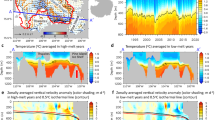
Abstract
Ocean acidification, induced by the increase in anthropogenic CO2 emissions, has a profound impact on marine organisms and biogeochemical processes1. The response of marine microbial activities to ocean acidification might play a crucial role in the future evolution of air–sea fluxes of biogenic gases such as nitrous oxide (N2O), a strong GHG and the dominant stratospheric ozone-depleting substance2. Here, we examine the response of N2O production from nitrification to acidification in a series of incubation experiments conducted in subtropical and subarctic western North Pacific. The experiments show that when pH was reduced, the N2O production rate during nitrification measured at subarctic stations increased significantly while nitrification rates remained stable or decreased. Contrary to previous findings, these results suggest that the effect of ocean acidification on N2O production during nitrification and nitrification rates are probably uncoupled. Collectively, these results suggest that if seawater pH continues to decline at the same rate, ocean acidification could increase marine N2O production during nitrification in the subarctic North Pacific by 185 to 491% by the end of the century.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the article and its Supplementary Information and at at http://www.godac.jamstec.go.jp/darwin/e.
References
Beman, J. M. et al. Global declines in oceanic nitrification rates as a consequence of ocean acidification. Proc. Natl Acad. Sci. USA 108, 208–213 (2011).
Freing, A., Wallace, D. W. R. & Bange, H. W. Global oceanic production of nitrous oxide. Phil. Trans. R. Soc. B 367, 1245–1255 (2012).
Caldeira, K. & Wickett, M. E. Oceanography: anthropogenic carbon and ocean pH. Nature 425, 365–365 (2003).
Orr, J. C. et al. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 437, 681–686 (2005).
Kump, L. R., Bralower, T. J. & Ridgwell, A. Ocean acidification in deep time. Oceanography 22, 94–107 (2009).
Pearson, P. N. & Palmer, M. R. Atmospheric carbon dioxide concentrations over the past 60 million years. Nature 406, 695–699 (2000).
Codispoti, L. A. Interesting times for marine N2O. Science 327, 1339–1340 (2010).
Breider, F., Yoshikawa, C., Abe, H., Toyoda, S. & Yoshida, N. Origin and fluxes of nitrous oxide along a latitudinal transect in western North Pacific: controls and regional significance. Glob. Biogeochem. Cycles 29, 1014–1027 (2015).
IPCC Climate Change 2007: The Physical Science Basis (eds Solomon, S. et al.) (Cambridge Univ. Press, 2007).
Frame, C. H., Lau, E., Nolan, E. J., Goepfert, T. J. & Lehmann, M. F. Acidification enhances hybrid N2O production associated with aquatic ammonia-oxidizing microorganisms. Front. Microbiol. 7, 2104 (2017).
Rees, A. P., Brown, I. J., Jayakumar, A. & Ward, B. B. The inhibition of N2O production by ocean acidification in cold temperate and polar waters. Deep Sea Res. Pt II 127, 93–101 (2016).
Honda, M. C. et al. Comparison of carbon cycle between the western Pacific subarctic and subtropical time-series stations: highlights of the K2S1 project. J. Oceanogr. 73, 647–667 (2017).
Cedervall, P., Hooper, A. B. & Wilmot, C. M. Structural studies of hydroxylamine oxidoreductase reveal a unique heme cofactor and a previously unidentified interaction partner. Biochemistry 52, 6211–6218 (2013).
Fernández, M. L., Estrin, D. A. & Bari, S. E. Theoretical insight into the hydroxylamine oxidoreductase mechanism. J. Inorg. Biochem. 102, 1523–1530 (2008).
Shiro, Y. Structure and function of bacterial nitric oxide reductases: nitric oxide reductase, anaerobic enzymes. Biochim. Biophys. Acta 1817, 1907–1913 (2012).
Hino, T. et al. Structural basis of biological N2O generation by bacterial nitric oxide reductase. Science 330, 1666–1670 (2010).
Yang, H., Gandhi, H., Ostrom, N. & Hegg, E. L. Isotopic fractionation by a fungal P450 nitric oxide reductase during the production of N2O. Environ. Sci. Technol. 48, 10707–10715 (2014).
Lehnert, N., Praneeth, V. K. K. & Paulat, F. Electronic structure of iron(II)–porphyrin nitroxyl complexes: molecular mechanism of fungal nitric oxide reductase (P450nor). J. Comput. Chem. 27, 1338–1351 (2006).
Hatzenpichler, R. Diversity, physiology, and niche differentiation of ammonia-oxidizing Archaea. Appl. Environ. Microbiol. 78, 7501–7510 (2012).
Fehling, C. & Friedrichs, G. Dimerization of HNO in aqueous solution: an interplay of solvation effects, fast acid–base equilibria, and intramolecular hydrogen bonding? J. Am. Chem. Soc. 133, 17912–17922 (2011).
Toyoda, S., Yoshida, N. & Koba, K. Isotopocule analysis of biologically produced nitrous oxide in various environments. Mass Spectrom. Rev. 36, 135–160 (2017).
Zhang, Y. Computational investigations of HNO in biology. J. Inorg. Biochem. 118, 191–200 (2013).
Caranto, J. D. & Lancaster, K. M. Nitric oxide is an obligate bacterial nitrification intermediate produced by hydroxylamine oxidoreductase. Proc. Natl Acad. Sci. USA 114, 8217–8222 (2017).
Bartberger, M. D., Fukuto, J. M. & Houk, K. N. On the acidity and reactivity of HNO in aqueous solution and biological systems. Proc. Natl Acad. Sci. USA 98, 2194–2198 (2001).
