Abstract
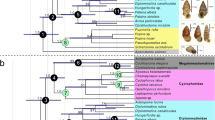
Ctenophora, comprising approximately 200 described species, is an important lineage for understanding metazoan evolution and is of great ecological and economic importance. Ctenophore diversity includes species with unique colloblasts used for prey capture, smooth and striated muscles, benthic and pelagic lifestyles, and locomotion with ciliated paddles or muscular propulsion. However, the ancestral states of traits are debated and relationships among many lineages are unresolved. Here, using 27 newly sequenced ctenophore transcriptomes, publicly available data and methods to control systematic error, we establish the placement of Ctenophora as the sister group to all other animals and refine the phylogenetic relationships within ctenophores. Molecular clock analyses suggest modern ctenophore diversity originated approximately 350 million years ago ± 88 million years, conflicting with previous hypotheses, which suggest it originated approximately 65 million years ago. We recover Euplokamis dunlapae—a species with striated muscles—as the sister lineage to other sampled ctenophores. Ancestral state reconstruction shows that the most recent common ancestor of extant ctenophores was pelagic, possessed tentacles, was bioluminescent and did not have separate sexes. Our results imply at least two transitions from a pelagic to benthic lifestyle within Ctenophora, suggesting that such transitions were more common in animal diversification than previously thought.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
18 October 2017
In the version of this Article originally published the location of Punta Arenas was incorrect and should have read 'Chile' in Figures 3–5 and in the Supplementary Information. This has been corrected in all versions of the Article.
References
Hyman, L. The Invertebrates Vol. 1 (McGraw-Hill, New York, 1940).
Mackie, G. O., Mills, C. E. & Singla, C. L. Structure and function of the prehensile tentialla of Euplokamis (Ctenophora, Cydippida). Zoomorphology 107, 319–337 (1988).
Moroz, L. L. et al. The ctenophore genome and the evolutionary origins of neural systems. Nature 510, 109–114 (2014).
Roohi, A. et al. Changes in biodiversity of phytoplanton, zooplankton, fishes and macrobenthos in the Southern Caspian Sea after the invasion of the ctenophore Mnemiopsis leidyi. Biol. Invasions 12, 2342–2361 (2010).
Ryan, J. F. et al. The genome of the ctenophore Mnemiopsis leidyi and its implications for cell type evolution. Science 342, 1242592 (2013).
Harbison, G. R. in The Origins and Relationships of Lower Invertebrates (eds Morris, S. C., George, J. D., Gibson, R. & Platt, H. M.) 78–100 (Oxford Univ. Press, Oxford, 1985).
Dunn, C. W., Leys, S. & Haddock, S. H. D. The hidden biology of sponges and ctenophores. Trends Ecol. Evol. 30, 282–291 (2015).
Whelan, N. V., Kocot, K. M. & Halanych, K. M. Employing phylogenomics to resolve the relationships among cnidarians, ctenophores, sponges, placozoans and bilaterians. Integr. Comp. Biol. 55, 1084–1095 (2015).
Dunn, C. W. et al. Broad phylogenomic sampling improves resolution of the animal tree of life. Nature 452, 745–749 (2008).
Hejnol, A. et al. Assessing the root of bilaterian animals with scalable phylogenomic models. Proc. Biol. Sci. 276, 4261–4270 (2009).
Whelan, N. V., Kocot, K. M., Moroz, L. L. & Halanych, K. M. Error, signal, and the placement of Ctenophora sister to all other animals. Proc. Natl. Acad. Sci. USA 112, 5773–2778 (2015).
Podar, M., Haddock, S. H. D., Sogin, M. L. & Harbison, G. R. A molecular phylogenetic framework for the phylum Ctenophora using 18S rRNA genes. Mol. Phylogen. Evol. 21, 218–230 (2001).
Simion, P., Bekkouche, N., Jager, M., Quéinnec, E. & Manuel, M. Exploring the potential of small RNA subunit and ITS sequences for resolving the phylogenetic relationships within the phylum Ctenophora. Zoology 118, 102–114 (2015).
Tang, F., Bengtson, S., Wang, Y., Wang, X.-l. & Yin, C.-Y. Eoandromeda and the origin of Ctenophora. Evol. Dev. 13, 408–414 (2011).
Morris, S. C. & Collins, D. H. Middle Cambiran ctenophores from Stephen Formation British Columbia, Canada. Phil. Trans. R. Soc. Lond. B 351, 279–308 (1996).
Chen, J.-Y. et al. Raman spectra of a Lower Cambrian ctenophore embryo from southwestern Shaanxi, China. Proc. Natl Acad. Sci USA 104, 6289–6292 (1997).
