Abstract
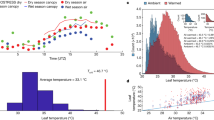
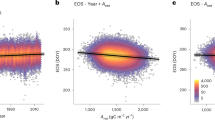
Tropical forest leaf albedo (reflectance) greatly impacts how much energy the planet absorbs; however; little is known about how it might be impacted by climate change. Here, we measure leaf traits and leaf albedo at ten 1-ha plots along a 3,200-m elevation gradient in Peru. Leaf mass per area (LMA) decreased with warmer temperatures along the elevation gradient; the distribution of LMA was positively skewed at all sites indicating a shift in LMA towards a warmer climate and future reduced tropical LMA. Reduced LMA was significantly (P < 0.0001) correlated with reduced leaf near-infrared (NIR) albedo; community-weighted mean NIR albedo significantly (P < 0.01) decreased as temperature increased. A potential future 2 °C increase in tropical temperatures could reduce lowland tropical leaf LMA by 6–7 g m−2 (5–6%) and reduce leaf NIR albedo by 0.0015–0.002 units. Reduced NIR albedo means that leaves are darker and absorb more of the Sun’s energy. Climate simulations indicate this increased absorbed energy will warm tropical forests more at high CO2 conditions with proportionately more energy going towards heating and less towards evapotranspiration and cloud formation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All the data in this paper can be found in a data repository at the following website: https://ora.ox.ac.uk/objects/uuid:4101e249-3cf5-443f-9c29-9204604c667b.
References
Betts, R. Implications of land ecosystem-atmosphere interactions for strategies for climate change adaptation and mitigation. Tellus B 59, 602–615 (2007).
Loarie, S. R., Lobell, D. B., Asner, G. P. & Field, C. B. Land-cover and surface water change drive large albedo increases in South America. Earth Interact. 15, 1–16 (2011).
Asner, G. P. Biophysical and biochemical sources of variability in canopy reflectance. Remote Sens. Environ. 64, 234–253 (1998).
Serbin, S. P., Singh, A., McNeil, B. E., Kingdon, C. C. & Townsend, P. A. Spectroscopic determination of leaf morphological and biochemical traits for northern temperate and boreal tree species. Ecol. Appl. 24, 1651–1669 (2014).
Richardson, A. D., Duigan, S. P. & Berlyn, G. P. An evaluation of noninvasive methods to estimate foliar chlorophyll content. New Phytol. 153, 185–194 (2002).
Jacquemoud, S. & Baret, F. PROSPECT: a model of leaf optical properties spectra. Remote Sens. Environ. 34, 75–91 (1990).
Asner, G. P. & Martin, R. E. Spectral and chemical analysis of tropical forests: scaling from leaf to canopy levels. Remote Sens. Environ. 112, 3958–3970 (2008).
Enquist, B. J. et al. Scaling from traits to ecosystems: developing a general trait driver theory via integratingtrait-based and metabolic scaling theories. Adv. Ecol. Res. 52, 249–318 (2015).
Enquist, B. J. et al. Assessing trait-based scaling theory in tropical forests spanning a broad temperature gradient. Glob. Ecol. Biogeogr. 26, 1357–1373 (2017).
Savage, V. M., Webb, C. T. & Norberg, J. A general multi-trait-based framework for studying the effects of biodiversity on ecosystem functioning. J. Theor. Biol. 247, 213–229 (2007).
Norberg, J. et al. Phenotypic diversity and ecosystem functioning in changing environments: a theoretical framework. Proc. Natl Acad. Sci. USA 98, 11376–11381 (2001).
Asner, G. P. et al. Scale dependence of canopy trait distributions along a tropical forest elevation gradient. New Phytol. 214, 973–988 (2017).
Feeley, K. J. et al. Upslope migration of Andean trees. J. Biogeogr. 38, 783–791 (2011).
Feeley, K. J. Distributional migrations, expansions, and contractions of tropical plant species as revealed in dated herbarium records. Glob. Chang. Biol. 18, 1335–1341 (2012).
Asner, G. P. & Martin, R. E. Convergent elevation trends in canopy chemical traits of tropical forests. Glob. Chang. Biol. 22, 2216–2227 (2016).
Körner, C. H., Bannister, P. & Mark, A. F. Altitudinal variation in stomatal conductance, nitrogen content and leaf anatomy in different plant life forms in New Zealand. Oecologia 69, 577–588 (1986).
Roderick, M. L., Berry, S. L. & Noble, I. R. A framework for understanding the relationship between environment and vegetation based on the surface area to volume ratio of leaves. Funct. Ecol. 14, 423–437 (2000).
Atkin, O. K., Loveys, B. R., Atkinson, L. J. & Pons, T. L. Phenotypic plasticity and growth temperature: understanding interspecific variability. J. Exp. Bot. 57, 267–281 (2006).
Ball, M. C. et al. Space and time dependence of temperature and freezing in evergreen leaves. Funct. Plant Biol. 29, 1259–1272 (2002).
