Abstract
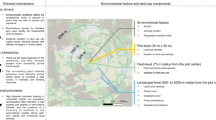
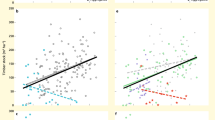
Biodiversity affects many ecosystem functions and services, including carbon cycling and retention. While it is known that the efficiency of carbon capture and biomass production by ecological communities increases with species diversity, the role of vertebrate animals in the carbon cycle remains undocumented. Here, we use an extensive dataset collected in a high-diversity Amazonian system to parse out the relationship between animal and plant species richness, feeding interactions, tree biomass and carbon concentrations in soil. Mammal and tree species richness is positively related to tree biomass and carbon concentration in soil—and the relationship is mediated by organic remains produced by vertebrate feeding events. Our research advances knowledge of the links between biodiversity and carbon cycling and storage, supporting the view that whole community complexity—including vertebrate richness and trophic interactions—drives ecosystem function in tropical systems. Securing animal and plant diversity while protecting landscape integrity will contribute to soil nutrient content and carbon retention in the biosphere.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
19 October 2017
In the version of this Article originally published, the surname of Ted K. Raab was misspelt. This error has now been corrected in all versions of the Article.
References
Cardinale, B. J. et al. Biodiversity loss and its impact on humanity. Nature 486, 59–67 (2012).
Aarssen, L. W. High productivity in grassland ecosystems: effected by species diversity or productive species? Oikos 80, 183–184 (1997).
Tilman, D., Lehman, C. L. & Thomson, K. T. Plant diversity and ecosystem productivity: theoretical considerations. Proc. Natl Acad. Sci. USA 94, 1857–1861 (1997).
Cardinale, B. J. et al. The functional role of producer diversity in ecosystems. Am. J. Bot. 98, 572–592 (2011).
Fornara, D. A. & Tilman, D. Plant functional composition influences rates of soil carbon and nitrogen accumulation. J. Ecol. 96, 314–322 (2008).
Cong, W. F. et al. Plant species richness promotes soil carbon and nitrogen stocks in grasslands without legumes. J. Ecol. 102, 1163–1170 (2014).
Poorter, L. et al. Diversity enhances carbon storage in tropical forests. Glob. Ecol. Biogeogr. 24, 1314–1328 (2015).
Estes, J. A. et al. Trophic downgrading of planet Earth. Science 333, 301–306 (2011).
Lange, M. et al. Plant diversity increases soil microbial activity and soil carbon storage. Nat. Commun. 6, 6707 (2015).
Fierer, N. et al. Reconstructing the microbial diversity and function of pre-agricultural tallgrass prairie soils in the United States. Science 342, 621–624 (2013).
Marichal, R. et al. Soil macroinvertebrate communities and ecosystem services in deforested landscapes of Amazonia. Appl. Soil. Ecol. 83, 177–185 (2014).
Allgeier, J. E., Layman, C. A., Mumby, P. J. & Rosemond, A. D. Biogeochemical implications of biodiversity and community structure across multiple coastal ecosystems. Ecol. Monogr. 85, 117–132 (2015).
Bello, C. et al. Defaunation affects carbon storage in tropical forests. Sci. Adv. 1, e1501105 (2015).
Dos Santos Neves, N., Feer, N., Salmon, S., Chateil, C. & Ponge, C. J. F. The impact of red howler monkey latrines on the distribution of main nutrients and on topsoil profiles in a tropical rainforest. Austral Ecol. 35, 549–559 (2010).
Sitters, J., Maechler, M. J., Edwards, P. J., Suter, W. & Venterink, H. O. Interactions between C:N:P stoichiometry and soil macrofauna control dung decomposition of savanna herbivores. Funct. Ecol. 28, 776–786 (2014).
Barton, P. S., Cunningham, S. A., Lindenmayer, D. B. & Manning, A. D. The role of carrion in maintaining biodiversity and ecological processes in terrestrial ecosystems. Oecologia 171, 761–772 (2013).
Parmenter, R. R. & MacMahon, J. A. Carrion decomposition and nutrient cycling in a semiarid shrub-steppe ecosystem. Ecol. Monogr. 79, 637–661 (2009).
Hawlena, D., Strickland, M. S., Bradford, M. A. & Schmitz, O. J. Fear of predation slows plant-litter decomposition. Science 336, 1434–1438 (2012).
Lehmann, J. & Kleber, M. The contentious nature of soil organic matter. Nature 528, 60–68 (2015).
Frank, D. A., Depriest, T., McLauchlan, K. & Risch, A. C. Topographic and ungulate regulation of soil C turnover in a temperate grassland ecosystem. Glob. Change Biol. 17, 495–504 (2011).
Ritchie, M. E. Plant compensation to grazing and soil carbon dynamics in a tropical grassland. PeerJ 2, e233 (2014).
McSherry, M. E. & Ritchie, M. E. Effects of grazing on grassland soil carbon: a global review. Glob. Change Biol. 19, 1347–1357 (2013).
Hooper, D. U., Schulze, E. D. & Mooney, H. A. Biodiversity and Ecosystem Function (Springer, Berlin, 1993).
Fragoso, J. M. V. & Huffman, J. M. Seed-dispersal and seedling recruitment patterns by the last Neotropical megafaunal element in Amazonia, the tapir. J. Trop. Ecol. 16, 369–385 (2000).
Stevenson, P. R. & Guzmán-Caro, D. C. Nutrient transport within and between habitats through seed dispersal processes by woolly monkeys in north-western Colombia. Am. J. Primatol. 72, 992–1003 (2010).
Nichols, E. et al. Ecological functions and ecosystem services provided by Scarabaeinae dung beetles. Biol. Conserv. 141, 1461–1474 (2008).
