Abstract
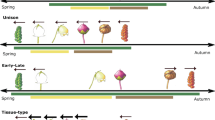
Advancing phenology is one of the most visible effects of climate change on plant communities, and has been especially pronounced in temperature-limited tundra ecosystems. However, phenological responses have been shown to differ greatly between species, with some species shifting phenology more than others. We analysed a database of 42,689 tundra plant phenological observations to show that warmer temperatures are leading to a contraction of community-level flowering seasons in tundra ecosystems due to a greater advancement in the flowering times of late-flowering species than early-flowering species. Shorter flowering seasons with a changing climate have the potential to alter trophic interactions in tundra ecosystems. Interestingly, these findings differ from those of warmer ecosystems, where early-flowering species have been found to be more sensitive to temperature change, suggesting that community-level phenological responses to warming can vary greatly between biomes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study have been archived in the Polar Data Catalogue: https://doi.org/10.21963/12961.
Change history
07 March 2019
In the version of this Article originally published, the following sentence was missing from the Acknowledgements: “This work was supported by the Norwegian Research Council SnoEco project, grant number 230970”. This text has now been added.
References
Fitter, A. H. & Fitter, R. S. R. Rapid changes in flowering time in British plants. Science 296, 1689–1691 (2002).
Parmesan, C. & Yohe, G. A globally coherent fingerprint of climate change impacts across natural systems. Nature 421, 37–42 (2003).
Thackeray, S. J. et al. Phenological sensitivity to climate across taxa and trophic levels. Nature 535, 241–245 (2016).
Arft, A. M. et al. Responses of tundra plants to experimental warming: meta-analysis of the International Tundra Experiment. Ecol. Monogr. 69, 491–511 (1999).
Høye, T. T., Post, E., Meltofte, H., Schmidt, N. M. & Forchhammer, M. C. Rapid advancement of spring in the High Arctic. Curr. Biol. 17, R449–R451 (2007).
Parmesan, C. Influences of species, latitudes and methodologies on estimates of phenological response to global warming. Glob. Change Biol. 13, 1860–1872 (2007).
Oberbauer, S. F. et al. Phenological response of tundra plants to background climate variation tested using the International Tundra Experiment. Phil. Trans. R. Soc. B 368, 20120481 (2013).
Xu, L. et al. Temperature and vegetation seasonality diminishment over northern lands. Nat. Clim. Change 3, 581–586 (2013).
Park, T. et al. Changes in growing season duration and productivity of northern vegetation inferred from long-term remote sensing data. Environ. Res. Lett. 11, 084001 (2016).
Xu, C., Liu, H., Williams, A. P., Yin, Y. & Wu, X. Trends toward an earlier peak of the growing season in Northern Hemisphere mid‐latitudes. Glob. Change Biol. 22, 2852–2860 (2016).
Bradley, N. L., Leopold, A. C., Ross, J. & Huffaker, W. Phenological changes reflect climate change in Wisconsin. Proc. Natl Acad. Sci. USA 96, 9701–9704 (1999).
Høye, T. T., Post, E., Schmidt, N. M., Trøjelsgaard, K. & Forchhammer, M. C. Shorter flowering seasons and declining abundance of flower visitors in a warmer Arctic. Nat. Clim. Change 3, 759–763 (2013).
Bjorkman, A. D., Elmendorf, S. C., Beamish, A. L., Vellend, M. & Henry, G. H. R. Contrasting effects of warming and increased snowfall on Arctic tundra plant phenology over the past two decades. Glob. Change Biol. 21, 4651–4661 (2015).
Panchen, Z. A. & Gorelick, R. Flowering and fruiting responses to climate change of two Arctic plant species, purple saxifrage (Saxifraga oppositifolia) and mountain avens (Dryas integrifolia). Arct. Sci. 1, 45–58 (2015).
Panchen, Z. A. & Gorelick, R. Prediction of Arctic plant phenological sensitivity to climate change from historical records. Ecol. Evol. 7, 1325–1338 (2017).
Price, M. V. & Waser, N. M. Effects of experimental warming on plant reproductive phenology in a subalpine meadow. Ecology 79, 1261–1271 (1998).
Dunne, J. A., Harte, J. & Taylor, K. J. Subalpine meadow flowering phenology responses to climate change: integrating experimental and gradient methods. Ecol. Monogr. 73, 69–86 (2003).
Menzel, A. et al. European phenological response to climate change matches the warming pattern. Glob. Change Biol. 12, 1969–1976 (2006).
Miller-Rushing, A. J. & Inouye, D. W. Variation in the impact of climate change on flowering phenology and abundance: an examination of two pairs of closely related wildflower species. Am. J. Bot. 96, 1821–1829 (2009).