Poorter, H., Niinemets, Ü., Poorter, L., Wright, I. J. & Villar, R. Causes and consequences of variation in leaf mass per area (LMA): a meta-analysis. New Phytol. 182, 565–588 (2009).
Asner, G. P. et al. Taxonomy and remote sensing of leaf mass per area (LMA) in humid tropical forests. Ecol. Appl. 21, 85–98 (2011).
Niinemets, Ü. Research review. Components of leaf dry mass per area—thickness and density—alter leaf photosynthetic capacity in reverse directions in woody plants. New Phytol. 144, 35–47 (1999).
Féret, J.-B. et al. PROSPECT-4 and 5: advances in the leaf optical properties model separating photosyntheticpigments. Remote Sens. Environ. 112, 3030–3043 (2008).
Féret, J-B., Gitelson, A. A., Noble, S. D. & Jacquemoud, S. PROSPECT-D: towards modeling leaf optical properties through a complete lifecycle. Remote Sens. Environ. 193, 204–215 (2017).
Ceccato, P., Flasse, S., Tarantola, S., Jacquemoud, S. & Grégoire, J-M. Detecting vegetation leaf water content using reflectance in the optical domain. Remote Sens. Environ. 77, 22–33 (2001).
Collins, W. D. et al. The Community Climate System Model version 3 (CCSM3). J. Clim. 19, 2122–2143 (2006).
Dickinson, R. E., Sellers, P. J. & Kimes, D. S. Albedos of homogeneous semi-infinite canopies: comparison of two-stream analytic and numerical solutions. J. Geophys. Res. Atmos. 92, 4282–4286 (1987).
Doughty, C. E., Field, C. B. & McMillan, A. M. S. Can crop albedo be increased through the modification of leaf trichomes, and could this cool regional climate? Clim. Change 104, 379–387 (2011).
Collins, M. et al. Climate Change 2013: The Physical Science Basis (eds Stocker, T. F. et al.) Ch. 29 (IPCC, Cambridge Univ. Press, 2013).
Doughty, C. E. et al. Can leaf spectroscopy predict leaf and forest traits along a Peruvian tropical forest elevation gradient? J. Geophys. Res. Biogeosci. 122, 2952–2965 (2017).
Swann, A. L. S., Fung, I. Y. & Chiang, J. C. H. Mid-latitude afforestation shifts general circulation and tropical precipitation. Proc. Natl Acad. Sci. USA 109, 712–716 (2012).
Doughty, C. E., Loarie, S. R. & Field, C. B. Theoretical impact of changing albedo on precipitation at the southernmost boundary of the ITCZ in South America. Earth Interact. 16, 1–14 (2012).
Brienen, R. J. W. et al. Long-term decline of the Amazon carbon sink. Nature 519, 344–348 (2015).
Wright, I. J. et al. The worldwide leaf economics spectrum. Nature 428, 821–827 (2004).
Chadwick, D. K. & Asner, G. P. Organismic-scale remote sensing of canopy foliar traits in lowland tropical forests. Remote Sens. 8, 87 (2016).
Fyllas, N. M. et al. Basin-wide variations in foliar properties of Amazonian forest: phylogeny, soils and climate. Biogeosciences 6, 2677–2708 (2009).
Reich, P. B. & Flores-Moreno, H. Peeking beneath the hood of the leaf economics spectrum. New Phytol. 214, 1395–1397 (2017).
Onoda, Y. et al. Physiological and structural tradeoffs underlying the leaf economics spectrum. New Phytol. 214, 1447–1463 (2017).
Chavana-Bryant, C. et al. Leaf aging of Amazonian canopy trees as revealed by spectral and physiochemical measurements. New Phytol. 214, 1049–1063 (2017).
Huang, M. et al. Velocity of change in vegetation productivity over northern high latitudes. Nat. Ecol. Evol. 1, 1649–1654 (2017).
Malhi, Y. & Wright, J. Spatial patterns and recent trends in the climate of tropical rainforest regions. Phil.Trans. R. Soc. B 359, 311–329 (2004).
Duffy, P. B., Brando, P., Asner, G. P. & Field, C. B. Projections of future meteorological drought and wet periods in the Amazon. Proc. Natl Acad. Sci. USA 112, 13172–13177 (2015).
Thomas, S. C. Increased leaf reflectance in tropical trees under elevated CO2. Glob. Chang. Biol. 11, 197–202 (2005).
Michaletz, S. T. et al. The energetic and carbon economic origins of leaf thermoregulation. Nat. Plants 2, 16129 (2016).
Helliker, B. R. & Richter, S. L. Subtropical to boreal convergence of tree-leaf temperatures. Nature 454, 511–514 (2008).
Keenan, T. F. et al. Increase in forest water-use efficiency as atmospheric carbon dioxide concentrations rise. Nature 499, 324–327 (2013).
van der Sleen, P. et al. No growth stimulation of tropical trees by 150 years of CO2 fertilization but water-use efficiency increased. Nat. Geosci. 8, 24–28 (2015).
