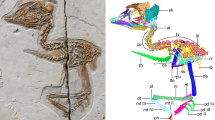
Abstract
The Cretaceous is a critical time interval that encompasses explosive diversifications of terrestrial vertebrates, particularly the period when the earliest-branching birds, after divergence from their theropod ancestors, evolved the characteristic avian Bauplan that led eventually to their global radiation. This early phylogenetic diversity is overwhelmed by the Ornithothoraces, consisting of the Enantiornithes and Ornithuromorpha, whose members evolved key derived features of crown birds. This disparity consequently circumscribes a large morphological gap between these derived clades and the oldest bird Archaeopteryx. The non-ornithothoracine pygostylians, with an intermediate phylogenetic position, are key to deciphering those evolutionary transformations, but progress in their study has been hampered by the limited diversity of known fossils. Here we report an Early Cetaceous non-ornithothoracine pygostylian, Cratonavis zhui gen. et sp. nov., that exhibits a unique combination of a non-avialan dinosaurian akinetic skull with an avialan post-cranial skeleton, revealing the key role of evolutionary mosaicism in early bird diversification. The unusually elongated scapular and metatarsal one preserved in Cratonavis highlights a breadth of skeletal plasticity, stemming from their distinct developmental modules and selection for possibly raptorial behaviour. Mapped changes in these two elements across theropod phylogeny demonstrate clade-specific evolutionary lability.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The specimen (IVPP V31106) described in this study is archived and available on request from the Institute of Vertebrate Paleontology and Paleoanthropology (IVPP), Chinese Academy of Sciences, Beijing, China. The data matrix used in the phylogenetic analysis is provided in Supplementary Information. The CT scanning results are archived and available on Open Science Framework (https://osf.io/6jd4h/?view_only=a68708fb3f8f4a4e88494ba44f85e624) or request from the corresponding author. This published work and the nomenclatural acts it contains have been registered in ZooBank, the proposed online registration system for the International Code of Zoological Nomenclature (ICZN). The ZooBank Life Science Identifiers (LSIDs) can be resolved and the associated information viewed through any standard web browser by appending the LSID to the prefix http://zoobank.org/. The LSIDs for this publication are: urn:lsid:zoobank.org:pub:2F4C81B7-E844-470C-9D35-FFAE62F04781.
Code availability
The R code that we used in comparative analyses is archived and available on OSF (https://osf.io/6jd4h/?view_only=a68708fb3f8f4a4e88494ba44f85e624).
References
Brusatte, S. L., O’Connor, J. K. & Jarvis, E. D. The origin and diversification of birds. Curr. Biol. 25, R888–R898 (2015).
Xu, X. et al. An integrative approach to understanding bird origins. Science 346, 1253293 (2014).
Chiappe, L. M. & Meng, Q. Birds of Stone: Chinese Avian Fossils from the Age of Dinosaurs (Johns Hopkins Univ. Press, 2016).
O’Connor, J. K, Chiappe, L. M. & Bell, A. in Living Dinosaurs: the Evolutionary History of Birds (eds Gareth J. D. & Gary, K.) 39–114 (Wiley, 2011).
Field, D. J. et al. Complete Ichthyornis skull illuminates mosaic assembly of the avian head. Nature 557, 96–100 (2018).
Lovette, I. J. & Fitzpatrick, J. W. Handbook of Bird Biology, 3rd edn (John Wiley & Sons, 2016).
Gauthier, J. Saurischian monophyly and the origin of birds. Mem. Calif. Acad. Sci. 8, 1–55 (1986).
Chiappe, L. M. & Walker, C. A. in Mesozoic Birds: Above the Heads of Dinosaurs (eds Chiappe, L. M. & Witmer, L. M.) 448–472 (Univ. California, 2002).
Wang, M., Stidham, T. A. & Zhou, Z. A new clade of basal Early Cretaceous pygostylian birds and developmental plasticity of the avian shoulder girdle. Proc. Natl Acad. Sci. USA 115, 10708–10713 (2018).
Zhou, Z., Meng, Q., Zhu, R. & Wang, M. Spatiotemporal evolution of the Jehol Biota: responses to the North China Craton destruction in the Early Cretaceous. Proc. Natl Acad. Sci. USA 118, e2107859118 (2021).
