Abstract
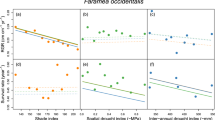
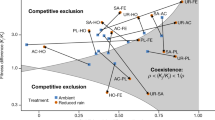
Occasional periods of drought are typical of most tropical forests, but climate change is increasing drought frequency and intensity in many areas across the globe, threatening the structure and function of these ecosystems. The effects of intermittent drought on tropical tree communities remain poorly understood and the potential impacts of intensified drought under future climatic conditions are even less well known. The response of forests to altered precipitation will be determined by the tolerances of different species to reduced water availability and the interactions among plants that alleviate or exacerbate the effects of drought. Here, we report the response of experimental monocultures and mixtures of tropical trees to simulated drought, which reveals a fundamental shift in the nature of interactions among species. Weaker competition for water in diverse communities allowed seedlings to maintain growth under drought while more intense competition among conspecifics inhibited growth under the same conditions. These results show that reduced competition for water among species in mixtures mediates community resistance to drought. The delayed onset of competition for water among species in more diverse neighbourhoods during drought has potential implications for the coexistence of species in tropical forests and the resilience of these systems to climate change.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chadwick, R., Good, P., Martin, G. & Rowell, D. P. Large rainfall changes consistently projected over substantial areas of tropical land. Nat. Clim. Change 6, 177–181 (2015).
Cai, W. et al. Increasing frequency of extreme El Niño events due to greenhouse warming. Nat. Clim. Change 5, 1–6 (2014).
O'Brien, M. J., Ong, R. & Reynolds, G. Intra-annual plasticity of growth mediates drought resilience over multiple years in tropical seedling communities. Glob. Change Biol. http://dx.doi.org/10.1111/gcb.13658 (2017).
Adler, P. B., HilleRisLambers, J., Kyriakidis, P. C., Guan, Q. & Levine, J. M. Climate variability has a stabilizing effect on the coexistence of prairie grasses. Proc. Natl Acad. Sci. USA 103, 12793–12798 (2006).
Bagchi, R. et al. Pathogens and insect herbivores drive rainforest plant diversity and composition. Nature 506, 85–88 (2014).
McDowell, N. G. et al. The interdependence of mechanisms underlying climate-driven vegetation mortality. Trends Ecol. Evol. 26, 523–532 (2011).
Sterck, F., Markesteijn, L., Schieving, F. & Poorter, L. Functional traits determine trade-offs and niches in a tropical forest community. Proc. Natl Acad. Sci. USA 108, 20627–20632 (2011).
Bell, T., Freckleton, R. P. & Lewis, O. T. Plant pathogens drive density-dependent seedling mortality in a tropical tree. Ecol. Lett. 9, 569–574 (2006).
Janzen, D. H. Herbivores and the number of tree species in tropical forests. Am. Nat. 104, 501–528 (1970).
Connell, J. H. Diversity in tropical rain forests and coral reefs. Science 199, 1302–1310 (1978).
Brienen, R. J. W. et al. Long-term decline of the Amazon carbon sink. Nature 519, 344–348 (2015).
Engelbrecht, B. M. J. et al. Drought sensitivity shapes species distribution patterns in tropical forests. Nature 447, 80–82 (2007).
Curran, L. M. et al. Impact of El Niño and logging on canopy tree recruitment in Borneo. Science 286, 2184–2188 (1999).
Doughty, C. E. et al. Allocation trade-offs dominate the response of tropical forest growth to seasonal and interannual drought. Ecology 95, 2192–2201 (2014).
Rowland, L. et al. Death from drought in tropical forests is triggered by hydraulics not carbon starvation. Nature 528, 119–122 (2015).
Isbell, F. et al. Biodiversity increases the resistance of ecosystem productivity to climate extremes. Nature 526, 574–577 (2015).
Morin, X., Fahse, L., de Mazancourt, C., Scherer-Lorenzen, M. & Bugmann, H. Temporal stability in forest productivity increases with tree diversity due to asynchrony in species dynamics. Ecol. Lett. 17, 1526–1535 (2014).
Sakschewski, B. et al. Resilience of Amazon forests emerges from plant trait diversity. Nat. Clim. Change 6, 1032–1036 (2016).
Lewis, S. L., Brando, P. M., Phillips, O. L., van der Heijden, G. M. F. & Nepstad, D. The 2010 Amazon drought. Science 331, 554 (2011).
Laurance, S. et al. Long-term variation in Amazon forest dynamics. J. Veg. Sci. 20, 323–333 (2009).
Levine, J. M. & HilleRisLambers, J. The importance of niches for the maintenance of species diversity. Nature 461, 254–257 (2009).
Darwin, C. On the Origins of Species by Means of Natural Selection (John Murray, London, 1859).
Ashton, P., Givnish, T. & Appanah, S. Staggered flowering in the Dipterocarpaceae: new insights into floral induction and the evolution of mast fruiting in the aseasonal tropics. Am. Nat. 132, 44–66 (1988).
Gibbons, J. M. & Newbery, D. M. Drought avoidance and the effect of local topography on trees in the understorey of Bornean lowland rain forest. Plant Ecol. 164, 1–18 (2003).