Kurten, E. L. Cascading effects of contemporaneous defaunation on tropical forest communities. Biol. Conserv. 163, 22–32 (2013).
Kurten, E. L., Wright, S. J. & Carson, W. P. Hunting alters seedling functional trait composition in a neotropical forest. Ecology 96, 1923–1932 (2015).
Hoehn, P., Tscharntke, T., Tylianakis, J. M. & Steffan-Dewenter, I. Functional group diversity of bee pollinators increases crop yield. Proc. R. Soc. B 275, 2283–2291 (2008).
Lefcheck, J. S. et al. Biodiversity enhances ecosystem multifunctionality across trophic levels and habitats. Nat. Commun. 6, 6936 (2015).
Iwamura, T., Lambin, E. F., Silvius, K. M., Luzar, J. B. & Fragoso, J. M. V. Agent-based modeling of hunting and subsistence agriculture on indigenous lands: understanding interactions between social and ecological systems. Environ. Model. Softw. 58, 109–127 (2014).
Read, J. M. et al. Space, place, and hunting patterns among indigenous peoples of the Guyanese Rupununi region. J. Lat. Am. Geogr. 9, 213–243 (2010).
Luzar, J. B. et al. Large-scale environmental monitoring by indigenous peoples. BioScience. 6, 771–781 (2011).
Luzar, J. B., Silvius, K. M. & Fragoso, J. M. V. Church affiliation and meat taboos in indigenous communities of Guyanese Amazonia. Hum. Ecol. 40, 833–845 (2012).
Fragoso, J. M. V. et al. Line transect surveys underdetect terrestrial mammals: implications for the sustainability of subsistence hunting. PLoS ONE 11, e0152659 (2016).
Hijmans, R. J., Cameron, S. E., Parra, J. L., Jones, P. G. & Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25, 1965–1978 (2005).
Anderson, D. R., Laake, J. L., Crain, B. R. & Burnham, K. P. Guidelines for line transect sampling of biological populations. J. Wildlife Manage. 43, 70–78 (1979).
Butt, N., Epps, K., Overman, H., Iwamura, T. & Fragoso, J. M. V. Assessing carbon stocks using indigenous peoples’ field measurements in Amazonian Guyana. Forest Ecol. Manage. 338, 191–199 (2015).
Batjes, N. H. Total carbon and nitrogen in the soils of the world. Eur. J. Soil Sci. 47, 151–163 (1996).
Silva, L. C. R. et al. Can savannas become forests? A coupled analysis of nutrient stocks and fire thresholds in central Brazil. Plant Soil 373, 829–842 (2013).
Sheil, D., Ladd, B., Silva, L. C., Laffan, S. W. & Van Heist, M. How are soil carbon and tropical biodiversity related? Environ. Conserv. 43, 231–241 (2016).
Burnham, K. P. & Anderson, D. R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach (Springer-Verlag, New York, 2002).
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Satistical Computing, Vienna, 2017).
Grace, J. B. Structural Equation Models and Natural Systems (Cambridge Univ. Press, Cambridge, 2006).
Statistica (Data Analysis Software System), Version 10 (StatSoft, Tulsa, 2001).
Acknowledgements
We thank the Guyana Environmental Protection Agency and the Ministry of Amerindian Affairs for permission to work in the Rupununi. We thank the National Science Foundation (NSF; Grant BE/CNH 05 08094), the Gordon and Betty Moore Foundation and Stanford University for financial and administrative support. We thank the Iwokrama International Centre for Rainforest Conservation, the North Rupununi District Development Board, and the Deep South Toshaos’ Council for support in Guyana. K. Epps, D. Turner and E. Kurten collaborated with soil sampling and analyses. N. Butts and A. Cummings sampled tree diversity and carbon in trees. Additionally, A. R. Larrinaga, A. Baselga and C. Gómez provided useful suggestions and collaborated with GIS work. We thank H. Mooney, C. Field, E. Garnier, C. Violle, A. Milcu, S. Hattenschwiler, B. Shipley, C. Tucker, M. Grennié, A. M. Cortizas, B. Glaser, A. da Rocha, L. García de Jalón and the Guitián lab for comments on the manuscript. S. García created the illustration. We thank the Makushi, Wapishana and Wai-Wai technicians whose fieldwork and local knowledge made this research possible.
Author information
Authors and Affiliations
Contributions
J.M.V.F., M.S., K.M.S. and L.F.B.O. conceptualized this study. M.S. completed the statistical analyses. J.M.V.F., K.M.S., H.O., L.F.B.O., T.K.R. and other colleagues designed the field study and/or oversaw data collection. M.S. and K.M.S. drafted the manuscript with all authors participating in revisions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary tables and figures.
Rights and permissions
About this article
Cite this article
Sobral, M., Silvius, K.M., Overman, H. et al. Mammal diversity influences the carbon cycle through trophic interactions in the Amazon. Nat Ecol Evol 1, 1670–1676 (2017). https://doi.org/10.1038/s41559-017-0334-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-017-0334-0
This article is cited by
-
Meta-analysis reveals that vertebrates enhance plant litter decomposition at the global scale
Nature Ecology & Evolution (2024)
-
Trophic rewilding can expand natural climate solutions
Nature Climate Change (2023)
-
Big trees drive forest structure patterns across a lowland Amazon regrowth gradient
Scientific Reports (2021)
-
Impact of natural salt lick on the home range of Panthera tigris at the Royal Belum Rainforest, Malaysia
Scientific Reports (2021)
-
Protecting irrecoverable carbon in Earth’s ecosystems
Nature Climate Change (2020)