Hone, D. W., Farke, A. A. & Wedel, M. J. Ontogeny and the fossil record: what, if anything, is an adult dinosaur? Biol. Lett. 12, 20150947 (2016).
Plateau, O. & Foth, C. Common patterns of skull bone fusion and their potential to discriminate different ontogenetic stages in extant birds. Frontiers Ecol. Evol. 9, (2021).
Campione, N. E., Evans, D. C., Brown, C. M. & Carrano, M. T. Body mass estimation in non-avian bipeds using a theoretical conversion to quadruped stylopodial proportions. Methods Ecol. Evol. 5, 913–923 (2014).
Mayr, G., Pohl, B., Hartman, S. & Peters, D. S. The tenth skeletal specimen of Archaeopteryx. Zool. J. Linn. Soc. 149, 97–116 (2007).
Rauhut, O. W., Foth, C. & Tischlinger, H. The oldest Archaeopteryx (Theropoda: Avialiae): a new specimen from the Kimmeridgian/Tithonian boundary of Schamhaupten, Bavaria. PeerJ 6, e4191 (2018).
Xu, X. et al. The taxonomic status of the Late Cretaceous dromaeosaurid Linheraptor exquisitus and its implications for dromaeosaurid systematics. Vertebr. Palasiat. 53, 29–62 (2015).
Hu, H. et al. Evolution of the vomer and its implications for cranial kinesis in Paraves. Proc. Natl Acad. Sci. USA 116, 19571–19578 (2019).
Wang, M., Stidham, T. A., Li, Z., Xu, X. & Zhou, Z. Cretaceous bird with dinosaur skull sheds light on avian cranial evolution. Nat. Commun. 12, 3890 (2021).
Wang, M., O’Connor, J. K. & Zhou, Z. A taxonomical revision of the Confuciusornithiformes (Aves: Pygostylia). Vertebr. Palasiat. 57, 1–37 (2019).
O’Connor, J. K. & Chiappe, L. M. A revision of enantiornithine (Aves: Ornithothoraces) skull morphology. J. Syst. Palaeontol. 9, 135–157 (2011).
Xu, X., Norell, M. A., Wang, X., Makovicky, P. J. & Wu, X. A basal troodontid from the Early Cretaceous of China. Nature 415, 780–784 (2002).
Zhang, Z., Chiappe, L. M., Han, G. & Chinsamy, A. A large bird from the Early Cretaceous of China: new information on the skull of enantiornithines. J. Vertebr. Paleontol. 33, 1176–1189 (2013).
Turner, A. H., Makovicky, P. J. & Norell, M. A. A review of dromaeosaurid systematics and paravian phylogeny. Bull. Am. Mus. Nat. Hist. 371, 1–206 (2012).
Witmer, L. in Encyclopedia of Dinosaurs (eds Currie, P. J. & Padian, K.) 151–159 (Academic Press, 1997).
Currie, P. J. New information on the anatomy and relationships of Dromaeosaurus albertensis (Dinosauria: Theropoda). J. Vertebr. Paleontol. 15, 576–591 (1995).
Xu, X., You, H., Du, K. & Han, F. An Archaeopteryx-like theropod from China and the origin of Avialae. Nature 475, 465–470 (2011).
O’Connor, J. K. A systematic review of Enantiornithes (Aves: Ornithothoraces) (University of Southern California, 2009).
Norell, M. A., Clark, J. M., Turner, A. H., Makovicky, P. J., Barsbold, R. & Rowe, T. A new dromaeosaurid theropod from Ukhaa Tolgod (Ömnögov, Mongolia). Am. Mus. Novit. 3545, 1–51 (2006).
Sullivan, C. & Xu, X. Morphological diversity and evolution of the jugal in dinosaurs. Anat. Rec. 300, 30–48 (2017).
O’Connor, P. M. et al. Late Cretaceous bird from Madagascar reveals unique development of beaks. Nature 588, 272–276 (2020).
Rauhut, O. W. The interrelationships and evolution of basal theropod dinosaurs. Spec. Pap. Palaeontol. 69, 1–213 (2003).
Elzanowski, A. & Wellnhofer, P. Cranial morphology of Archaeopteryx: evidence from the seventh skeleton. J. Vertebr. Paleontol. 16, 81–94 (1996).
