Abstract
Climate change-driven increases in drought frequency and severity could compromise forest ecosystems and the terrestrial carbon sink1,2,3. While the impacts of single droughts on forests have been widely studied4,5,6, understanding whether forests acclimate to or become more vulnerable to sequential droughts remains largely unknown and is crucial for predicting future forest health. We combine cross-biome datasets of tree growth, tree mortality and ecosystem water content to quantify the effects of multiple droughts at a range of scales from individual trees to the globe from 1900 to 2018. We find that subsequent droughts generally have a more deleterious impact than initial droughts, but this effect differs enormously by clade and ecosystem, with gymnosperms and conifer-dominated ecosystems more often exhibiting increased vulnerability to multiple droughts. The differential impacts of multiple droughts across clades and biomes indicate that drought frequency changes may have fundamentally different ecological and carbon-cycle consequences across ecosystems.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All datasets are publicly available. The International Tree-Ring Data Bank is available from the National Oceanic and Atmospheric Administration (https://www.ncdc.noaa.gov/data-access/paleoclimatology-data/datasets/tree-ring); the US Forest Inventory and Analysis plot data are available from the US Department of Agriculture (https://www.fia.fs.fed.us/); and the vegetation optical depth data are available from the University of Montana (https://www.ntsg.umt.edu/project/default.php).
Code availability
All analysis was done in the open-source software R with the packages that are documented and cited in the Methods section of the paper. Code will be made available on request.
References
Ciais, P. et al. Europe-wide reduction in primary productivity caused by the heat and drought in 2003. Nature 437, 529–533 (2005).
Reichstein, M. et al. Climate extremes and the carbon cycle. Nature 500, 287–295 (2013).
IPCC Special Report on Managing the Risks of Extreme Events and Disasters to Advance Climate Change Adaptation (eds Field, C. B. et al.) (Cambridge Univ. Press, 2012).
Schwalm, C. R. et al. Reduction in carbon uptake during turn of the century drought in western North America. Nat. Geosci. 5, 551–556 (2012).
Phillips, O. L. et al. Drought sensitivity of the Amazon rainforest. Science 323, 1344–1347 (2009).
Allen, C. D., Breshears, D. D. & McDowell, N. G. On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere 6, 129 (2015).
Dai, A. Drought under global warming: a review. Wiley Interdiscip. Rev. Clim. Change 2, 45–65 (2011).
Cook, B. I., Smerdon, J. E., Seager, R. & Coats, S. Global warming and 21st century drying. Clim. Dyn. 43, 2607–2627 (2014).
Cox, P. M., Betts, R. A., Jones, C. D., Spall, S. A. & Totterdell, I. J. Acceleration of global warming due to carbon-cycle feedbacks in a coupled climate model. Nature 408, 184–187 (2000).
Friedlingstein, P. et al. Uncertainties in CMIP5 climate projections due to carbon cycle feedbacks. J. Clim. 27, 511–526 (2014).
Zscheischler, J. et al. A few extreme events dominate global interannual variability in gross primary production. Environ. Res. Lett. 9, 035001 (2014).
Miao, S., Zou, C. B. & Breshears, D. D. Vegetation responses to extreme hydrological events: sequence matters. Am. Nat. 173, 113–118 (2008).
Anderegg, W. R. L. et al. Pervasive drought legacies in forest ecosystems and their implications for carbon cycle models. Science 349, 528–532 (2015).
Schwalm, C. R. et al. Global patterns of drought recovery. Nature 548, 202–205 (2017).
Holling, C. S. Resilience and stability of ecological systems. Annu. Rev. Ecol. Syst. 4, 1–23 (1973).
Gunderson, L. H. et al. Ecological resilience—in theory and application. Annu. Rev. Ecol. Syst. 31, 425–439 (2000).
Ingrisch, J. & Bahn, M. Towards a comparable quantification of resilience. Trends Ecol. Evol. 33, 251–259 (2018).
Bartlett, M. K. et al. Global analysis of plasticity in turgor loss point, a key drought tolerance trait. Ecol. Lett. 17, 1580–1590 (2014).
Martínez‐Vilalta, J. et al. Hydraulic adjustment of Scots pine across Europe. New Phytol. 184, 353–364 (2009).
Hacke, U. G., Stiller, V., Sperry, J. S., Pittermann, J. & McCulloh, K. A. Cavitation fatigue. Embolism and refilling cycles can weaken the cavitation resistance of xylem. Plant Physiol. 125, 779–786 (2001).
Sala, A., Woodruff, D. R. & Meinzer, F. C. Carbon dynamics in trees: feast or famine? Tree Physiol. 32, 764–775 (2012).