Prevéy, J. S. et al. Greater temperature sensitivity of plant phenology at colder sites: implications for convergence across northern latitudes. Glob. Change Biol. 23, 2660–2671 (2017).
Shaver, G. R. & Kummerow, J. in Arctic Ecosystems in a Changing Climate: An Ecophysiological Perspective (eds Chapin, F. S., Jefferies, R. L., Reynolds, J. F., Shaver, G. R. & Svoboda, J.) 193–211 (Academic Press, San Diego, 1992).
Molau, U. Relationships between flowering phenology and life history strategies in tundra plants. Arct. Alp. Res. 25, 391–402 (1993).
Keller, F. & Körner, C. The role of photoperiodism in alpine plant development. Arct. Antarct. Alp. Res. 35, 361–368 (2003).
Hollister, R. D., Webber, P. J. & Tweedie, C. E. The response of Alaskan arctic tundra to experimental warming: differences between short- and long-term responses. Glob. Change Biol. 11, 525–536 (2005).
Semenchuk, P. R., Elberling, B. & Cooper, E. J. Snow cover and extreme winter warming events control flower abundance of some, but not all species in High Arctic Svalbard. Ecol. Evol. 3, 2586–2599 (2013).
Iler, A. M. et al. Maintenance of temporal synchrony between syrphid flies and floral resources despite differential phenological responses to climate. Glob. Change Biol. 19, 2348–2359 (2013).
Post, E., Kerby, J., Pedersen, C. & Steltzer, H. Highly individualistic rates of plant phenological advance associated with arctic sea ice dynamics. Biol. Lett. 12, 20160332 (2016).
CaraDonna, P. J. & Inouye, D. W. Phenological responses to climate change do not exhibit phylogenetic signal in a subalpine plant community. Ecology 96, 355–361 (2015).
Körner, C. & Basler, D. Phenology under global warming. Science 327, 1461–1462 (2010).
Cleland, E. E. et al. Phenological tracking enables positive species responses to climate change. Ecology 93, 1765–1771 (2012).
Wheeler, H. C., Høye, T. T., Schmidt, N. M., Svenning, J.-C. & Forchhammer, M. C. Phenological mismatch with abiotic conditions—implications for flowering in Arctic plants. Ecology 96, 775–787 (2015).
Inouye, D. W. Effects of climate change on phenology, frost damage, and floral abundance of montane wildflowers. Ecology 89, 353–362 (2008).
Wipf, S., Stoeckli, V. & Bebi, P. Winter climate change in alpine tundra: plant responses to changes in snow depth and snowmelt timing. Clim. Change 94, 105–121 (2009).
Wheeler, J. A. et al. Increased spring freezing vulnerability for alpine shrubs under early snowmelt. Oecologia 175, 219–229 (2014).
Wheeler, J. A. et al. The snow and the willows: earlier spring snowmelt reduces performance in the low-lying alpine shrub Salix herbacea. J. Ecol. 104, 1041–1050 (2016).
Cooper, E. J., Dullinger, S. & Semenchuk, P. Late snowmelt delays plant development and results in lower reproductive success in the High Arctic. Plant Sci. 180, 157–167 (2011).
Parsons, A. N. et al. Growth responses of four sub-Arctic dwarf shrubs to simulated environmental change. J. Ecol. 82, 307–318 (1994).
Molau, U., Nordenhäll, U. & Eriksen, B. Onset of flowering and climate variability in an alpine landscape: a 10-year study from Swedish Lapland. Am. J. Bot. 92, 422–431 (2005).
Grubb, P. J. The maintenance of species-richness in plant communities: the importance of the regeneration niche. Biol. Rev. 52, 107–145 (1977).
Higgins, S. I., Delgado-Cartay, M. D., February, E. C. & Combrink, H. J. Is there a temporal niche separation in the leaf phenology of savanna trees and grasses? J. Biogeogr. 38, 2165–2175 (2011).
Sanz-Aguilar, A., Carrete, M., Edelaar, P., Potti, J. & Tella, J. L. The empty temporal niche: breeding phenology differs between coexisting native and invasive birds. Biol. Invasions 17, 3275–3288 (2015).
Wolkovich, E. M. & Cleland, E. E. The phenology of plant invasions: a community ecology perspective. Front. Ecol. Environ. 9, 287–294 (2011).
Hegland, S. J., Nielsen, A., Lázaro, A. & Bjerknes, A.-L. How does climate warming affect plant–pollinator interactions?. Ecol. Lett. 12, 184–195 (2009).
McKinnon, L., Picotin, M., Bolduc, E., Juillet, C. & Bêty, J. Timing of breeding, peak food availability, and effects of mismatch on chick growth in birds nesting in the High Arctic. Can. J. Zool. 90, 961–971 (2012).