O’Brien, M. J., Burslem, D. F. R. P., Caduff, A., Tay, J. & Hector, A. Contrasting nonstructural carbohydrate dynamics of tropical tree seedlings under water deficit and variability. New Phytol. 205, 1083–1094 (2015).
Brassard, B. W. et al. Tree species diversity increases fine root productivity through increased soil volume filling. J. Ecol. 101, 210–219 (2013).
Wiley, E. & Helliker, B. A re-evaluation of carbon storage in trees lends greater support for carbon limitation to growth. New Phytol. 195, 285–289 (2012).
Körner, C. Paradigm shift in plant growth control. Curr. Opin. Plant Biol. 25, 107–114 (2015).
Muller, B. et al. Water deficits uncouple growth from photosynthesis, increase C content, and modify the relationships between C and growth in sink organs. J. Exp. Bot. 62, 1715–1729 (2011).
O’Brien, M. J., Leuzinger, S., Philipson, C. D., Tay, J. & Hector, A. Drought survival of tropical tree seedlings enhanced by non-structural carbohydrate levels. Nat. Clim. Change 4, 710–714 (2014).
Adams, H. et al. A multi-species synthesis of physiological mechanisms in drought-induced tree mortality. Nat. Ecol. Evol. 1, 1285–1291 (2017).
Have, P. et al. Predators have large effects on ecosystem properties by changing plant diversity, not plant biomass. Control 87, 1432–1437 (2006).
Güsewell, S. N. P ratios in terrestrial plants: variation and functional significance. New Phytol. 164, 243–266 (2004).
Adler, P. B., HilleRislambers, J. & Levine, J. M. A niche for neutrality. Ecol. Lett. 10, 95–104 (2007).
Sack, L. & Grubb, P. J. The combined impacts of deep shade and drought on the growth and biomass allocation of shade-tolerant woody seedlings. Oecologia 131, 175–185 (2002).
Nakagawa, M. et al. Impact of severe drought associated with the 1997–1998 El Niño in a tropical forest in Sarawak. J. Trop. Ecol. 16, 355–367 (2000).
Newbery, D. M. & Lingenfelder, M. Resistance of a lowland rain forest to increasing drought intensity in Sabah, Borneo. J. Trop. Ecol. 20, 613–624 (2004).
Martinez-Vilalta, J. & Lloret, F. Drought-induced vegetation shifts in terrestrial ecosystems: the key role of regeneration dynamics. Glob. Planet. Change 144, 94–108 (2016).
Granados, A., Brobie, J. F., Bernard, H. & O’Brien, M. J. Defaunation and habitat disturbance interact synergistically to alter seedling recruitment. Ecol. Appl. http://dx.doi.org/10.1002/eap.1592 (2017).
Bartlett, M. K. et al. Drought tolerance as a driver of tropical forest assembly: resolving spatial signatures for multiple processes. Ecology 97, 503–514 (2016).
Deka, R. et al. Use and accuracy of the filter-paper technique for measurement of soil matric potential. Eur. J. Soil Sci. 46, 233–238 (1995).
O’Brien, M. J., Philipson, C. D., Tay, J. & Hector, A. The influence of variable rainfall frequency on germination and early growth of shade-tolerant dipterocarp seedlings in Borneo. PLoS ONE 8, e70287 (2013).
Acknowledgements
This research was supported by the University Research Priority Programme on Global Change and Biodiversity at the University of Zurich, the Southeast Asian Rainforest Research Partnership and the NERC Human Modified Tropical Forests programme (BALI project). M.J.O.B. was funded by a Swiss National Science Foundation Postdoc Mobility Grant (P300PA_167758). We greatly appreciate the hard work of staff at the Malua Field Station, especially site manager P. Ulok and R. Murus. We thank F. Osman and V. Saidi for their tireless work on this experiment and L. Turnbull for comments on the manuscript. This Article is number 19 of the Sabah Biodiversity Experiment.
Author information
Authors and Affiliations
Contributions
M.J.O.B. came up with the concept, designed and carried out the experiment, analysed the data and wrote the manuscript. G.R. and R.O. contributed to the logistics and implementation of the experiment in Sabah. A.H. contributed to the design, analysis and writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary tables, figures and R code.
Rights and permissions
About this article
Cite this article
O’Brien, M.J., Reynolds, G., Ong, R. et al. Resistance of tropical seedlings to drought is mediated by neighbourhood diversity. Nat Ecol Evol 1, 1643–1648 (2017). https://doi.org/10.1038/s41559-017-0326-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-017-0326-0
This article is cited by
-
Climate-induced tree-mortality pulses are obscured by broad-scale and long-term greening
Nature Ecology & Evolution (2024)
-
Drought resistance enhanced by tree species diversity in global forests
Nature Geoscience (2022)
-
Drought-exposure history increases complementarity between plant species in response to a subsequent drought
Nature Communications (2022)
-
The genome of Shorea leprosula (Dipterocarpaceae) highlights the ecological relevance of drought in aseasonal tropical rainforests
Communications Biology (2021)
-
Conspecific negative density dependence in rainy season enhanced seedling diversity across habitats in a tropical forest
Oecologia (2020)