Abstract
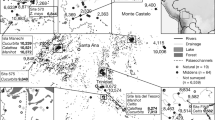
The development of agriculture is one of humankind’s most pivotal achievements. Questions about plant domestication and the origins of agriculture have engaged scholars for well over a century, with implications for understanding its legacy on global subsistence strategies, plant distribution, population health and the global methane budget. Rice is one of the most important crops to be domesticated globally, with both Asia (Oryza sativa L.) and Africa (Oryza glaberrima Steud.) discussed as primary centres of domestication. However, until now the pre-Columbian domestication of rice in the Americas has not been documented. Here we document the domestication of Oryza sp. wild rice by the mid-Holocene residents of the Monte Castelo shell mound starting at approximately 4,000 cal. yr bp, evidenced by increasingly larger rice husk phytoliths. Our data provide evidence for the domestication of wild rice in a region of the Amazon that was also probably the cradle of domestication of other major crops such as cassava (Manihot esculenta), peanut (Arachis hypogaea) and chilli pepper (Capsicum sp.). These results underline the role of wetlands as prime habitats for plant domestication worldwide.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
14 December 2018
In the HTML version of this Article originally published, Fig. 4 was a duplicate of Fig. 1. This has now been amended.
References
Lampe, K. Rice research: food for 4 billion people. GeoJournal 35, 253–261 (1995).
Sweeney, M. & McCouch, S. The complex history of the domestication of rice. Ann. Bot. 100, 951–957 (2007).
Callaway, E. Domestication: the birth of rice. Nature 514, S58–S59 (2014).
Linares, O. F. African rice (Oryza glaberrima): history and future potential. Proc. Natl Acad. Sci. USA 99, 16360–16365 (2002).
Kroeber, A. L. Cultural and Natural Areas of Native North America (Univ. California Press, Berkeley, 1939).
Eltis, D. et al. Agency and diaspora in Atlantic history: reassessing the African contribution to rice cultivation in the Americas. Am. Hist. Rev. 112, 1329–1358 (2007).
Schmitz, P. I., Rogge, J. H., Osorio Rosa, A. & Beber, M. V. Aterros indígenas no Pantanal do Mato Grosso do Sul Vol. 54 (Instituto anchietano de Pesquisas, São Leopoldo, 1998).
Hartt, C. F. & Agassiz, L. Thayer Expedition: Scientific Results of a Journey in Brazil: by Louis Agassiz and His Travelling Companions: Geology and Physical Geography of Brazil (Fields Osgood, Boston, 1870).
May, W. Die Reiskultur, insbesonders in Brasilien. Bot. Zeitung 50, 50–66 (1862).
Barman, R. J. The forgotten journey: Georg Heinrich Langsdorff and the Russian imperial scientific expedition to Brazil, 1821–1829. Terrae Incognitae 3, 67–96 (1971).
Winkler, H. Reis. (Monographien zur Landwirtschaft warmer Länder) Vol. 138 (Deutscher Auslandverlag W. Bangert, Hamburg, 1926).
De Azara, F. Descripción e Historia de Paraguay y del Río de la Plata (Biblioteca Virtual Miguel de Cervantes, Alicante, 2002).
Cardim, F. Tratados da Terra e Gente do Brasil (Editora Hedra, São Paulo, 2009).
de Acosta, J. Historia Natural y Moral de las Indias (Ramon Angles, Madrid, 1894).
Da Silva, C. A. A Dinâmica do Uso da Terra nos Locais Onde há Sitios Arqueológicos: o Caso da Comunidade cai n’Agua. Maniquiri-AM MA thesis (Univ. Federal do Amazonas, 2010).
Bortolotto, I. M., de Mello Amorozo, M. C., Neto, G. G., Oldeland, J. & Damasceno-Junior, G. A. Knowledge and use of wild edible plants in rural communities along Paraguay River, Pantanal, Brazil. J. Ethnobiol. Ethnomed. 11, 46 (2015).
Brochado, J. P. The Social Ecology of the Marajoara Culture. MA thesis (Univ. Illinois, 1980).