Kerby, J. T. & Post, E. Advancing plant phenology and reduced herbivore production in a terrestrial system associated with sea ice decline. Nat. Commun. 4, 2514 (2013).
Wipf, S. Phenology, growth, and fecundity of eight subarctic tundra species in response to snowmelt manipulations. Plant Ecol. 207, 53–66 (2010).
Post, E., Pedersen, C., Wilmers, C. C. & Forchhammer, M. C. Warming, plant phenology and the spatial dimension of trophic mismatch for large herbivores. Proc. R. Soc. B 275, 2005–2013 (2008).
Schmidt, N. M. et al. An ecological function in crisis? The temporal overlap between plant flowering and pollinator function shrinks as the Arctic warms. Ecography 39, 1250–1252 (2016).
Sherry, R. A. et al. Divergence of reproductive phenology under climate warming. Proc. Natl Acad. Sci. USA 104, 198–202 (2007).
Steltzer, H. & Post, E. Seasons and life cycles. Science 324, 886–887 (2009).
Wolkovich, E. M. et al. Warming experiments underpredict plant phenological responses to climate change. Nature 485, 494–497 (2012).
Prevéy, J. S. & Seastedt, T. R. Seasonality of precipitation interacts with exotic species to alter composition and phenology of a semi-arid grassland. J. Ecol. 102, 1549–1561 (2014).
Diez, J. M. et al. Forecasting phenology: from species variability to community patterns. Ecol. Lett. 15, 545–553 (2012).
Aldridge, G., Inouye, D. W., Forrest, J. R. K., Barr, W. A. & Miller-Rushing, A. J. Emergence of a mid-season period of low floral resources in a montane meadow ecosystem associated with climate change. J. Ecol. 99, 905–913 (2011).
CaraDonna, P. J., Iler, A. M. & Inouye, D. W. Shifts in flowering phenology reshape a subalpine plant community. Proc. Natl Acad. Sci. USA 111, 4916–4921 (2014).
Cook, B. I. et al. Sensitivity of spring phenology to warming across temporal and spatial climate gradients in two independent databases. Ecosystems 15, 1283–1294 (2012).
Høye, T. T. et al. Phenology of High-Arctic butterflies and their floral resources: species-specific responses to climate change. Curr. Zool. 60, 243–251 (2014).
Hocking, B. Insect–flower associations in the High Arctic with special reference to nectar. Oikos 19, 359–387 (1968).
Janzen, D. H. Synchronization of sexual reproduction of trees within the dry season in Central America. Evolution 21, 620–637 (1967).
Meng, F. D. et al. Changes in flowering functional group affect responses of community phenological sequences to temperature change. Ecology 98, 734–740 (2017).
Hulme, P. E. Contrasting impacts of climate-driven flowering phenology on changes in alien and native plant species distributions. New Phytol. 189, 272–281 (2011).
Craine, J. M., Wolkovich, E. M., Gene Towne, E. & Kembel, S. W. Flowering phenology as a functional trait in a tallgrass prairie. New Phytol. 193, 673–682 (2012).
Miller-Rushing, A. J., Høye, T. T., Inouye, D. W. & Post, E. The effects of phenological mismatches on demography. Philos. Trans. R. Soc. Lond. B 365, 3177–3186 (2010).
Elmendorf, S. C. et al. Experiment, monitoring, and gradient methods used to infer climate change effects on plant communities yield consistent patterns. Proc. Natl Acad. Sci. USA 112, 448–452 (2015).
Hollister, R. D. et al. Warming experiments elucidate the drivers of observed directional changes in tundra vegetation. Ecol. Evol. 5, 1881–1895 (2015).
Molau, U. & Mølgaard, P. International Tundra Experiment (ITEX) Manual (Danish Polar Center, 1996).
Henry, G. H. R. & Molau, U. Tundra plants and climate change: the International Tundra Experiment (ITEX). Glob. Change Biol. 3, 1–9 (1997).
Harris, I., Jones, P, Osborn, T. J. & Lister, D. H. Updated high-resolution grids of monthly climatic observations—the CRU TS3.10 dataset. Int. J. Climatol. 34, 623–642 (2014).
Marion, G. M. et al. Open-top designs for manipulating field temperature in high-latitude ecosystems. Glob. Change Biol. 3, 20–32 (1997).
Hollister, R. D., Webber, P. J., Nelson, F. E. & Tweedie, C. E. Soil thaw and temperature response to air warming varies by plant community: results from an open-top chamber experiment in Northern Alaska. Arct. Antarct. Alp. Res. 38, 206–215 (2006).
Walker, M. D. et al. Plant community responses to experimental warming across the tundra biome. Proc. Natl Acad. Sci. USA 103, 1342–1346 (2006).
Latimer, A. M. Geography and resource limitation complicate metabolism-based predictions of species richness. Ecology 88, 1895–1898 (2007).