Mills, C. E. Revised classification of the genus Euplokamis Chun, 1880 (Ctenophora: Cydippida: Euplokamidae n. fam.) with a description of the new species Euplokamis dunlapae. Can. J. Zool. 65, 2661–2668 (1987).
Harbison, G. R. & Miller, R. L. Not all ctenophores are hermaphrodites. Studies on the sytematics, distribution, sexuality and development of two species of Ocyropsis. Mar. Biol. 90, 413–424 (1986).
Uyeno, D., Lasley, R. M., Moore, J. M. & Berumen, M. L. New records of Lobatolampea tetragona (Ctenophora: Lobata: Lobatolampeidae) from the Red Sea. Mar. Biodivers. Rec. 8, e33 (2015).
Pisani, D. et al. Genomic data do not support comb jellies as the sister group to all other animals. Proc. Natl Acad. Sci. USA 112, 15402–15407 (2015).
Simion, P. et al. A large and consistent phylogenomic dataset supports sponges as the sister group to all other animals. Curr. Biol. 27, 958–967 (2017).
Halanych, K. M., Whelan, N. V., Kocot, K. M., Kohn, A. B. & Moroz, L. L. Miscues misplace sponges. Proc. Natl Acad. Sci. USA 113, E946–E949 (2016).
Moroz, L. L. & Halanych, K. M. A sisterly dispute: methodological misconceptions. Nature 529, 286–287 (2016).
Whelan, N. V. & Halanych, K. M. Who let the CAT out of the bag? Accurately dealing with subtitutional heterogeneity in phylogenomics analyses. Syst. Biol. 66, 232–255 (2017).
Arcila, D. et al. Genome-wide interrogation advances resolution of recalcitrant groups in the tree of life. Nat. Ecol. Evol. 1, 0020 (2017).
Shen, X.-X., Hittinger, C. T. & Rokas, A. Contentious relationships in phylogenomic studies can be driven by a handful of genes. Nat. Ecol. Evol. 1, 0126 (2017).
Heath, T. A., Hedtke, S. M. & Hillis, D. M. Taxon sampling and the accuracy of phylogenetic analyses. J. Syst. Evol. 46, 239–257 (2008).
Hedtke, S. M., Townsend, T. M. & Hillis, D. M. Resolution of phylogenetic conflict in large data sets by increased taxon sampling. Syst. Biol. 55, 522–529 (2006).
Zwickl, D. J. & Hillis, D. M. Increased taxon sampling greatly reduces phylogenetic error. Syst. Biol. 51, 588–598 (2002).
Benton, M. J. & Twitchett, R. J. How to kill (almost all life): the end-Permian extinctoin event. Trends Ecol. Evol. 18, 358–365 (2003).
Lartillot, N. & Philippe, H. A Bayesian mixture model for across-site heterogeneities in the amino-acid replacement process. Mol. Biol. Evol. 21, 1095–1109 (2004).
Steinmetz, P. R. H. et al. Independent evolution of striated muscles in cnidarians and bilaterians. Nature 487, 231–234 (2012).
Martindale, M. Q. in Atlas of Marine Invertebrate Larvae (eds Young, C. M., Sewell, M. A. & Rice, M. E.) 109–122 (Academic Press, Cambridge, MA, 2002).
Rigby, S. & Milsom, C. Benthic origins of zooplankton: an environmentaly determined macroevolutionary effect. Geology 24, 52–54 (1996).
Haddock, S. H. D. & Case, J. F. Not all ctenophores are bioluminescent: Pleurobrachia. Biol. Bull. 189, 356–362 (1995).
Widder, E. A. Bioluminescence in the ocean: origins of biological, chemical, and ecological diversity. Science 328, 704–708 (2010).
Gershwin, L.-A., Zeidler, W. & Davie, P. J. F. Ctenophora of Australia. Mem. Queensl. Mus. 54, 1–45 (2010).
Haddock, S. H. D. Comparative feeding behaviour of planktonic ctenophores. Integr. Comp. Biol. 47, 847–853 (2007).
Moroz, L. L. Covergent evolution of neural systems in ctenophores. J. Exp. Biol. 218, 598–611 (2015).
Moroz, L. L. & Kohn, A. B. Independent origins of neurons and synapses: insights from ctenophores. Phil. Trans. R. Soc. Lond. B 371, 20150041 (2016).
Borowiec, M. L., Lee, E. K., Chiu, J. C. & Plachetzki, D. C. Extracting phylogenetic signal and accounting for bias in whole-genome data sets supports the Ctenophora as sister to remaining Metazoa. BMC Genomics 16, 987 (2015).
Cannon, J. T. et al. Xenacoelomorpha is the sister group to Nephrozoa. Nature 530, 89–93 (2016).