Chiappe, L. M., Norell, M. & Clark, J. A new skull of Gobipteryx minuta (Aves: Enantiornithes) from the Cretaceous of the Gobi Desert. Am. Mus. Novit. 3346,, 1–15 (2001).
Ostrom, J. H. Osteology of Deinonychus antirrhopus, an unusual theropod from the Lower Cretaceous of Montana. 30, 1–165 (1969).
Currie, P. J. & Zhao, X. A new carnosaur (Dinosauria, Theropoda) from the Jurassic of Xinjiang, People’s Republic of China. Can. J. Earth Sci. 30, 2037–2081 (1993).
Gingerich, P. D. Evolutionary significance of the Mesozoic toothed birds. Smithson. Contrib. Paleobiol. 27, 23–33 (1976).
McDowell, S. The bony palate of birds. Part I. The Palaeognathae. Auk 65, 520–549 (1948).
Baumel, J. J. & Witmer, L. M. in Handbook of Avian Anatomy: Nomina Anatomica Avium (eds Baumel J. J. et al.) 45–132 (Nuttall Ornithological Club, 1993).
Zhou, Z. & Zhang, F. A long-tailed, seed-eating bird from the Early Cretaceous of China. Nature 418, 405–409 (2002).
Vickaryous, M. K. & Hall, B. K. Homology of the reptilian coracoid and a reappraisal of the evolution and development of the amniote pectoral apparatus. J. Anat. 208, 263–285 (2006).
Zhou, Z. & Zhang, F. Jeholornis compared to Archaeopteryx, with a new understanding of the earliest avian evolution. Naturwissenschaften 90, 220–225 (2003).
Wang, M. & Zhou, Z. A new confuciusornithid (Aves: Pygostylia) from the Early Cretaceous increases the morphological disparity of the Confuciusornithidae. Zool. J. Linn. Soc. 185, 417–430 (2019).
Chiappe, L. M., Ji, S. A., Ji, Q. & Norell, M. A. Anatomy and systematics of the Confuciusornithidae (Theropoda: Aves) from the Late Mesozoic of northeastern China. Bull. Am. Mus. Nat. Hist. 242, 1–89 (1999).
O’Connor, J. K. et al. Evolution and functional significance of derived sternal ossification patterns in ornithothoracine birds. J. Evol. Biol. 28, 1550–1567 (2015).
Chiappe, L. M. & Walker, C. A. in Mesozoic Birds: Above the Heads of Dinosaurs (eds Chiappe, L. M. & Witmer, L. M.) 240–267 (Univ. California, 2002).
Wang, M., Wang, X., Wang, Y. & Zhou, Z. A new basal bird from China with implications for morphological diversity in early birds. Sci. Rep. 6, 19700 (2016).
Rauhut, O. W. M., Tischlinger, H. & Foth, C. A non-archaeopterygid avialan theropod from the Late Jurassic of southern Germany. eLife 8, e43789 (2019).
Osmólska, H., Currie, P. J. & Barsbold, R. in The Dinosauria (eds. Weishampel, D., Dodson, P. & Osmólska, H.) 165–183 (University of California Press, 2004).
Hutchinson, J. R. The evolution of pelvic osteology and soft tissues on the line to extant birds (Neornithes). Zool. J. Linn. Soc. 131, 123–168 (2001).
Wellnhofer, P. Archaeopteryx: The Icon of Evolution (Verlag Dr. Friedrich Pfeil, 2009).
Hattori, S. Evolution of the hallux in non-avian theropod dinosaurs. J. Vertebr. Paleontol. 36, e1116995 (2016).
Zhou, Z. & Zhang, F. Anatomy of the primitive bird Sapeornis chaoyangensis from the Early Cretaceous of Liaoning, China. Can. J. Earth Sci. 40, 731–747 (2003).
Botelho, J. F., Smith-Paredes, D., Soto-AcuÑA, S., NÚÑEz-LeÓN, D., Palma, V. & Vargas, A. O. Greater growth of proximal metatarsals in bird embryos and the evolution of hallux position in the grasping foot. J. Exp. Zool. 00, 1–13 (2016).