Clement, C. R. 1492 and the loss of Amazonian crop genetic resources. I. The relation between domestication and human population decline. Econ. Bot. 53, 188–202 (1999).
Piperno, D. R. & Pearsall, D. M. The Origins of Agriculture in the Lowland Neotropics (Academic Press, New York, 1998).
Soreng, R. J. et al. A worldwide phylogenetic classification of the Poaceae (Gramineae). J. System. Evol. 53, 117–137 (2015).
Judziewicz, E. J. et al. Catalogue of New World Grasses (Poaceae): I. Subfamilies Anomochlooideae, Bambusoideae, Ehrhartoideae, and Pharoideae (Smithsonian Institution, Washington DC, 2000).
Sanchez, P. L., Wing, R. A. & Brar, D. S. in Genetics and Genomics of Rice (eds Zhang, Q. & Wing, R. A.) 9–25 (Springer, New York, 2013).
Bertazzoni, E. C. & Damasceno-Júnior, G. A. Aspectos da biologia e fenologia de Oryza latifolia Desv.(Poaceae) no Pantanal sul-mato-grossense. Acta Bot. Bras. 2, 476–786 (2011).
Karasawa, M. M. et al. Mating system of Brazilian Oryza glumaepatula populations studied with microsatellite markers. Ann. Bot. 99, 245–253 (2007).
Santos, K. F., Silveira, R. D. D., Martin-Didonet, C. C. G. & Brondani, C. Storage protein profile and amino acid content in wild rice Oryza glumaepatula. Pesqui. Agropecu. Bras. 48, 66–72 (2013).
Vennum, T. Wild Rice and the Ojibway People (Minnesota Historical Society, St. Paul, 1988).
Oelke, E. A. in New Crops (eds Janick, J. & Simon, J. E.) 235–243 (Wiley, New York, 1993).
Gu, Y., Zhao, Z. & Pearsall, D. M. Phytolith morphology research on wild and domesticated rice species in East Asia. Quat. Int. 287, 141–148 (2013).
Pearsall, D. M. et al. Distinguishing rice (Oryza sativa Poaceae) from wild Oryza species through phytolith analysis: results of preliminary research. Econ. Bot. 49, 183–196 (1995).
Zhao, Z., Pearsall, D. M., Benfer, R. A. & Piperno, D. R. Distinguishing rice (Oryza sativa poaceae) from wild Oryza species through phytolith analysis, II Finalized method. Econ. Bot. 52, 134–145 (1998).
Harvey, E. L. & Fuller, D. Q. Investigating crop processing using phytolith analysis: the example of rice and millets. J. Archaeol. Sci. 32, 739–752 (2005).
Weisskopf, A. et al. Phytoliths and rice: from wet to dry and back again in the Neolithic Lower Yangtze. Antiquity 89, 1051–1063 (2015).
Purugganan, M. D. & Fuller, D. Q. The nature of selection during plant domestication. Nature 457, 843–848 (2009).
Piperno, D. R. Phytoliths: A Comprehensive Guide for Archaeologists and Paleoecologists (AltaMira Press, New York, 2006).
Piperno, D. R. & Stothert, K. E. Phytolith evidence for early Holocene Cucurbita domestication in southwest Ecuador. Science 299, 1054–1057 (2003).
Vrydaghs, L. et al. Differentiating the volcaniform phytoliths of bananas: Musa acuminata. Ethnobot. Res. Appl. 7, 239–246 (2009).
Lu, H. Y., Wu, N. Q. & Liu, B. Z. in The State-of-the-Art of Phytoliths in Soils and Plants (eds Pinilla, A., Trasseras, J. & Machado, M. J.) 159–174 (Monografias del Centro de Ciencias Medioambientales, Consejo Superior de Investigaciones Cientificas, Madrid, 1997).
Lombardo, U. et al. Early and middle holocene hunter-gatherer occupations in western Amazonia: the hidden shell middens. PLoS ONE 8, e72746 (2013).
Roosevelt, A. C. & Housley, R. A. Eighth millennium pottery from a prehistoric shell midden in the Brazilian Amazon. Science 254, 1621–1624 (1991).