Schymanski, S. J., Roderick, M. L., Sivapalan, M., Hutley, L. B. & Beringer, J. A canopy-scale test of the optimal water-use hypothesis. Plant Cell Environ. 31, 97–111 (2008).
Zhang, T., Niinemets, Ü., Sheffield, J. & Lichstein, J. W. Shifts in tree functional composition amplify the response of forest biomass to climate. Nature 556, 99–102 (2018).
Anderegg, W. R. et al. Hydraulic diversity of forests regulates ecosystem resilience during drought. Nature 561, 538–541 (2018).
Royer, P. D. et al. Extreme climatic event-triggered overstorey vegetation loss increases understorey solar input regionally: primary and secondary ecological implications. J. Ecol. 99, 714–723 (2011).
Raffa, K. F. et al. Cross-scale drivers of natural disturbances prone to anthropogenic amplification: the dynamics of bark beetle eruptions. BioScience 58, 501–517 (2008).
Anderegg, W. R., Trugman, A. T., Bowling, D. R., Salvucci, G. & Tuttle, S. E. Plant functional traits and climate influence drought intensification and land–atmosphere feedbacks. Proc. Natl Acad. Sci. USA 116, 14071–14076 (2019).
Cailleret, M. et al. A synthesis of radial growth patterns preceding tree mortality. Glob. Change Biol. 23, 1675–1690 (2017).
Camarero, J. J., Gazol, A., Sangüesa-Barreda, G., Oliva, J. & Vicente-Serrano, S. M. To die or not to die: early warnings of tree dieback in response to a severe drought. J. Ecol. 103, 44–57 (2015).
Jump, A. S. et al. Structural overshoot of tree growth with climate variability and the global spectrum of drought-induced forest dieback. Glob. Change Biol. 23, 3742–3757 (2017).
Saatchi, S. et al. Persistent effects of a severe drought on Amazonian forest canopy. Proc. Natl Acad. Sci. USA 110, 565–570 (2013).
Konings, A. G., Williams, A. P. & Gentine, P. Sensitivity of grassland productivity to aridity controlled by stomatal and xylem regulation. Nat. Geosci. 10, 284–288 (2017).
Carnicer, J. et al. Widespread crown condition decline, food web disruption, and amplified tree mortality with increased climate change-type drought. Proc. Natl Acad. Sci. USA 108, 1474–1478 (2011).
Brienen, R. J. W. et al. Long-term decline of the Amazon carbon sink. Nature 519, 344–348 (2015).
Lenton, T. M. et al. Tipping elements in the Earth’s climate system. Proc. Natl Acad. Sci. USA 105, 1786–1793 (2008).
Duffy, P. B., Brando, P., Asner, G. P. & Field, C. B. Projections of future meteorological drought and wet periods in the Amazon. Proc. Natl Acad. Sci. USA 112, 13172–13177 (2015).
Johnson, D. M., McCulloh, K. A., Woodruff, D. R. & Meinzer, F. C. Hydraulic safety margins and embolism reversal in stems and leaves: why are conifers and angiosperms so different? Plant Sci. 196, 48–53 (2012).
Morris, H. et al. A global analysis of parenchyma tissue fractions in secondary xylem of seed plants. New Phytol. 209, 1553–1565 (2016).
DeSoto, L. et al. Low growth resilience to drought is related to future mortality risk in trees. Nat. Commun. 11, 1–9 (2020).
Fisher, R. A. et al. Vegetation demographics in Earth system models: a review of progress and priorities. Glob. Change Biol. 24, 35–54 (2018).
Kennedy, D. et al. Implementing plant hydraulics in the Community Land Model, version 5. J. Adv. Model. Earth Syst. 11, 485–513 (2019).
Trugman, A. T. et al. Tree carbon allocation explains forest drought-kill and recovery patterns. Ecol. Lett. 21, 1552–1560 (2018).
Trugman, A. T. et al. Climate and plant trait strategies determine tree carbon allocation to leaves and mediate future forest productivity. Glob. Change Biol. 25, 3395–3405 (2019).
Scheiter, S., Langan, L. & Higgins, S. I. Next-generation dynamic global vegetation models: learning from community ecology. New Phytol. 198, 957–969 (2013).
Vicente-Serrano, S. M., Beguería, S., López-Moreno, J. I., Angulo, M. & El Kenawy, A. A new global 0.5 gridded dataset (1901–2006) of a multiscalar drought index: comparison with current drought index datasets based on the Palmer Drought Severity Index. J. Hydrometeorol. 11, 1033–1043 (2010).
