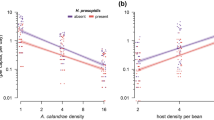
Abstract
Host–parasite interactions often fuel coevolutionary change. However, parasitism is one of a myriad of possible ecological interactions in nature. Biotic (for example, predation) and abiotic (for example, temperature) variation can amplify or dilute parasitism as a selective force on hosts and parasites, driving population variation in (co)evolutionary trajectories. We dissected the relationships between wider ecology and coevolutionary trajectory using 16 ecologically complex Daphnia magna–Pasteuria ramosa ponds seeded with an identical starting host (Daphnia) and parasite (Pasteuria) population. We show, using a time-shift experiment and outdoor population data, how multivariate biotic and abiotic ecological differences between ponds caused coevolutionary divergence. Wider ecology drove variation in host evolution of resistance, but not parasite infectivity; parasites subsequently coevolved in response to the changing complement of host genotypes, such that parasites adapted to historically resistant host genotypes. Parasitism was a stronger interaction for the parasite than for its host, probably because the host is the principal environment and selective force, whereas for hosts, parasite-mediated selection is one of many sources of selection. Our findings reveal the mechanisms through which wider ecology creates coevolutionary hotspots and coldspots in biologically realistic arenas of host–parasite interaction, and sheds light on how the ecological theatre can affect the (co)evolutionary play.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data are available on Dryad at https://doi.org/10.5061/dryad.qv9s4mwd6.
Code availability
All companion code is available on Dryad at https://doi.org/10.5061/dryad.qv9s4mwd6. As we are actively researching these datasets, we ask that researchers kindly contact us if they are planning to use the data for reasons other than reproducing the findings of our paper.
References
Paterson, S. et al. Antagonistic coevolution accelerates molecular evolution. Nature 464, 275–278 (2010).
Schulte, R. D., Makus, C., Hasert, B., Michiels, N. K. & Schulenburg, H. Multiple reciprocal adaptations and rapid genetic change upon experimental coevolution of an animal host and its microbial parasite. Proc. Natl Acad. Sci. USA 107, 7359–7364 (2010).
Koskella, B. & Lively, C. M. Evidence for negative frequency-dependent selection during experimental coevolution of a freshwater snail and a sterilizing trematode. Evolution 63, 2213–2221 (2009).
Decaestecker, E. et al. Host–parasite ‘Red Queen’ dynamics archived in pond sediment. Nature 450, 870–873 (2007).
Gómez, P. & Buckling, A. Bacteria–phage antagonistic coevolution in soil. Science 332, 106–109 (2011).
Refardt, D. & Ebert, D. Inference of parasite local adaptation using two different fitness components. J. Evol. Biol. 20, 921–929 (2007).
Duffy, M. A., Hall, S. R., Cáceres, C. E. & Ives, A. R. Rapid evolution, seasonality, and the termination of parasite epidemics. Ecology 90, 1441–1448 (2009).
Springer, Y. P. Clinical resistance structure and pathogen local adaptation in a serpentine flax–flax rust interaction. Evolution 61, 1812–1822 (2007).
Tack, A. J. M., Laine, A.-L., Burdon, J. J., Bissett, A. & Thrall, P. H. Below-ground abiotic and biotic heterogeneity shapes above-ground infection outcomes and spatial divergence in a host–parasite interaction. New Phytol. 207, 1159–1169 (2015).
Wolinska, J. & King, K. C. Environment can alter selection in host–parasite interactions. Trends Parasitol. 25, 236–244 (2009).
Auld, S. K. J. R., Hall, S. R., Ochs, J. H., Sebastian, M. & Duffy, M. A. Predators and patterns of within-host growth can mediate both among-host competition and evolution of transmission potential of parasites. Am. Nat. 184, S77–S90 (2014).
Wright, R. C. T., Brockhurst, M. A. & Harrison, E. Ecological conditions determine extinction risk in co-evolving bacteria–phage populations. BMC Evol. Biol. 16, 227 (2016).
Duffy, M. A. et al. Ecological context influences epidemic size and parasite-driven evolution. Science 335, 1636–1638 (2012).
Auld, S. K. J. R. & Brand, J. Environmental variation causes different (co) evolutionary routes to the same adaptive destination across parasite populations. Evol. Lett. 1, 245–254 (2017).
Su, M. & Boots, M. The impact of resource quality on the evolution of virulence in spatially heterogeneous environments. J. Theor. Biol. 416, 1–7 (2017).
Auld, S. K. J. R. & Tinsley, M. C. The evolutionary ecology of complex lifecycle parasites: linking phenomena with mechanisms. Heredity 114, 125–132 (2015).
Cardon, M., Loot, G., Grenouillet, G. & Blanchet, S. Host characteristics and environmental factors differentially drive the burden and pathogenicity of an ectoparasite: a multilevel causal analysis. J. Anim. Ecol. 80, 657–667 (2011).
Mahmud, M. A., Bradley, J. E. & MacColl, A. D. C. Abiotic environmental variation drives virulence evolution in a fish host–parasite geographic mosaic. Funct. Ecol. 31, 2138–2146 (2017).
Arruda, J. A., Marzolf, G. R. & Faulk, R. T. The role of suspended sediments in the nutrition of zooplankton in turbid reservoirs. Ecology 64, 1225–1235 (1983).
Mostowy, R. & Engelstädter, J. The impact of environmental change on host–parasite coevolutionary dynamics. Proc. R. Soc. B 278, 2283–2292 (2011).