Zimpel, C. A. & Pugliese, F. A. in Cerâmicas Arqueológicas da Amazônia: Rumo a Uma Nova Síntese (eds Barreto, C., Lima, H. P. & Betancourt, C. J.) 420–434 (Museu Paraense Emilio Goeldi, Belém do Pará, 2016).
Miller, E. T. Algumas culturas ceramistas, do noroeste do Pantanal do Guaporé à encosta e altiplano sudoeste do Chapadão dos Parecis. Origem, difusão/migração e adaptação – do noroeste da América do Sul ao Brasil. Revista Brasileira de Linguística Antropológica 5, 335–383 (2013).
Miller, E. T. Arqueologia nos empreendimentos hidrelétricos da Eletronorte: resultados preliminares (Eletronorte. Ministerio de Minas e Energia, Brasilia, 1992).
Zohary, D. Unconscious selection and the evolution of domesticated plants. Econ. Bot. 58, 5–10 (2004).
Iriarte, J. et al. Out of Amazonia: late-Holocene climate change and the Tupi–Guarani trans-continental expansion. Holocene 27, 967–975 (2016).
Austin, D. F. Fox-tail millets (Setaria: Poaceae)—abandoned food in two hemispheres. Econ. Bot. 60, 143–158 (2006).
Yarnell, R. A. Iva annua var. macrocarpa: extinct American cultigen? Am. Anthropol. 74, 335–341 (1972).
Kanapeckas, K. L. et al. Escape to ferality: the endoferal origin of weedy rice from crop rice through de-domestication. PLoS ONE 11, e0162676 (2016).
Fuller, D. Q. & Qin, L. Water management and labour in the origins and dispersal of Asian rice. World Archaeol. 41, 88–111 (2009).
Pohl, M. D. et al. Early agriculture in the Maya lowlands. Latin Am. Antiq. 7, 355–372 (1996).
Global Biodiversity Information Facility Version 1.0 (GBIS, Copenhagen, accessed 5 May 2017); https://www.gbif.org/.
Acknowledgements
The research was funded by the European Research Council project ‘Pre-Columbian Amazon-Scale Transformations’ (ERC-CoG 616179) to J.I. L.M.H. was funded by CAPES (Ministry of Education, Brazil) and Monte Castelo fieldwork was funded by grants from the Brazilian National Science Development Council (CNPq-307179/2013-3) and The National Geographic Society (W243-12) to E.G.N.
Author information
Authors and Affiliations
Contributions
L.H., J.I. and E.G.N. designed research; E.G.N., F.P., M.S. and C.A.Z. performed archaeological excavations at Monte Castelo; L.H. undertook phytolith analysis; B.S.W. carried out statistical analyses; E.V. provided Oryza reference collection samples for analysis; J.I. and L.H. led the writing of the paper with inputs from all other authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary notes, tables, figures, and references.
Rights and permissions
About this article
Cite this article
Hilbert, L., Neves, E.G., Pugliese, F. et al. Evidence for mid-Holocene rice domestication in the Americas. Nat Ecol Evol 1, 1693–1698 (2017). https://doi.org/10.1038/s41559-017-0322-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-017-0322-4
This article is cited by
-
A glimpse into shell mound builders’ diet during mid-to-late Holocene on Marajó island
Vegetation History and Archaeobotany (2023)
-
A New Methodological Approach to Detect Microcenters and Regions of Maize Genetic Diversity in Different Areas of Lowland South America
Economic Botany (2023)
-
Vernacular Names of Traditional Rice Varieties Reveal the Unique History of Maroons in Suriname and French Guiana
Economic Botany (2023)
-
Phytoliths in spikelets of selected Oryzoideae species: new findings from in situ observation
Archaeological and Anthropological Sciences (2022)
-
Palaeoenvironmental reconstruction through phytolith analysis in the Casa de Pedra shell mound archaeological site, São Francisco do Sul, Santa Catarina, Brazil
Vegetation History and Archaeobotany (2022)