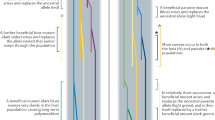
Abstract
The parasite Leishmania donovani causes a fatal disease termed visceral leishmaniasis. The process through which the parasite adapts to environmental change remains largely unknown. Here we show that aneuploidy is integral for parasite adaptation and that karyotypic fluctuations allow for selection of beneficial haplotypes, which impact transcriptomic output and correlate with phenotypic variations in proliferation and infectivity. To avoid loss of diversity following karyotype and haplotype selection, L. donovani utilizes two mechanisms: polyclonal selection of beneficial haplotypes to create coexisting subpopulations that preserve the original diversity, and generation of new diversity as aneuploidy-prone chromosomes tolerate higher mutation rates. Our results reveal high aneuploidy turnover and haplotype selection as a unique evolutionary adaptation mechanism that L. donovani uses to preserve genetic diversity under strong selection. This unexplored process may function in other human diseases, including fungal infection and cancer, and stimulate innovative treatment options.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Pallen, M. J. & Wren, B. W. Bacterial pathogenomics. Nature 449, 835–842 (2007).
Torres, E. M. et al. Effects of aneuploidy on cellular physiology and cell division in haploid yeast. Science 317, 916–924 (2007).
Pavelka, N. et al. Aneuploidy confers quantitative proteome changes and phenotypic variation in budding yeast. Nature 468, 321–325 (2010).
Pavelka, N., Rancati, G. & Li, R. Dr Jekyll and Mr Hyde: role of aneuploidy in cellular adaptation and cancer. Curr. Opin. Cell Biol. 22, 809–815 (2010).
Selmecki, A. M., Dulmage, K., Cowen, L. E., Anderson, J. B. & Berman, J. Acquisition of aneuploidy provides increased fitness during the evolution of antifungal drug resistance. PLoS Genet. 5, e1000705 (2009).
Sabeti, P. C. et al. Detecting recent positive selection in the human genome from haplotype structure. Nature 419, 832–837 (2002).
Staaf, J. et al. Landscape of somatic allelic imbalances and copy number alterations in human lung carcinoma. Int. J. Cancer 132, 2020–2031 (2013).
Scheinfeldt, L. B. & Tishkoff, S. A. Recent human adaptation: genomic approaches, interpretation and insights. Nat. Rev. Genet. 14, 692–702 (2013).
Gao, R. et al. Punctuated copy number evolution and clonal stasis in triple-negative breast cancer. Nat. Genet. 48, 1119–1130 (2016).
Sterkers, Y., Crobu, L., Lachaud, L., Pagès, M. & Bastien, P. Parasexuality and mosaic aneuploidy in Leishmania: alternative genetics. Trends Parasitol. 30, 429–435 (2014).
Imamura, H. et al. Evolutionary genomics of epidemic visceral leishmaniasis in the Indian subcontinent. Elife 5, e12613 (2016).
Alvar, J. et al. Leishmaniasis worldwide and global estimates of its incidence. PLoS ONE 7, e35671 (2012).
Zilberstein, D. & Shapira, M. The role of pH and temperature in the development of Leishmania parasites. Annu. Rev. Microbiol. 48, 449–470 (1994).
Leprohon, P., Fernandez-Prada, C., Gazanion, É., Monte-Neto, R. & Ouellette, M. Drug resistance analysis by next generation sequencing in Leishmania. Int. J. Parasitol. Drugs Drug Resist. 5, 26–35 (2015).
Downing, T. et al. Whole genome sequencing of multiple Leishmania donovani clinical isolates provides insights into population structure and mechanisms of drug resistance. Genome Res. 21, 2143–2156 (2011).
Dujardin, J.-C., Mannaert, A., Durrant, C. & Cotton, J. A. Mosaic aneuploidy in Leishmania: the perspective of whole genome sequencing. Trends Parasitol. 30, 554–555 (2014).
Rogers, M. B. et al. Chromosome and gene copy number variation allow major structural change between species and strains of Leishmania. Genome Res. 21, 2129–2142 (2011).
Sterkers, Y., Lachaud, L., Crobu, L., Bastien, P. & Pagès, M. FISH analysis reveals aneuploidy and continual generation of chromosomal mosaicism in Leishmania major. Cell. Microbiol. 13, 274–283 (2011).
Dumetz, F. et al. Modulation of aneuploidy in Leishmania donovani during adaptation to different in vitro and in vivo environments and its impact on gene expression. MBio 8, e00599-17 (2017).
Legrand, M. et al. Haplotype mapping of a diploid non-meiotic organism using existing and induced aneuploidies. PLoS Genet. 4, e1 (2008).
Yona, A. H. et al. Chromosomal duplication is a transient evolutionary solution to stress. Proc. Natl Acad. Sci. USA 109, 21010–21015 (2012).