Krulwich, T. A., Sachs, G. & Padan, E. Molecular aspects of bacterial pH sensing and homeostasis. Nat. Rev. Microbiol. 9, 330–343 (2011).
Slonczewski, J. L., Fujisawa, M., Dopson, M. & Krulwich, T. A. in Advances in Microbial Physiology Vol. 55 (ed. Poole, R. K.) 1–79 (Academic, 2009).
Wakita, M. et al. Ocean acidification from 1997 to 2011 in the subarctic western North Pacific Ocean. Biogeosciences 10, 7817–7827 (2013).
Wilks, J. C. & Slonczewski, J. L. pH of the cytoplasm and periplasm of Escherichia coli: rapid measurement by green fluorescent protein fluorimetry. J. Bacteriol. 189, 5601–5607 (2007).
Ishii, M. et al. Ocean acidification off the south coast of Japan: a result from time series observations of CO2 parameters from 1994 to 2008. J. Geophys. Res. Oceans 116, C06022 (2011).
Schlitzer, R. Ocean Data View v.5.1.7 (Alfred Wegener Institute, 2019); http://odv.awi.de
Riebesell, U., Fabry, V. J., Hansson, L. & Gattuso, J.-P. (eds) Guide to Best Practices for Ocean Acidification Research and Data Reporting (European Commission, 2010).
Gattuso, J.-P. & Lavigne, H. Technical Note: approaches and software tools to investigate the impact of ocean acidification. Biogeosciences 6, 2121–2133 (2009).
Schulz, K. G., Barcelos e Ramos, J., Zeebe, R. E. & Riebesell, U. CO2 perturbation experiments: similarities and differences between dissolved inorganic carbon and total alkalinity manipulations. Biogeosciences 6, 2145–2153 (2009).
Berg, I. A., Kockelkorn, D., Buckel, W. & Fuchs, G. A 3-hydroxypropionate/4-hydroxybutyrate autotrophic carbon dioxide assimilation pathway in Archaea. Science 318, 1782–1786 (2007).
Badger, M. R. & Bek, E. J. Multiple Rubisco forms in proteobacteria: their functional significance in relation to CO2 acquisition by the CBB cycle. J. Exp. Bot. 59, 1525–1541 (2008).
Kock, A. & Bange, H. W. Nitrite removal improves hydroxylamine analysis in aqueous solution by conversion with iron(III). Environ. Chem. 10, 64–71 (2013).
Frame, C. H. & Casciotti, K. L. Biogeochemical controls and isotopic signatures of nitrous oxide production by a marine ammonia-oxidizing bacterium. Biogeosciences 7, 2695–2709 (2010).
Santoro, A. E., Buchwald, C., McIlvin, M. R. & Casciotti, K. L. Isotopic signature of N2O produced by marine ammonia-oxidizing Archaea. Science 333, 1282–1285 (2011).
Yamagishi, H. et al. Role of nitrification and denitrification on the nitrous oxide cycle in the eastern tropical North Pacific and Gulf of California. J. Geophys. Res. Biogeosci. 112, G02015 (2007).
Toyoda, S. & Yoshida, N. Determination of nitrogen isotopomers of nitrous oxide on a modified isotope ratio mass spectrometer. Anal. Chem. 71, 4711–4718 (1999).
Toyoda, S., Mutobe, H., Yamagishi, H., Yoshida, N. & Tanji, Y. Fractionation of N2O isotopomers during production by denitrifier. Soil Biol. Biochem. 37, 1535–1545 (2005).
Sigman, D. M. et al. A bacterial method for the nitrogen isotopic analysis of nitrate in seawater and freshwater. Anal. Chem. 73, 4145–4153 (2001).
Peng, X. et al. Revisiting nitrification in the Eastern Tropical South Pacific: a focus on controls. Geophys. Res. Oceans 121, 1667–1684 (2016).
Charpentier, J., Farias, L., Yoshida, N., Boontanon, N. & Raimbault, P. Nitrous oxide distribution and its origin in the central and eastern South Pacific Subtropical Gyre. Biogeosciences 4, 729–741 (2007).
Acknowledgements
We thank the captain, officers, crew, scientists and technicians for their assistance during the KS-16-8 and YK16-16 cruises and M. C. Honda, PI of the MR13-04 cruise. This project was financially supported by Kakenhi grants (project nos 23224013, 15H05822, 15H05471 and 17H06105) of the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT) and the Swiss National Science Foundation (project no. PBNEP2-142954).
Author information
Authors and Affiliations
Contributions
F.B., C.Y., A.M. and S.T. conceived this study, designed and executed the experiments, analysed the results and wrote the paper. M.W. calculated pH values from DIC and total alkalinity for all cruises. Y.M. measured δ15NO3− values to estimate nitrification rates. S.K., T.F. and N.H. contributed to the organization of the sampling campaigns. All authors contributed to the interpretation of the results and the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Climate Change thanks Hermann Bange, Shuh-Ji Kao and Brian Popp for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1 and 2 and Figs. 1–4.
Rights and permissions
About this article
Cite this article
Breider, F., Yoshikawa, C., Makabe, A. et al. Response of N2O production rate to ocean acidification in the western North Pacific. Nat. Clim. Chang. 9, 954–958 (2019). https://doi.org/10.1038/s41558-019-0605-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41558-019-0605-7
This article is cited by
-
Effects of acidification on nitrification and associated nitrous oxide emission in estuarine and coastal waters
Nature Communications (2023)
-
Epipelagic nitrous oxide production offsets carbon sequestration by the biological pump
Nature Geoscience (2023)
-
The marine nitrogen cycle: new developments and global change
Nature Reviews Microbiology (2022)
-
Nitrous oxide and methane in a changing Arctic Ocean
Ambio (2022)
-
Microbial N2O consumption in and above marine N2O production hotspots
The ISME Journal (2021)