Chang, E. S. et al. Genomic insights into the evolutionary origin of Myxozoa within Cnidaria. Proc. Natl Acad. Sci. USA 112, 14912–14917 (2015).
Nabhan, A. R. & Sarkar, I. N. The impact of taxon sampling on phylogenetic inference: a review of two decades of controversy. Brief. Bioinformatics 13, 122–134 (2011).
Brown, T., Howe, C., Zhang, A., Pyrkosz, Q. & Brom, A. B. A reference-free algorithm for computational normalization of shotgun sequencing data. Preprint at https://arxiv.org/abs/1203.4802 (2012).
Haas, B. J. et al. De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat. Protoc. 8, 1494–1512 (2013).
Ebersberger, I., Strauss, S. & von Haeseler, A. HaMStR: profile hidden Markov model based search for orthologs in ESTs. BMC Evol. Biol. 9, 157 (2009).
Altschul, S. F. et al. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 25, 3389–3402 (1997).
Li, L., Stoeckert, C. J. & Roos, D. S. OrthoMCL: identification of ortholog groups for eukaryotic genomes. Genome Res. 13, 2178–2189 (2003).
Kocot, K. M. et al. Phylogenomics of Lophotrochozoa with consideration of systematic error. Syst. Biol. 66, 256–282 (2017).
Katoh, K. & Standley, D. M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Price, M. N., Dehal, P. S. & Arkin, A. P. FastTree 2—approximately maximum-likelihood trees for large alignments. PLoS ONE 5, e9490 (2010).
Kocot, K. M., Citarella, M. R., Moroz, L. L. & Halanych, K. M. PhyloTreePruner: a phylogenetic tree-based approach for selection of orthologous sequences for phylogenomics. Evol. Bioinform. 9, 429–435 (2013).
Finn, R. D., Clements, J. & Eddy, S. R. HMMER web server: interactive sequence similarity searching. Nucleic Acids Res. 39, W29–W37 (2011).
Stamatakis, A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313 (2014).
Struck, T. H. TreSpEx—detection of misleading signal in phylogenetic reconstructions based on tree information. Evol. Bioinform. 10, 51–67 (2014).
R Development Core Team. R: A Language and Environment for Statistical Computing http://www.R-project.org/ (R Foundation for Statistical Computing, Vienna, 2015).
Zhong, M. et al. Detecting the symplesiomorphy trap: a multigene phylogenetic analysis of terebelliform annelids. BMC Evol. Biol. 11, 369 (2011).
Kück, P. & Struck, T. H. BaCoCa—a heuristic software tool for the parallel assessment of sequence biases in hundreds of gene and taxon partitions. Mol. Phylogen. Evol. 70, 94–98 (2014).
Lartillot, N., Rodrigue, N., Stubbs, D. & Richer, J. PhyloBayes MPI: phylogenetic reconstruction with infinite mixtures of profiles in a parallel environment. Syst. Biol. 62, 611–615 (2013).
Nosenko, T. et al. Deep metazoan phylogeny: when different genes tell different stories. Mol. Phylogen. Evol. 67, 223–233 (2013).
Brown, J. M. & Lemmon, A. R. The importance of data partitioning and the utility of Bayes factors in Bayesian phylogenetics. Syst. Biol. 56, 643–655 (2007).
Lanfear, R., Frandsen, P. B., Wright, A. M., Senfeld, T. & Calcott, B. PartitionFinder 2: new methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol. Biol. Evol., 34, 772–773 (2016).
Lanfear, R., Calcott, B., Kainer, D., Mayer, C. & Stamatakis, A. Selecting optimal partitioning schemes for phylogenomic datasets. BMC Evol. Biol. 14, 82 (2014).
Jékely, G., Paps, J. & Nielsen, C. The phylogenetic position of ctenophores and the origin(s) of nervous systems. EvoDevo 6, 1 (2015).
Bouckaert, R. et al. BEAST 2: A software platform for Bayesian evolutionary analysis. PLoS Comput. Biol. 10, e1003537 (2014).
Rice, P., Longden, I. & Bleasby, A. EMBOSS: the European Molecular Biology Open Software Suite. Trends Genet. 16, 276–277 (2000).
Duchêne, S., Molak, M. & Ho, S. Y. W. ClockstaR: choosing the number of relaxed-clock models in molecular phylogenetic analysis. Bioinformatics 30, 1017–1019 (2014).
Drummond, A. J., Ho, S. Y. W., Phillips, M. J. & Rambaut, A. Relaxed phylogenetics and dating with confidence. PLoS Biol. 4, e88 (2006).
Dos Reis, M. et al. Uncertainty in the timing of origin of animals and the limits of precision in molecular timescales. Curr. Biol. 25, 29392950 (2015).