Sterkers, Y. et al. Novel insights into genome plasticity in eukaryotes: mosaic aneuploidy in Leishmania. Mol. Microbiol. 86, 15–23 (2012).
Rougeron, V., De Meeûs, T., Kako Ouraga, S., Hide, M. & Bañuls, A.-L. ‘Everything you always wanted to know about sex (but were afraid to ask)’ in Leishmania after two decades of laboratory and field analyses. PLoS Pathog. 6, e1001004 (2010).
Lande, R. Natural selection and random genetic drift in phenotypic evolution. Evolution 30, 314–334 (1976).
Lynch, M. & Conery, J. S. The evolutionary fate and consequences of duplicate genes. Science 290, 1151–1155 (2000).
Victoir, K. & Dujardin, J.-C. How to succeed in parasitic life without sex? Asking Leishmania. Trends Parasitol. 18, 81–85 (2002).
Bastien, P., Blaineau, C. & Pages, M. Leishmania: sex, lies and karyotype. Parasitol. Today 8, 174–177 (1992).
Dujardin, J.-C. et al. Clonal propagation and the fast generation of karyotype diversity: an in vitro Leishmania model. Parasitology 134, 33–39 (2007).
Gazanion, É., Fernández-Prada, C., Papadopoulou, B., Leprohon, P. & Ouellette, M. Cos-Seq for high-throughput identification of drug target and resistance mechanisms in the protozoan parasite Leishmania. Proc. Natl Acad. Sci. USA 113, E3012–E3021 (2016).
Pescher, P., Blisnick, T., Bastin, P. & Späth, G. F. Quantitative proteome profiling informs on phenotypic traits that adapt Leishmania donovani for axenic and intracellular proliferation. Cell. Microbiol. 13, 978–991 (2011).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Mortazavi, A., Williams, B. A., McCue, K., Schaeffer, L. & Wold, B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods 5, 621–628 (2008).
Acknowledgements
Work on samples from the Indian subcontinent was supported by the European Union 7th Framework Programme (EU FP7) (Kaladrug-R, contract 222895), the Belgian Science Policy Office (TRIT, P7/41), the Department of Economy, Science and Innovation in Flanders (ITM-SOFIB) and the Flemish Fund for Scientific Research (G.0.B81.12). We thank Life Science Editors for editing assistance. This study recieved funding from Plan Nacional (BFU2011-28575 to C.N., P.P.B. and D.K.), Center for Genomic Regulation (CRG), Spanish Ministry of Economy and Competitiveness, ‘Centro de Excelencia Severo Ochoa 2013–2017’ (SEV-2012–0208), a grant from the Institut Pasteur International Department strategic fund to the LeiSHield consortium (P.P.B., P.P, C.N., G.F.S., G.B. and Y.S.), Agence Nationale de la Recherche (ANR) within the framework of the 'Investissements d’avenir' programme (ANR-11-LABX-0024-01 “PARAFRAP”) (V.C., M.P. and Y.S.), the EU FP7 (Kaladrug-R, contract 222895), the Belgian Science Policy Office (TRIT, P7/41), the Department of Economy, Science and Innovation in Flanders (ITM-SOFIB) and the Flemish Fund for Scientific Research (G.0.B81.12) (J.C.D., F.D., H.I. and M.D.).
Author information
Authors and Affiliations
Contributions
P.P.B., P.P, C.N. and G.F.S worked on all aspects of work, contributed to the design of the project and wrote the article. G.B contributed to in silico analysis. F.D. and M.D. managed the hamster infection experiment with field isolates. H.I., M.D. and J.-C.D. contributed to the field isolates analyses and revised the paper. D.K. helped analyse the sequencing data. H.H. was responsible for the genomic sequencing of the in vitro clones. V.C., M.P. and Y.S. contributed the DNA-FISH analysis.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary Figures 1–12, Supplementary Tables 1–2.
Rights and permissions
About this article
Cite this article
Prieto Barja, P., Pescher, P., Bussotti, G. et al. Haplotype selection as an adaptive mechanism in the protozoan pathogen Leishmania donovani . Nat Ecol Evol 1, 1961–1969 (2017). https://doi.org/10.1038/s41559-017-0361-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-017-0361-x
This article is cited by
-
Leishmania genetic exchange is mediated by IgM natural antibodies
Nature (2023)
-
Laboratory diagnostics for human Leishmania infections: a polymerase chain reaction-focussed review of detection and identification methods
Parasites & Vectors (2022)
-
Effects of aneuploidy on cell behaviour and function
Nature Reviews Molecular Cell Biology (2022)
-
Colonization and genetic diversification processes of Leishmania infantum in the Americas
Communications Biology (2021)
-
Evaluation of whole genome amplification and bioinformatic methods for the characterization of Leishmania genomes at a single cell level
Scientific Reports (2020)