Wang, M. et al. An Early Cretaceous enantiornithine bird with a pintail. Curr. Biol. 31, 4845–4852 (2021).
Wang, M., Lloyd, G. T., Zhang, C. & Zhou, Z. The patterns and modes of the evolution of disparity in Mesozoic birds. Proc. R. Soc. B 288, 20203105 (2021).
Hu, H., O’Connor, J. K. & Zhou, Z. A new species of Pengornithidae (Aves: Enantiornithes) from the Lower Cretaceous of China suggests a specialized scansorial habitat previously unknown in early birds. PLoS ONE 10, e0126791 (2015).
O’Connor, J. K., Wang, M. & Hu, H. A new ornithuromorph (Aves) with an elongate rostrum from the Jehol Biota, and the early evolution of rostralization in birds. J. Syst. Palaeontol. 14, 939–948 (2016).
Blomberg, S. P., Garland, T. & Ives, A. R. Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57, 717–745 (2003).
Pagel, M. Inferring the historical patterns of biological evolution. Nature 401, 877–884 (1999).
Holliday, C. M. & Witmer, L. M. Cranial kinesis in dinosaurs: intracranial joints, protractor muscles, and their significance for cranial evolution and function in diapsids. J. Vertebr. Paleontol. 28, 1073–1088 (2008).
Gussekloo, S. W. S. & Bout, R. G. Cranial kinesis in palaeognathous birds. J. Exp. Biol. 208, 3409–3419 (2005).
Plateau, O. & Foth, C. Birds have peramorphic skulls, too: anatomical network analyses reveal oppositional heterochronies in avian skull evolution. Commun. Biol. 3, 195 (2020).
Clarke, J. A. & Norell, M. A. The morphology and phylogenetic position of Apsaravis ukhaana from the Late Cretaceous of Mongolia. Am. Mus. Novit. 3387, 1–46 (2002).
Huang, R., Zhi, Q., Patel, K., Wilting, J. & Christ, B. Dual origin and segmental organisation of the avian scapula. Development 127, 3789–3794 (2000).
Jenkins, F. A. The evolution of the avian shoulder joint. Am. J. Sci. 293, 253–253 (1993).
Novas, F. E., Motta, M. J., Agnolín, F. L., Rozadilla, S., Lo Coco, G. E. & Brissón Egli, F. Comments on the morphology of basal paravian shoulder girdle: new data based on unenlagiid theropods and paleognath birds. Front. Earth Sci. 9, (2021).
Ostrom, J. H. Some hypothetical anatomical stages in the evolution of avian flight. Smithson. Contrib. Paleobiol. 27, 1–21 (1976).
Dial, K. P. Activity patterns of the wing muscles of the pigeon (Columba livia) during different modes of flight. J. Exp. Zool. 262, 357–373 (1992).
Kovacs, C. E. & Meyers, R. A. Anatomy and histochemistry of flight muscles in a wing-propelled diving bird, the Atlantic Puffin, Fratercula arctica. J. Morphol. 244, 109–125 (2000).
Brusatte, S. L. et al. The osteology of Balaur bondoc, an Island-Dwelling Dromaeosaurid (Dinosauria: Theropoda) from the Late Cretaceous of Romania. Bull. Am. Mus. Nat. Hist. 2013, 1–100 (2013).
Romer, A. S. Osteology of the Reptiles (Univ. Chicago Press, 1956).
Gatesy, S. M. Hind limb scaling in birds and other theropods: implications for terrestrial locomotion. J. Morphol. 209, 83–96 (1991).
Middleton, K. M. Morphology, Evolution, and Function of the Avian Hallux (Brown Univ., 2003).
Kamilar, J. M. & Cooper, N. Phylogenetic signal in primate behaviour, ecology and life history. Philos. Trans. R. Soc. B 368, 20120341 (2013).
Goloboff, P. A. & Catalano, S. A. TNT version 1.5, including a full implementation of phylogenetic morphometrics. Cladistics 32, 221–238 (2016).
Benson, R. B. J. et al. Rates of dinosaur body mass evolution indicate 170 million years of sustained ecological innovation on the avian stem lineage. PLoS Biol. 12, e1001853 (2014).
Xu, X. et al. Two Early Cretaceous fossils document transitional stages in alvarezsaurian dinosaur evolution. Curr. Biol. 28, 1–8 (2018).