Brusca, R. C., Moore, W. & Shuster, S. M. in Invertebrates 3rd edn 1104 (Sinauer Associates, Sunderland, MA, 2016).
Eechkaut, I., Flammang, P., Bue, C. L. & Jangoux, M. Functional morphology of the tentacles and tentilla of Coeloplana bannworthi (Ctenophora, Platyctenida), and ectosymbiont of Diadema setosum (Echinodermata, Echinoida). Zoomorphology 117, 165–174 (1997).
Harbison, G. R. & Madin, L. P. in Synopsis and Classification of Living Organisms (ed. Parker, S. P.) 707–715 (McGraw-Hill, New York, 1982).
Horita, T. An undescribed lobate ctenophore, Lobatolampea tetragona gen. nov. & spec. nov., representing a new family, from Japan. Zool. Meded. 73, 457–464 (2000).
Matsumoto, G. I. & Harbison, G. R. In situ observations of foraging, feeding, and escape behavior in three orders of oceanic ctenophores: Lobata, Cestida, and Beroida. Mar. Biol. 117, 279–287 (1993).
Purcell, J. E., Sturdevant, M. V. & Galt, C. P. Response of Marine Ecosystems to Global Change: Ecological Impact of Appendicularians (eds Gorsky, G., Yongbluth, M. J. & Deibel, D.) 359–435 (GB Science Publishers-Editions Scientifiques, Paris, 2005).
Stretch, J. J. Observations on the abundance and feeding behavior of the cestid ctenophore. Velamen Parallelum. Bull. Mar. Sci. 32, 796–799 (1982).
Blomberg, S. P., Garland, T. Jr & Ives, A. R. Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57, 717–745 (2003).
Revell, L. J. Phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 3, 217–223 (2012).
Huelsenbeck, J. P., Nielsen, R. & Bollback, J. P. Stochastic mapping of morphological characters. Syst. Biol. 52, 131–158 (2003).
Nielsen, R. Mapping mutations on phylogenies. Syst. Biol. 51, 729–739 (2002).
Paradis, E., Claude, J. & Strimmer, K. APE: analysis of phylogenetics and evolution in R language. Bioinformatics 20, 289–290 (2004).
Harmon, L. J., Weir, J. T., Brock, C. D., Glor, R. E. & Challenger, W. GEIGER: investigating evolutionary radiations. Bioinformatics 24, 129–131 (2008).
Acknowledgements
This work was made possible in part by a grant of high-performance computing resources and technical support from the Alabama Supercomputer Authority and was supported by the United States National Aeronautics and Space Administration (grant NASA-NNX13AJ31G), the National Science Foundation (grants ANT-1043670, ANT-1043745, 1557923, 1548121 and 1645219) and the Ministry of Education of the Russian Federation (#14W03.31.0015). We thank the International SeaKeepers Society, captains and crew of RV Laurence M Gold, RV Capasetic, RV Penny Mae, RV Defiance, RV Basic Explorer, RV Harle of Fleet Miami and RV Miss Phebe II, as well as J. Jacoby for their help with the collection of ctenophores around the globe. A. Kohn helped with laboratory work and data curation. C. Mills helped with species identifications. The findings and conclusions in this paper are those of the authors and do not necessarily represent the views of the United States Fish and Wildlife Service. This is Molette Biology Laboratory contribution 70 and Auburn University Marine Biology Program contribution 166.
Author information
Authors and Affiliations
Contributions
N.V.W., K.M.K., L.L.M. and K.M.H. designed the study. K.M.K. collected and identified the Australian speices. L.L.M. and G.P. collected and identified all other ctenophores. P.W., K.M., T.P.M. and L.L.M. sequenced and assembled the ctenophore transcriptomes. N.V.W. and K.M.K. performed phylogenetic analyses and ancestral state reconstruction. N.V.W., K.M.K., L.L.M. and K.M.H. wrote the manuscript. All authors edited manuscript versions and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary Discussion, Tables, and Figures
Rights and permissions
About this article
Cite this article
Whelan, N.V., Kocot, K.M., Moroz, T.P. et al. Ctenophore relationships and their placement as the sister group to all other animals. Nat Ecol Evol 1, 1737–1746 (2017). https://doi.org/10.1038/s41559-017-0331-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-017-0331-3
This article is cited by
-
Neuromuscular organization of the benthic ctenophore, Vallicula multiformis
Zoological Letters (2024)
-
The ctenophore Mnemiopsis leidyi deploys a rapid injury response dating back to the last common animal ancestor
Communications Biology (2024)
-
The origin and evolution of Wnt signalling
Nature Reviews Genetics (2024)