Abstract
Proboscideans were keystone Cenozoic megaherbivores and present a highly relevant case study to frame the timing and magnitude of recent megafauna extinctions against long-term macroevolutionary patterns. By surveying the entire proboscidean fossil history using model-based approaches, we show that the dramatic Miocene explosion of proboscidean functional diversity was triggered by their biogeographical expansion beyond Africa. Ecomorphological innovations drove niche differentiation; communities that accommodated several disparate proboscidean species in sympatry became commonplace. The first burst of extinctions took place in the late Miocene, approximately 7 million years ago (Ma). Importantly, this and subsequent extinction trends showed high ecomorphological selectivity and went hand in hand with palaeoclimate dynamics. The global extirpation of proboscideans began escalating from 3 Ma with further extinctions in Eurasia and then a dramatic increase in African extinctions at 2.4 Ma. Overhunting by humans may have served as a final double jeopardy in the late Pleistocene after climate-triggered extinction trends that began long before hominins evolved suitable hunting capabilities.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Datasets are available in Supplementary Data 1 and from figshare (https://doi.org/10.6084/m9.figshare.14035109).
Code availability
PyRate v.3.0 is a Python-based (v.3.) program available at https://github.com/dsilvestro/PyRate. Computation in R used functions in the packages mvMORPH v.1.1.3, phytools v.0.7-70, ape v.5.5 and lme4 v.1.1-23. Input files can be obtained from Supplementary Data 1 and figshare (https://doi.org/10.6084/m9.figshare.14035109).
References
Surovell, T., Waguespack, N. & Brantingham, P. J. Global archaeological evidence for proboscidean overkill. Proc. Natl Acad. Sci. USA 102, 6231–6236 (2005).
Smith, F. A., Smith, R. E. E., Lyons, S. K. & Payne, J. L. Body size downgrading of mammals over the late Quaternary. Science 360, 310–313 (2018).
Faith, J. T., Rowan, J., Du, A. & Barr, W. A. The uncertain case for human-driven extinctions prior to Homo sapiens. Quat. Res. 96, 88–104 (2020).
Cuvier, G. Mémoires sur les Espèces d’Éléphants Vivants et Fossiles. Mémoires de l’Institut des Sciences et Arts 2, 1–22 (1800); https://www.biodiversitylibrary.org/page/16303001#page/175/mode/1up
Osborn, H. F. The ancestral tree of the Proboscidea. Discovery, evolution, migration and extinction over a 50,000,000 year period. Proc. Natl Acad. Sci. USA 21, 404–412 (1935).
International Union for Conservation of Nature. The IUCN Red List of Threatened Species Version 2021-1 (IUCN, 2021); https://www.iucnredlist.org
Maglio, V. J. Origin and evolution of the Elephantidae. Trans. Am. Philos. Soc. 63, 1–149 (1973).
Zhang, H., Wang, Y., Janis, C. M., Goodall, R. H. & Purnell, M. A. An examination of feeding ecology in Pleistocene proboscideans from southern China (Sinomastodon, Stegodon, Elephas), by means of dental microwear texture analysis. Quat. Int. 445, 60–70 (2017).
Saegusa, H. Stegodontidae and Anancus: keys to understanding dental evolution in Elephantidae. Quat. Sci. Rev. 231, 106176 (2020).
Hempson, G. P., Archibald, S. & Bond, W. J. A continent-wide assessment of the form and intensity of large mammal herbivory in Africa. Science 350, 1056–1061 (2015).
Silvestro, D., Salamin, N., Antonelli, A. & Meyer, X. Improved estimation of macroevolutionary rates from fossil data using a Bayesian framework. Paleobiology 45, 546–570 (2019).
Clavel, J., Escarguel, G. & Merceron, G. mvMORPH: an R package for fitting multivariate evolutionary models to morphometric data. Methods Ecol. Evol. 6, 1311–1319 (2015).
Cantalapiedra, J. L., Hernández Fernández, M., Azanza, B. & Morales, J. Congruent phylogenetic and fossil signatures of mammalian diversification dynamics driven by Tertiary abiotic change. Evolution 69, 2941–2953 (2015).
Silvestro, D., Antonelli, A., Salamin, N. & Quental, T. B. The role of clade competition in the diversification of North American canids. Proc. Natl Acad. Sci. USA 112, 8684–8689 (2015).
Sepkoski, J. J. A kinetic model of Phanerozoic taxonomic diversity I. Analysis of marine orders. Paleobiology 4, 223–251 (1978).
Tassy, P. in European Neogene Mammal Chronology (eds Lindsay, E. H. et al.) 237–252 (Plenus Press, 1989).
van der Made, J. in Elefantentreich: eine Fossilwelt in Europa (ed. Meller, H.) 340–360 (Landesamt für Denkmalpflege und Archäologie Sachsen-Anhalt-Landesmuseum für Vorgeschichte, 2010).
Saarinen, J. J. et al. Patterns of maximum body size evolution in Cenozoic land mammals: eco-evolutionary processes and abiotic forcing. Proc. Biol. Sci. 281, 20132049 (2014).