Lee, Y. N. et al. Resolving the long-standing enigmas of a giant ornithomimosaur Deinocheirus mirificus. Nature 515, 257–260 (2014).
Rauhut, O. W. M. & Pol, D. Probable basal allosauroid from the early Middle Jurassic Cañadón Asfalto Formation of Argentina highlights phylogenetic uncertainty in tetanuran theropod dinosaurs. Sci. Rep. 9, 18826 (2019).
Novas, F. E., Agnolin, F. L., Ezcurra, M. D., Temp Müller, R., Martinelli, A. G. & Langer, M. C. Review of the fossil record of early dinosaurs from South America, and its phylogenetic implications. J. S. Am. Earth Sci. 110, 103341 (2021).
Paradis, E., Claude, J. & Strimmer, K. APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20, 289–290 (2004).
Brusatte, S. L. in Computational Paleontology (ed. Elewa, A. M. T) 53–74 (Springer, 2011).
Bapst, D. W. Paleotree: an R package for paleontological and phylogenetic analyses of evolution. Methods Ecol. Evol. 3, 803–807 (2012).
Felsenstein, J. Phylogenies and the comparative method. Am. Nat. 125, 1–15 (1985).
Revell, L. J. Phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 3, 217–223 (2012).
Blomberg, S. P., Garland, T. & Ives, A. R. Testing for phylogenetic signal in comparative data: Behavioral traits are more labile. Evolution 57, 717–745 (2003).
Warton, D. I., Duursma, R. A., Falster, D. S. & Taskinen, S. SMATR 3—an R package for estimation and inference about allometric lines. Methods Ecol. Evol. 3, 257–259 (2012).
Legendre, P. lmodel2. R package version 1.7-2 http://cran.r-project.org/web/packages/lmodel2/lmodel2.pdf. (2013)
Pinheiro, J. et al. nlme: Linear and Nonlinear Mixed EffectsModels. R package version 3.1-158 https://svn.r-project.org/R-packages/trunk/nlme/ (2022)
Dececchi, T. A. & Larsson, H. C. E. Assessing arboreal adaptations of bird antecedents: testing the ecological setting of the origin of the avian flight stroke. PLoS ONE 6, e22292 (2011).
Hastie, T., Tibshirani, R., Leisch, F., Hornik, K. & Ripley, B. mda: mixture and flexible discriminant analysis. R package version 0.5-3 https://cran.r-project.org/package=mda (2022).
Acknowledgements
We thank P. Yin for help with CT scanning, and W. Gao for photographing. This research is supported by the National Natural Science Foundation of China (42288201), the Key Research Program of Frontier Sciences, CAS (ZDBS-LY-DQC002) and the Tencent Foundation (through the XPLORER PRIZE).
Author information
Authors and Affiliations
Contributions
M.W. conceived the project; Z.L. and M.W. conducted the digital reconstruction; M.W. collected the data; M.W. performed the phylogenetic analysis and comparative analyses; M.W., Z.L., T.A.S. and Z.Z. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Fernando Novas and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Additional cranial anatomy of Cratonavis zhui, IVPP V31106.
a, Photograph. b, c, CT Isosurface of the whole skull in dorsal (a) and (b) ventral views. ba, basisphenoid-parasphenoid; bp, basipterygoid process; car, caudal ramus of lacrimal; de, dentary; ecp, ectopterygoid; fp, frontal process of premaxilla; fr, frontal; jmx, jugal process of maxilla; ju, jugal; jup, jugal process of postorbital; lc, lacrimal; mx, maxilla; na, nasal; pa, palatine; pi, parietal; pm, premaxilla; pmd, post-dentary mandible; po, postorbital; poq, postorbital process of jugal; pr, parasphenoid rostrum; pt, pterygoid; qju, quadratojugal process of jugal; qu, quadrate; quj, quadratojugal; rea, retroarticular process; sqq, squamosal process of quadratojugal; sr, subcellar recess; vo, vomer; l/r, left/right side. The arrowhead (a) denotes the lateral flange of the lacrimal. Scale bars, 10 mm (a–c).