Doughty, C. E. & Goulden, M. L. Are tropical forests near a high temperature threshold?. J. Geophys. Res. Biogeosci 113, G00B07 (2008).
Mau, A. C., Reed, S. C., Wood, T. E. & Cavaleri, M. A. Temperate and tropical forest canopies are already functioning beyond their thermal thresholds for photosynthesis. Forests 9, 47 (2018).
Zhu, P. et al. Elevated atmospheric CO2 negatively impacts photosynthesis through radiative forcing and physiology-mediated climate feedback. Geophys. Res. Lett. 44, 1956–1963 (2017).
GEMTraits: A Database and R Package for Accessing and Analyzing Plant Functional Traits from the Global Ecosystems Monitoring Network version 1 (Univ. Oxford, 2017); https://ora.ox.ac.uk/objects/uuid:4101e249-3cf5-443f-9c29-9204604c667b
Malhi, Y. et al. The variation of productivity and its allocation along a tropical elevation gradient: a whole carbon budget perspective. New Phytol. 214, 1019–1032 (2017).
Hurrell, J. W., Hack, J. J., Shea, D., Caron, J. M. & Rosinski, J. A new sea surface temperature and sea ice boundary dataset for the community atmosphere model. J. Clim. 21, 5145–5153 (2008).
IPCC Climate Change 2014: Synthesis Report (eds Core Writing Team, Pachauri, R. K. & Meyer L. A.) (IPCC, 2014).
Olivier, J. G. J., Janssens-Maenhout, G., Muntean, M. & Peters, J. A. H. W. Trends in Global CO 2 Emissions: 2016 Report (PBL Netherlands Environmental Assessment Agency and European Commission, Joint Research Centre, 2016); http://edgar.jrc.ec.europa.eu/news_docs/jrc-2016-trends-in-global-co2-emissions-2016-report-103425.pdf
Acknowledgements
This work is a product of the GEM network (gem.tropicalforests.ox.ac.uk), ABERG (andesresearch.org), the Amazon Forest Inventory Network (www.rainfor.org) and the Carnegie Spectranomics Project (spectranomics.carnegiescience.edu) research consortia. The field campaign was funded by a grant to Y.M. from the UK Natural Environment Research Council (NERC) (grant no. NE/J023418/1), with additional support from European Research Council advanced investigator grants GEM-TRAITS (no. 321131) and T-FORCES (no. 291585), and a John D. and Catherine T. MacArthur Foundation grant to G.P.A. We thank the Servicio Nacional de Áreas Naturales Protegidas por el Estado and the personnel of the Manu and Tambopata National Parks for logistical assistance and permission to work in the protected areas. We also thank the Explorers’ Inn and the Pontifical Catholic University of Peru, as well as Asociación para la Conservación de la Cuenca Amazónica. We thankE. Cosio (Pontifical Catholic University of Peru) for his assistance with research permissions and sample analysis and storage. Taxonomic work at the Carnegie Institution was helped by R. Tupayachi, F. Sinca and N. Jaramillo. B.B. was supported by a United States National Science Foundation (NSF) graduate research fellowship and doctoral dissertation improvement grant (no. DEB-1209287), as well as an NERC independent research fellowship (grant no. NE/M019160/1). G.P.A. and the Spectranomics team were supported by the endowment of the Carnegie Institution for Science and a grant from the NSF (no. DEB-1146206). S.D. was partially supported by a Visiting Professorship grant from the Leverhulme Trust, UK. Y.M. was also supported by the Jackson Foundation. G.R.G. was supported by funding from the European Community’s Seventh Framework Program (FP7/2007–2013) under grant agreement no. 290605 (COFUND: PSI-FELLOW). C.E.D. received funding from the John Fell Fund, Google and a NASA grant (no. 80NSSC17K0749). Climate simulations were run on Monsoon, Northern Arizona University’s supercomputer.
Author information
Authors and Affiliations
Contributions
C.E.D. wrote the paper with contributions from G.P.A., B.B., G.R.G. and R.E.M. P.E.S.-A. and C.E.D. collected the spectral data. P.E.S.-A., A.S., L.P.B., G.R.G., B.B., N.S., B.J.E., R.E.M., G.P.A., S.D. and Y.M. provided data or support. C.E.D. analysed the data and ran the climate and leaf reflectance simulations.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures and Tables
Rights and permissions
About this article
Cite this article
Doughty, C.E., Santos-Andrade, P.E., Shenkin, A. et al. Tropical forest leaves may darken in response to climate change. Nat Ecol Evol 2, 1918–1924 (2018). https://doi.org/10.1038/s41559-018-0716-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-018-0716-y
This article is cited by
-
Joint optimization of land carbon uptake and albedo can help achieve moderate instantaneous and long-term cooling effects
Communications Earth & Environment (2023)
-
Robust leaf trait relationships across species under global environmental changes
Nature Communications (2020)