Fortelius, M. et al. Evolution of Neogene mammals in Eurasia: environmental forcing and biotic interactions. Annu. Rev. Earth Planet Sci. 42, 579–604 (2014).
Marshall, C. R. & Quental, T. B. The uncertain role of diversity dependence in species diversification and the need to incorporate time-varying carrying capacities. Philos. Trans. R. Soc. Lond. B 371, 20150217 (2016).
Vrba, E. S. Evolution, species and fossils: how does life evolve? S. Afr. J. Sci. 76, 61–84 (1980).
Cantalapiedra, J. L., Prado, J. L., Hernández Fernández, M. & Alberdi, M. T. Decoupled ecomorphological evolution and diversification in Neogene-Quaternary horses. Science 355, 627–630 (2017).
Calandra, I., Göhlich, U. B. & Merceron, G. How could sympatric megaherbivores coexist? Example of niche partitioning within a proboscidean community from the Miocene of Europe. Naturwissenschaften 95, 831–838 (2008).
Sanders, W. J. Proboscidea from Kanapoi, Kenya. J. Hum. Evol. 140, 102547 (2020).
Wang, S. et al. Evolution of Protanancus (Proboscidea, Mammalia) in East Asia. J. Vertebr. Paleontol. 35, e881830 (2015).
Lister, A. M. The role of behaviour in adaptive morphological evolution of African proboscideans. Nature 500, 331–334 (2013).
Lister, A. M., Sher, A. V., van Essen, H. & Wei, G. The pattern and process of mammoth evolution in Eurasia. Quat. Int. 126, 49–64 (2005).
Wei, G. et al. New materials of the steppe mammoth, Mammuthus trogontherii, with discussion on the origin and evolutionary patterns of mammoths. Sci. China Earth Sci. 53, 956–963 (2010).
Stanley, S. M. Macroevolution: Patterns and Processes (W. H. Freeman and Company, 1979).
Edwards, E. J. et al. The origins of C4 grasslands: integrating evolutionary and ecosystem science. Science 328, 587–591 (2010).
Faith, J. T., Rowan, J., Du, A. & Koch, P. L. Plio-Pleistocene decline of African megaherbivores: no evidence for ancient hominin impacts. Science 362, 938–941 (2018).
Kaya, F. et al. The rise and fall of the Old World savannah fauna and the origins of the African savannah biome. Nat. Ecol. Evol. 2, 241–246 (2018).
Saarinen, J. & Lister, A. M. Dental mesowear reflects local vegetation and niche separation in Pleistocene proboscideans from Britain. J. Quat. Sci. 31, 799–808 (2016).
Rivals, F., Semprebon, G. M. & Lister, A. M. Feeding traits and dietary variation in Pleistocene proboscideans: a tooth microwear review. Quat. Sci. Rev. 219, 145–153 (2019).
Vrba, E. S. in Living Fossils (eds Eldredge, N. & Stanley, S. M.) 62–79 (Springer, 1984).
Herrera‐Flores, J. A., Stubbs, T. L. & Benton, M. J. Macroevolutionary patterns in Rhynchocephalia: is the tuatara (Sphenodon punctatus) a living fossil? Palaeontology 60, 319–328 (2017).
Todd, N. E. Trends in proboscidean diversity in the African Cenozoic. J. Mamm. Evol. 13, 1–10 (2006).
Rivals, F., Mol, D., Lacombat, F., Lister, A. M. & Semprebon, G. M. Resource partitioning and niche separation between mammoths (Mammuthus rumanus and Mammuthus meridionalis) and gomphotheres (Anancus arvernensis) in the Early Pleistocene of Europe. Quat. Int. 379, 164–170 (2015).
Sanders, W. J. & Haile-Selassie, Y. A new assemblage of mid-Pliocene proboscideans from the Woranso-Mille area, Afar region, Ethiopia: taxonomic, evolutionary, and paleoecological considerations. J. Mamm. Evol. 19, 105–128 (2012).
van der Geer, A. A. E. et al. The effect of area and isolation on insular dwarf proboscideans. J. Biogeogr. 43, 1656–1666 (2016).
Westerhold, T. et al. An astronomically dated record of Earth’s climate and its predictability over the last 66 million years. Science 369, 1383–1387 (2020).
Vrba, E. S. in African Biogeography, Climate Change, and Hominid Evolution (eds Bromage, T. G. & Shrenk, F.) 19–39 (Oxford Univ. Press, 1999).
Stuart, A. J. Late Quaternary megafaunal extinctions on the continents: a short review. Geol. J. 50, 338–363 (2015).
Jukar, A. M., Lyons, S. K., Wagner, P. J. & Uhen, M. D. Late Quaternary extinctions in the Indian subcontinent. Palaeogeogr. Palaeoclimatol. Palaeoecol. 562, 110137 (2021).
Raup, D. M. Extinction: Bad Genes or Bad Luck? (Norton, 1991).
Cantalapiedra, J. L. et al. Conserving evolutionary history does not result in greater diversity over geological time scales. Proc. Biol. Sci. 286, 20182896 (2019).
Ronquist, F. et al. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 61, 539–542 (2012).
