. Coindat and S. Fevre of Hydroptic, and L. Stemman and M. Picheral for support during work with the UVP5.
Author information
Authors and Affiliations
Contributions
D.B. and T.S.W. conceived the project, developed the microenvironment and particle models, conducted the simulations and analysed output, with contributions from C.D. R.K. provided UVP5 particle observations and suggested their use for model validation. D.B. and T.S.W. wrote the paper, with contributions from C.D. and R.K.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Information, Tables and Figures
Numerical functions
Code for the particle microenvironment model, in Matlab
Rights and permissions
About this article
Cite this article
Bianchi, D., Weber, T.S., Kiko, R. et al. Global niche of marine anaerobic metabolisms expanded by particle microenvironments. Nature Geosci 11, 263–268 (2018). https://doi.org/10.1038/s41561-018-0081-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-018-0081-0
This article is cited by
-
Distinct Assembly Processes Structure Planktonic Bacterial Communities Among Near- and Offshore Ecosystems in the Yangtze River Estuary
Microbial Ecology (2024)
-
Deep-sea Bacteroidetes from the Mariana Trench specialize in hemicellulose and pectin degradation typically associated with terrestrial systems
Microbiome (2023)
-
Network analysis of 16S rRNA sequences suggests microbial keystone taxa contribute to marine N2O cycling
Communications Biology (2023)
-
Particle-associated denitrification is the primary source of N2O in oxic coastal waters
Nature Communications (2023)
-
Nitrite accumulation and anammox bacterial niche partitioning in Arctic Mid-Ocean Ridge sediments
ISME Communications (2023)