Beguería, S., Vicente-Serrano, S. M. & Angulo-Martínez, M. A multiscalar global drought dataset: the SPEIbase: a new gridded product for the analysis of drought variability and impacts. Bull. Am. Meteorol. Soc. 91, 1351–1356 (2010).
Beguería, S., Vicente-Serrano, S. M., Reig, F. & Latorre, B. Standardized Precipitation Evapotranspiration Index (SPEI) revisited: parameter fitting, evapotranspiration models, tools, datasets and drought monitoring. Int. J. Climatol. 34, 3001–3023 (2014).
Vicente-Serrano, S. M. et al. Response of vegetation to drought time-scales across global land biomes. Proc. Natl Acad. Sci. USA 110, 52–57 (2013).
Gazol, A., Camarero, J. J., Anderegg, W. R. L. & Vicente-Serrano, S. M. Impacts of droughts on the growth resilience of Northern Hemisphere forests. Glob. Ecol. Biogeogr. 26, 166–176 (2017).
Klesse, S. et al. Sampling bias overestimates climate change impacts on forest growth in the southwestern United States. Nat. Commun. 9, 5336 (2018).
Bechtold, W. A. & Patterson, P. L. The Enhanced Forest Inventory and Analysis Program—National Sampling Design and Estimation Procedures General Technical Report SRS-80 (USDA, 2005).
Bechtold, W. & Scott, C. T. in The Enhanced Forest Inventory and Analysis Program—National Sampling Design and Estimation Procedures General Technical Report SRS-80 (eds Bechtold, W. A. & Patterson, P. L.) 37–52 (USDA, 2005).
Woudenberg, S. W. et al. The Forest Inventory and Analysis Database: Database Description and Users Manual Version 4.0 for Phase 2 General Technical Report RMRS-GTR-245 (USDA, 2010).
Jacobi, W. R., Kearns, H. S. J. & Johnson, D. W. Persistence of pinyon pine snags and logs in southwestern Colorado. West. J. Appl. For. 20, 247–252 (2005).
Shaw, J. D. et al. Arizona’s Forest Resources, 2001–2014 Resource Bulletin RMRS-RB-25 (USDA, 2018).
Shaw, J. D., Steed, B. E. & DeBlander, L. T. Forest inventory and analysis (FIA) annual inventory answers the question: what is happening to pinyon-juniper woodlands? J. For. 103, 280–285 (2005).
Breshears, D. D. et al. Regional vegetation die-off in response to global-change-type drought. Proc. Natl Acad. Sci. USA 102, 15144–15148 (2005).
Williams, A. P. et al. Forest responses to increasing aridity and warmth in the southwestern United States. Proc. Natl Acad. Sci. USA 107, 21289–21294 (2010).
Anderegg, W. R. et al. Tree mortality from drought, insects, and their interactions in a changing climate. New Phytol. 208, 674–683 (2015).
Jackson, T. J. & Schmugge, T. J. Vegetation effects on the microwave emission of soils. Remote Sens. Environ. 36, 203–212 (1991).
Tian, F. et al. Remote sensing of vegetation dynamics in drylands: evaluating vegetation optical depth (VOD) using AVHRR NDVI and in situ green biomass data over West African Sahel. Remote Sens. Environ. 177, 265–276 (2016).
Liu, Y. Y. et al. Recent reversal in loss of global terrestrial biomass. Nat. Clim. Change 5, 470–474 (2015).
Momen, M. et al. Interacting effects of leaf water potential and biomass on vegetation optical depth. J. Geophys. Res. Biogeosci. 122, 3031–3046 (2017).
Konings, A. G. & Gentine, P. Global variations in ecosystem-scale isohydricity. Glob. Change Biol. 23, 891–905 (2017).
Van de Griend, A. A. & Wigneron, J.-P. The b-factor as a function of frequency and canopy type at H-polarization. IEEE Trans. Geosci. Remote Sens. 42, 786–794 (2004).
Konings, A. G., Rao, K. & Steele-Dunne, S. C. Macro to micro: microwave remote sensing of plant water content for physiology and ecology. New Phytol. https://doi.org/10.1111/nph.15808 (2019).
Du, J. et al. A global satellite environmental data record derived from AMSR-E and AMSR2 microwave earth observations. Earth Syst. Sci. Data Discuss. https://doi.org/10.5194/essd-2017-27 (2017).
Du, J., Kimball, J. S., Jones, L. A. & Member, S. Passive microwave remote sensing of soil moisture based on dynamic vegetation scattering properties for AMSR-E. IEEE Trans. Geosci. Remote Sens. 54, 597–608 (2015).