Extended Data Fig. 2 CT scanning of pectoral region of C. zhui.
am, alular metacarpal; bi, bicipital tubercle; co, coracoid; dp, deltopectoral crest; dv, dorsal vertebra; fu, furcula; gl, glenoid; hu, humerus; ma, major metacarpal; m1 to m3, major digit phalanx 1 to 3; mi, minor metacarpal; mi1, minor digit phalanx 1; ol, olecranon; ra, radius; rd, radiale; sp, scapula; uc, uncinate; un, ulna; l/r, left/right side. The arrowheads denote the lateral fossae of the dorsal centra. Scale bar, 10 mm.
Extended Data Fig. 3 Additional pelvis and hindlimb anatomy of C. zhui.
a, Photograph. b, CT Scanning. fe, femur; fi, fibula; il, ilium; isp, ischiatic peduncle; mt I–V, metatarsal I to V; pop, postacetabular process; prp, preacetabular process; pt, posterior trochanter; pu, pubis; pup, pubic peduncle; py, pygostyle; ti, tibiotarsus; 1–8, sacral vertebrae one to eight; l/r, left/right side. Scale bar, 10 mm.
Extended Data Fig. 4 Time-calibrated phylogeny of theropod dinosaurs.
The phylogeny is a super tree encompassing major theropod groups that preserve complete appendicular elements used as the backbone for comparative analysis (see Methods).
Extended Data Fig. 5 Evolution of scapula across theropod dinosaurs.
Scapula length changes among major theropod groups (line drawing of scapulocoracoid/scapula scaled with humerus in selected taxa). The phylogenetical signals were quantified using the Blomberg’s K and Pagel’s lambd with P-value of the likelihood radio test. Node name: a: Allosauroidea, b: Tyrannosauroidae, c: Compsognathidae, d: Therizinosauria, e: Alvarezsauria, f: Ornithomimosauria, g: Oviraptorosauria, h: Scansoriorpterygidae, i: Troodontidae, j: Dromaeosauridae.
Extended Data Fig. 6 Scaling relationship between scapula and humerus/femur length across theropod dinosaurs using phylogenetic generalized least squares (pgls).
a, Scapula against humerus. b, Scapula against femur. Statistically significant relationship is denoted by p-value (*<0.01).
Extended Data Fig. 7 Evolution of metatarsals across theropod dinosaurs.
Changes of metatarsal I length along the line to early avialans (metatarsal I and hallux colored in red and green, respectively). The phylogenetical signals were quantified using the Blomberg’s K and Pagel’s lambd with P-value of the likelihood radio test. Node name: a: Allosauroidea, b: Tyrannosauroidae, c: Compsognathidae, d: Therizinosauria, e: Alvarezsauria, f: Ornithomimosauria, g: Oviraptorosauria, h: Scansoriorpterygidae, i: Troodontidae, j: Dromaeosauridae.
Extended Data Fig. 8 Changes of metatarsal I length across theropod dinosaurs.
a, Metatarsals I/III length ratio mapped onto time-calibrated theropod phylogeny. b, Size and phylogenetically corrected metatarsal I length mapped onto time-calibrated paravian phylogeny. The phylogenetical signals were quantified using the Blomberg’s K and Pagel’s lambd with P-value of the likelihood radio test.
Extended Data Fig. 9 Scaling relationship between metatarsals I and III length across theropod dinosaurs.
Results using the phylogenetic generalized least squares regression (pgls). Statistically significant relationship is denoted by p-value (*<0.01).
Extended Data Fig. 10 Results of canonical variate analysis to predicate the ecologies of modern bird samples.
The modern samples can be 87.5% correctly assigned to their original ecological classifications using selected morphological traits.
Supplementary information
Supplementary Information
Supplementary Note 1, Supplementary Tables 1–5, supplementary information about the morphological characters and dataset used in the phylogenetic analyses, and references.
Supplementary Tables 2 and 3
Supplementary Table 2. Appendicular element length dataset of theropods used in phylogenetic comparative analyses. Supplementary Table 3. Dataset used in morphometric analysis.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Z., Wang, M., Stidham, T.A. et al. Decoupling the skull and skeleton in a Cretaceous bird with unique appendicular morphologies. Nat Ecol Evol 7, 20–31 (2023). https://doi.org/10.1038/s41559-022-01921-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-022-01921-w