Hublin, J.-J. et al. New fossils from Jebel Irhoud, Morocco and the pan-African origin of Homo sapiens. Nature 546, 289–292 (2017).
O’Connell, J. F. et al. When did Homo sapiens first reach Southeast Asia and Sahul? Proc. Natl Acad. Sci. USA 115, 8482–8490 (2018).
Ardelean, C. F. et al. Evidence of human occupation in Mexico around the Last Glacial Maximum. Nature 584, 87–92 (2020).
Paradis, E. Analysis of Phylogenetics and Evolution with R (Springer, 2012).
Revell, L. J. phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 3, 217–223 (2012).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
MacLatchy, L. M., Desilva, J., Sanders, W. J. & Wood, B. in Cenozoic Mammals of Africa (eds Werdelin, L. & Sanders, W. J.) 471–545 (Univ. California Press, 2010).
Acknowledgements
We thank D. Silvestro, J. Calatayud and J. Clavel for technical assistance and F. Bibi, M. J. Benton and W. J. Sanders for helpful comments. S. Wang and G. van den Bergh provided important constructive comments on the informal supertree. We also thank E. M. Dunne, D. Mothé and F. Rivals, whose comments and recommendations improved the presentation of this paper. This work was supported by the Talent Attraction Program of the Madrid Government (no. 2017-T1/AMB5298), the German Research Foundation (AOBJ no. 637491) and the Academy of Finland (post-doctoral research fund no. 315691).
Author information
Authors and Affiliations
Contributions
J.L.C., O.S. and J.S. conceptualized the research. J.L.C., O.S., J.S., H.Z., M.T.A. and J.L.P. gathered the data. J.L.C., O.S. and F.B. designed and performed the analysis. J.S. wrote the description and relevance of functional traits. J.L.C. and O.S. wrote the first version of the paper. H.Z. and J.S. contributed to the final version (main text and supplement).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Ecology & Evolution thanks Emma Dunne, Pasquale Raia, Dimila Mothé, Florent Rivals and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Description of the 17 ecomorphological traits.
Further details regarding traits scoring can be found in the Supplementary Methods.
Extended Data Fig. 2 Phylo-functional space of proboscideans.
Species coloured according to taxonomic affinities (a) and proboscidean functional type (b). Correlations of the different ecomorphological features with the functional space (c). Correlations of ordered variables (for example body size) are shown in one plot, whereas unordered variables have been broken down into binary traits prior to each correlation analysis.
Extended Data Fig. 3 Phenograms of NMDS axes 1 and 2.
PFTs reconstructed on the tree using a stochastic mapping as implemented in the make.simmap function in the R library phytools52.
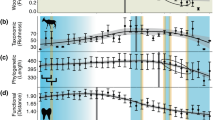
Extended Data Fig. 4 Global and continental proboscidean diversity through time as estimated using PyRate.
Shaded areas represent 95% credible intervals. Diversity axis is log-scaled.
Extended Data Fig. 5 Results from macroevolutionary phylogenetic models.
Support is indicated by AICc scores (smaller is better) and AICc Weights (higher is better). Average parameter estimates were obtained from 100 trees. Evolutionary rates are colour-coded for comparison (reddish being faster rates).
Extended Data Fig. 6 Parameter estimates of the community-level general linear mixed-effects models.
Mean, upper and lower confidence limits are provided for intercepts and slopes.
Extended Data Fig. 7 Representation of the mixed-effects models.
Relationship of local species richness with age (a), and averaged DMD (b) across different phases. Density plots show the distribution of average DMD scores in each phase. A schematic comparison of slopes and intercepts is included, showing whether parameters estimates are significantly larger (>), significantly smaller (<), or not significantly different (≈).
Extended Data Fig. 8 Effect of ecology (NMDS scores in axis 1 and 2) on speciation (𝛼λ, blue) and extinction (𝛼𝜇, red).
Posterior estimates of the correlation parameters. Horizontal lines beneath the histograms represent 95% credible intervals, which in all cases are significantly different from 0. NMDS-1 is associated with differential speciation potencial, whereas NMDS-2 is associated with differential extinction.
Extended Data Fig. 9 Aggregated effect of ecology on speciation (blue), and extinction (red).
The effect of specific species ecology as the departure from baseline rates plotted over time (a,b) and on the tree (c,d). Phylogenetic reconstructions were conducted with the contMap function in the R library phytools52.
Extended Data Fig. 10 Extinction selectivity through time.
Differences in mean pairwise disparity between random extinctions and observed extinctions across the entire proboscidean history (a), and the last 10 Myr (b). Positive values represent ecologically-restricted extinction; negative values result when extinction hits on broad regions of the functional space; the grey points show values consistent with a random signal (non-significant P-values).
Supplementary information
Supplementary Information
Supplementary Methods
Supplementary Data 1
Proboscidean occurrences, traits and PFTs.
Rights and permissions
About this article
Cite this article
Cantalapiedra, J.L., Sanisidro, Ó., Zhang, H. et al. The rise and fall of proboscidean ecological diversity. Nat Ecol Evol 5, 1266–1272 (2021). https://doi.org/10.1038/s41559-021-01498-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-021-01498-w