Du, J. et al. A global satellite environmental data record derived from AMSR-E and AMSR2 microwave Earth observations. Earth Syst. Sci. Data 9, 791–808 (2017).
Jones, L. A. et al. Satellite microwave remote sensing of daily land surface air temperature minima and maxima from AMSR-E. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 3, 111–123 (2010).
Olson, D. M. et al. Terrestrial Ecoregions of the World: A New Map of Life on Earth: a new global map of terrestrial ecoregions provides an innovative tool for conserving biodiversity. BioScience 51, 933–938 (2001).
Zar, J. H. in Biostatistical Analysis 1st edn, 185–205 (Prentice-Hall International, 1984).
Fox, J. et al. Package ‘car’ (R Foundation for Staistical Computing, 2012).
Fox, J., Friendly, M. & Weisberg, S. Hypothesis tests for multivariate linear models using the car package. R J. 5, 39–52 (2013).
Dormann, C. F. et al. Methods to account for spatial autocorrelation in the analysis of species distributional data: a review. Ecography 30, 609–628 (2007).
Pinheiro, J. et al. nlme: linear and nonlinear mixed effects models. R package v.3.1-117 (R Foundation for Statistical Computing, 2014).
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2012).
Acknowledgements
All correspondence and requests for materials should be addressed to W. Anderegg. W.R.L.A. acknowledges funding from the David and Lucille Packard Foundation, NSF Grants 1714972 and 1802880, and the USDA National Institute of Food and Agriculture, Agricultural and Food Research Initiative Competitive Programme, Ecosystem Services and Agro-ecosystem Management, grant no. 2018–67019–27850. A.T.T. acknowledges funding from the USDA National Institute of Food and Agriculture, Agricultural and Food Research Initiative Competitive Programme grant no. 2018-67012-31496 and the University of California Laboratory Fees Research Program Award No. LFR-20-652467. A.G.K. acknowledges funding from the NASA Carbon Cycle Science Program, and through the New Investigator Program (award 80NSSC18K0715), and from NOAA grant NA17OAR4310127.
Author information
Authors and Affiliations
Contributions
W.R.L.A., A.T.T. and G.B. designed the study. A.G.K. and J.S. provided key datasets. W.R.L.A. and A.T.T. analysed the data. W.R.L.A. wrote the first draft of the paper, and all authors contributed to writing and revising the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Climate Change thanks Adriaan J. Teuling, Alistair Jump and Tao Zhang for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
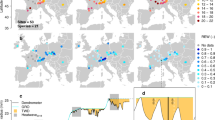
Extended Data Fig. 1 Impacts of a subsequent drought are more deleterious than an initial drought for trees.
Growth declines (Δring width index; a) from 1,208 sites in the International Tree-Ring Data Bank to an initial drought (Initial, light red) and subsequent drought (Subseq, dark red), categorized by drought severity of both droughts via the Standardized Precipitation Evapotranspiration Index (SPEI) thresholds. Identical data as Fig. 1a but shown as a violin plot. Blue dots are the mean. Numbers in italics are the number of chronologies in each bin. b, Growth declines differences from the International Tree-Ring Data Bank by clade where negative numbers indicate a more deleterious effect of the subsequent drought (left-to-right Nchronologies = 106, 410, 40, 174, 56, 291, 34, 257). Stars indicate statistically significant differences (*p < 0.05, **p < 0.01, ***p < 0.001).
Extended Data Fig. 2 Drought severity was typically similar between initial and subsequent droughts.
Drought severity differences for tree-ring (a), forest inventory (b), and vegetation optical depth (c) data. Initial drought (ID, light red) and subsequent drought (SD, dark red) drought severity of both droughts via the Standardized Precipitation Evapotranspiration Index (SPEI) thresholds. For sample sizes, see Figs. 1a, b, 3a. Error bars indicate ± 1 S.D. Stars indicate statistically significant differences (*p < 0.05, **p < 0.01).
Extended Data Fig. 3 Growth decline results were consistent across multiple tree-ring analysis methods.
Ring width difference during drought compared to average growth for an initial drought (ID, light red) and subsequent drought (SD, dark red) at SPEI < −2 drought severity threshold for the standardized (that is standard detrended “.crn” file presented in ITRDB) tree-ring chronology (“Std”; as in Fig. 1), a consistent spline de-trending method applied to all chronologies (“Detrd”), and a detrended and “prewhitened” (that is autoregressive model removed) (“PreWh”). Error bars indicate ± 1 S.E.M. Stars indicate statistically significant differences (**p < 0.01).
Extended Data Fig. 4 Impacts of a subsequent drought are more deleterious than an initial drought for trees.