Stan Modeling Language User’s Guide and Reference Manual Version 2.17.0 (Stan Development Team, 2017).
RStan: the R Interface to Stan R Package Version 2.17.3 (Stan Development Team, 2018).
R Development Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2017).
Gelman, A. & Rubin, D. B. Inference from iterative simulation using multiple sequences. Stat. Sci. 7, 457–472 (1992).
Miller-Rushing, A. J., Inouye, D. W. & Primack, R. B. How well do first flowering dates measure plant responses to climate change? The effects of population size and sampling frequency. J. Ecol. 96, 1289–1296 (2008).
Acknowledgements
We are grateful to the many individuals who established experiments and collected detailed phenological observations. There are too many to name them all; however, we especially thank: M. Dalle Fratte, D. Cooley, O. Durey, C. Eckert, J. F. Johnstone, C. Kennedy, V. Lamarre, G. Levasseur, C. Spiech, J. Svoboda and R. Wising; the Herschel Island Qikiqtaruk Territorial Park staff, including E. McLeod, S. McLeod, R. Joe, P. Lennie, D. Arey, L. Meyook, J. McLeod, P. Foisy, C. Gordon, J. Hansen, A. Rufus and R. Gordon; Quttinirpaaq National Park staff; the Greenland Ecosystem Monitoring team; and Warming and species Removal in Mountains (WaRM) coordinators N. Sanders, A. Classen and M. Sundqvist. These observations were made possible with the support of many funding agencies and grants, including: ArcticNet; the Natural Sciences and Engineering Research Council of Canada; the Canadian International Polar Year Program; the Polar Continental Shelf Program of Natural Resources Canada; the Danish Environmental Protection Agency; the Swiss Federal Institute for Forest, Snow and Landscape Research; the National Geographic Society; the US National Science Foundation (grant numbers PLR1525636, PLR1504141, PLR1433063, PLR1107381, PLR0119279, PLR0902125, PLR0856728, PLR1312402, PLR1019324, LTER 1026415, OPP1525636, OPP9907185, DEB1637686, 0856710, 9714103, 0632263, 0856516, 1432277, 1432982, 1504381, 1504224, 1433063, 0856728, 0612534, 0119279 and 9421755; the Danish National Research Foundation (grant CENPERM DNRF100); the Danish Council for Independent Research (Natural Sciences grant DFF 4181-00565); the Deutsche Forschungsgemeinschaft (grant: RU 1536/3-1); the Natural Environment Research Council (grant NE/M016323/1); the Department of Energy (grant SC006982); a Semper Ardens grant from the Carlsberg Foundation to N. J. Sanders; and an INTERACT Transnational Access grant. This work was supported by the Norwegian Research Council SnoEco project, grant number 230970.
Author information
Authors and Affiliations
Contributions
J.S.P. and C.R. designed and led the study. J.S.P. and C.R. led the collection of data for the phenology database. J.S.P., N.R., A.D.B., I.H.M.-S. and S.C.E. performed the statistical analyses. J.S.P., C.R., N.R., T.T.H., A.D.B., I.H.M.-S. and S.C.E. drafted the paper. J.S.P., C.R., A.D.B., I.H.M.-S., I.W.A., N.C., K.C., C.C., E.J.C., B.E., A.M.F., G.H.R.H., R.D.H., I.S.J., K.K., C.W.K., E.L., M.M., U.M., S.N., S.O., Z.A.P., E.P., S.B.R., N.M.S., E.S., P.R.S., J.G.S., K.N.S., Ø.T., T.T., S.V., C.-H.W., J.M.W. and S.W. contributed data. All authors were involved in writing and editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–7, Supplementary Figure 1, Supplementary Code and Supplementary References
Rights and permissions
About this article
Cite this article
Prevéy, J.S., Rixen, C., Rüger, N. et al. Warming shortens flowering seasons of tundra plant communities. Nat Ecol Evol 3, 45–52 (2019). https://doi.org/10.1038/s41559-018-0745-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-018-0745-6
This article is cited by
-
Plasticity and not adaptation is the primary source of temperature-mediated variation in flowering phenology in North America
Nature Ecology & Evolution (2024)
-
Warming in combination with increased precipitation mediate the sexual and clonal reproduction in the desert steppe dominant species Stipa breviflora
BMC Plant Biology (2023)
-
Virus diversity and activity is driven by snowmelt and host dynamics in a high-altitude watershed soil ecosystem
Microbiome (2023)
-
Diverging trends and drivers of Arctic flower production in Greenland over space and time
Polar Biology (2023)
-
Climate warming–driven phenological shifts are species-specific in woody plants: evidence from twig experiment in Kashmir Himalaya
International Journal of Biometeorology (2022)