Thompson, J. N. The Geographic Mosaic of Coevolution (Univ. Chicago Press, 2005).
Brett, M. T. Chaoborus and fish-mediated influences on Daphnia longispina population structure, dynamics and life history strategies. Oecologia 89, 69–77 (1992).
Goss, L. B. & Bunting, D. L. Daphnia development and reproduction: responses to temperature. J. Therm. Biol. 8, 375–380 (1983).
Luijckx, P., Fienberg, H., Duneau, D. & Ebert, D. A matching-allele model explains host resistance to parasites. Curr. Biol. 23, 1085–1088 (2013).
Bento, G. et al. The genetic basis of resistance and matching-allele interactions of a host–parasite system: the Daphnia magna–Pasteuria ramosa model. PLoS Genet. 13, e1006596 (2017).
Grosberg, R. K. Mate selection and the evolution of highly polymorphic self/nonself recognition genes. Science 289, 2111–2114 (2000).
Hutchinson, G. E. The Ecological Theater and the Evolutionary Play (Yale Univ. Press, 1965).
Stuart, Y. E. et al. Contrasting effects of environment and genetics generate a continuum of parallel evolution. Nat. Ecol. Evol. 1, 0158 (2017).
Klüttgen, B., Dülmer, U., Engels, M. & Ratte, H. ADaM, an artificial freshwater for the culture of zooplankton. Water Res. 28, 743–746 (1994).
Ebert, D., Zschokke-Rohringer, C. D. & Carius, H. J. Within- and between-population variation for resistance of Daphnia magna to the bacterial endoparasite Pasteuria ramosa. Proc. R. Soc. B 265, 2127–2134 (1998).
Auld, S. K. J. R. & Brand, J. Simulated climate change, epidemic size, and host evolution across host–parasite populations. Glob. Change Biol. 23, 5045–5053 (2017).
Holm, S. A simple sequentially rejective multiple test procedure. Scand. J. Stat. 6, 65–70 (1979).
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2019).
Brereton, R. G. & Lloyd, G. R. Re-evaluating the role of the Mahalanobis distance measure. J. Chemom. 30, 134–143 (2016).
D’Orazio, M. StatMatch: Statistical Matching or Data Fusion. R package version 1.4.0 (2019).
Goslee, S. C. & Urban, D. L. The ecodist package for dissimilarity-based analysis of ecological data. J. Stat. Softw. 22, 1–22 (2007).
Lefcheck, J. S. piecewiseSEM: piecewise structural equation modelling in R for ecology, evolution and systematics. Methods Ecol. Evol. 7, 573–579 (2016).
Auld, S. K. J. R., Wilson, P. J. & Little, T. J. Rapid change in parasite infection traits over the course of an epidemic in a wild host–parasite population. Oikos 123, 232–238 (2014).
Shocket, M. S. et al. Parasite rearing and infection temperatures jointly influence disease transmission and shape seasonality of epidemics. Ecology 99, 1975–1987 (2018).
Duncan, A. B., Mitchell, S. E. & Little, T. J. Parasite-mediated selection and the role of sex and diapause in Daphnia. J. Evol. Biol. 19, 1183–1189 (2006).
Auld, S. K. J. R. et al. Variation in costs of parasite resistance among natural host populations. J. Evol. Biol. 26, 2479–2486 (2013).
Laine, A.-L. Evolution of host resistance: looking for coevolutionary hotspots at small spatial scales. Proc. R. Soc. B 273, 267–273 (2006).
Lohse, K., Gutierrez, A. & Kaltz, O. Experimental evolution of resistance in Paramecium caudatum against the bacterial parasite Holospora undulata. Evolution 60, 1177–1186 (2006).
Duffy, M. A. & Sivars-Becker, L. Rapid evolution and ecological host–parasite dynamics. Ecol. Lett. 10, 44–53 (2007).
Brewer, M. J., Butler, A. & Cooksley, S. L. The relative performance of AIC, AICC and BIC in the presence of unobserved heterogeneity. Methods Ecol. Evol. 7, 679–692 (2016).
Shipley, B. A new inferential test for path models based on directed acyclic graphs. Struct. Equ. Model. 7, 206–218 (2000).
Acknowledgements
We thank M. Tinsley, S. Thackeray and the Stirling Eco-Evo group for comments on this manuscript. This work was supported by NERC Independent Research Fellowship (NE/L011549/1) and Royal Society Research Grant (RG130657) to S.K.J.R.A.
Author information
Authors and Affiliations
Contributions
Conceptualization: S.K.J.R.A.; data curation: S.K.J.R.A.; formal analysis: S.P. and S.K.J.R.A.; funding acquisition: S.K.J.R.A.; investigation: S.P., J.B. and S.K.J.R.A.; methodology: S.P., J.B. and S.K.J.R.A.; supervision: S.K.J.R.A.; writing original draft: S.P. and S.K.J.R.A.; writing, review and editing: all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Ecology & Evolution thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1 and 2, and Tables 1–3.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Paplauskas, S., Brand, J. & Auld, S.K.J.R. Ecology directs host–parasite coevolutionary trajectories across Daphnia–microparasite populations. Nat Ecol Evol 5, 480–486 (2021). https://doi.org/10.1038/s41559-021-01390-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-021-01390-7
This article is cited by
-
Virulence evolution during a naturally occurring parasite outbreak
Evolutionary Ecology (2023)
-
Environmental variation shapes and links parasitism to sexual selection
Evolutionary Ecology (2023)