Growth declines (Δring width index) from 1,208 sites in the International Tree-Ring Data Bank to an initial drought (Initial, light red) and subsequent drought (Subseq, dark red), categorized by drought severity of both droughts via the Standardized Precipitation Evapotranspiration Index (SPEI) thresholds. For multi-year droughts, the first year of the drought was analyzed. Error bars indicate ± 1 standard error. Stars indicate statistically significant differences (*p < 0.05, **p < 0.01).
Extended Data Fig. 5 All results are robust to accounting for spatial autocorrelation.
a, Difference in tree ring width index between a subsequent drought (SD) and an initial drought (SD) at two drought severity thresholds, where negative numbers indicate a subsequent drought is more harmful. b, Difference in mortality in forest inventory plots between a subsequent drought (SD) and an initial drought (SD) at two drought severity thresholds, where positive numbers indicate a subsequent drought is more harmful. c, Difference in vegetation optical depth (VOD) anomaly between a subsequent drought (SD) and an initial drought (SD) at two drought severity thresholds, where negative numbers indicate a subsequent drought is more harmful. Error bars indicate ± 1 S.E.M. Stars indicate statistically significant differences from zero (*p < 0.05, **p < 0.01).
Extended Data Fig. 6 Ecosystem impacts of multiple droughts are robust to accounting for drought severity differences.
Vegetation optical depth (VOD) anomaly (a) in response to an initial drought (Initial, light red) and subsequent drought (Subseq, dark red), categorized by drought severity of both droughts via the Standardized Precipitation Evapotranspiration Index (SPEI) thresholds. Panels (b) and (c) show the predicted minus observed VOD anomalies after construing a grid cell specific linear (b) and quadratic (c) regression between SPEI and VOD anomaly. Error bars indicate ± 1 S.E.M. Stars indicate statistically significant differences (***p < 0.001).
Extended Data Fig. 7 Ecosystem impacts of a subsequent drought are more deleterious than an initial drought, accounting for potential drought legacy effects.
Vegetation optical depth (VOD) anomaly in response to an initial drought (Initial, light red) and subsequent drought (Subseq, dark red), categorized by drought severity of both droughts via the Standardized Precipitation Evapotranspiration Index (SPEI) thresholds. All analyses used a 3+ year gap between initial and subsequent droughts. For biome definitions, see Fig. 3c. Error bars indicate + /− 1 standard error. Stars indicate statistically significant differences following other figures.
Extended Data Fig. 8 Analysis included broad geographic coverage of tree growth and mortality.
Geographical coverage of (a) the International Tree-Ring Data Bank (ITRDB) tree-ring chronologies, and (b) U.S. Forest Inventory and Analysis (FIA) long-term inventory plots included in this analysis.
Extended Data Fig. 9 Similar qualitative mortality patterns are observed in terms of mortality differences between initial and subsequent droughts.
This figure presents these patterns when excluding FIA mortality data from Intermountain West states (Nplots: 2848, 1270, 2559, 920; Ngrid-cells: 99, 43, 93, 54 left-to-right bar pairs). Figure legend otherwise the same as in Fig. 1b.
Extended Data Fig. 10 The deleterious impacts of subsequent droughts are robust to accounting for temporal trends.
Vegetation optical depth (VOD) responses to initial (Initial) and subsequent (Subseq) droughts are robust to accounting for potential trends in VOD. Left of the dashed line indicates the approach of selecting for grid cells without significant trends, while right of the dashed lines indicate detrending individual grid cells at the outset. Otherwise, legend is identical to Fig. 3a. Stars indicate statistically significant differences (***p < 0.001).
Rights and permissions
About this article
Cite this article
Anderegg, W.R.L., Trugman, A.T., Badgley, G. et al. Divergent forest sensitivity to repeated extreme droughts. Nat. Clim. Chang. 10, 1091–1095 (2020). https://doi.org/10.1038/s41558-020-00919-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41558-020-00919-1
This article is cited by
-
Responses of stem growth and canopy greenness of temperate conifers to dry spells
International Journal of Biometeorology (2024)
-
Species-specific, size-dependent, and environmentally modulated growth resilience to drought in conifer forests on the Eastern Tibetan Plateau
European Journal of Forest Research (2024)
-
Hydroclimatic extremes contribute to asymmetric trends in ecosystem productivity loss
Communications Earth & Environment (2023)
-
Evidence and attribution of the enhanced land carbon sink
Nature Reviews Earth & Environment (2023)
-
Approaching a thermal tipping point in the Eurasian boreal forest at its southern margin
Communications Earth & Environment (2023)