Abstract
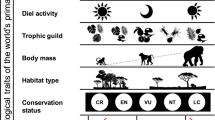
Climate-change-driven alterations in the extent and intensity of extreme weather events may have catastrophic consequences for primate populations. Using a trait-based approach, we assessed the vulnerability of the world’s 607 primate taxa to effects of cyclones and droughts—two types of extreme climatic events that are expected to increase and/or intensify in the future. We found that 16% of primate taxa are vulnerable to cyclones, particularly taxa in Madagascar; 22% are vulnerable to droughts, mainly taxa in the Malaysia Peninsula, North Borneo, Sumatra and tropical moist forests of West Africa. These findings will help with prioritization of primate conservation efforts. They indicate a need for increased efforts to investigate the context-specific mechanisms underpinning primates’ vulnerability to extreme climatic events.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data supporting the findings of this study are available through the references provided within the article or the supplemental materials. Additional data related to this paper may be requested from the corresponding author.
Change history
10 September 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Lambert, J. E. & Garber, P. A. Evolutionary and ecological implications of primate seed dispersal. Am. J. Primatol. 45, 9–28 (1998).
Nuñez‐Iturri, G. & Howe, H. F. Bushmeat and the fate of trees with seeds dispersed by large primates in a lowland rain forest in western Amazonia. Biotropica 39, 348–354 (2007).
Wich, S. A. & Marshall, A. J. An Introduction to Primate Conservation (Oxford Univ. Press, 2016).
Cowlishaw, G. & Dunbar, R. I. Primate Conservation Biology (Univ. of Chicago Press, 2000)
Estrada, A. et al. Impending extinction crisis of the world’s primates: why primates matter. Sci. Adv. 3, e1600946 (2017).
Ameca y Juárez, E. I., Mace, G. M., Cowlishaw, G. & Pettorelli, N. Natural population die-offs: causes and consequences for terrestrial mammals. Trends Ecol. Evol. 27, 272–277 (2012).
Anderson, S. C., Branch, T. A., Cooper, A. B. & Dulvy, N. K. Black-swan events in animal populations. Proc. Natl Acad. Sci. USA 114, 3252–3257 (2017).
Ameca y Juárez, E. I., Mace, G. M., Cowlishaw, G., Cornforth, W. A. & Pettorelli, N. Assessing exposure to extreme climatic events for terrestrial mammals. Conserv. Lett. 6, 145–153 (2013).
Wiederholt, R. & Post, E. Tropical warming and the dynamics of endangered primates. Biol. Lett. 6, 257–260 (2010).
Wiederholt, R. & Post, E. Birth seasonality and offspring production in threatened neotropical primates related to climate. Glob. Change Biol. 17, 3035–3045 (2011).
Campos, F. A., Jack, K. M. & Fedigan, L. M. Climate oscillations and conservation measures regulate white-faced capuchin population growth and demography in a regenerating tropical dry forest in Costa Rica. Biol. Conserv. 186, 204–213 (2015).
Dunham, A. E., Erhart, E. M., Overdorff, D. J. & Wright, P. C. Evaluating effects of deforestation, hunting, and El Niño events on a threatened lemur. Biol. Conserv. 141, 287–297 (2008).
Dunham, A. E., Erhart, E. M. & Wright, P. C. Global climate cycles and cyclones: consequences for rainfall patterns and lemur reproduction in southeastern Madagascar. Glob. Change Biol. 17, 219–227 (2011).
Tarnaud, L. & Simmen, B. A major increase in the population of brown lemurs on Mayotte since the decline reported in 1987. Oryx 36, 297–300 (2002).
Waite, T. A., Chhangani, A. K., Campbell, L. G., Rajpurohit, L. S. & Mohnot, S. M. Sanctuary in the city: urban monkeys buffered against catastrophic die-off during ENSO-related drought. EcoHealth 4, 278–286 (2007).
Diez, J. M. et al. Will extreme climatic events facilitate biological invasions? Front. Ecol. Environ. 10, 249–257 (2012).
Wernberg, T. et al. An extreme climatic event alters marine ecosystem structure in a global biodiversity hotspot. Nat. Clim. Change 3, 78 (2013).
Foden, W. B. & Young, B. E. IUCN Guidelines for Assessing Species’ Vulnerability to Climate Change (IUCN Species Survival Commission, 2017).
Pacifici, M. et al. Assessing species vulnerability to climate change. Nat. Clim. Change 5, 215–224 (2015).
Dawson, T. P., Jackson, S. T., House, J. I., Prentice, I. C. & Mace, G. M. Beyond predictions: biodiversity conservation in a changing climate. Science 332, 53–58 (2011).
Pacifici, M. et al. Species’ traits influenced their response to recent climate change. Nat. Clim. Change 7, 205 (2017).
Small-Lorenz, S. L., Culp, L. A., Ryder, T. B., Will, T. C. & Marra, P. P. A blind spot in climate change vulnerability assessments. Nat. Clim. Change 3, 91 (2013).
The IUCN Red List of Threatened Species (IUCN, accessed 5 January 2017); http://www.iucnredlist.org.
Climate Change 2014: Synthesis Report (eds Core Writing Team, Pachauri, R. K. and Meyer, L. A.) (IPCC, 2014).
Foden, W. B. et al. Identifying the world’s most climate change vulnerable species: a systematic trait-based assessment of all birds, amphibians and corals. PLoS ONE 8, e65427 (2013).
Knutson, T. R. et al. Tropical cyclones and climate change. Nat. Geosci. 3, 157–163 (2010).
Bender, M. A. et al. Modeled impact of anthropogenic warming on the frequency of intense Atlantic hurricanes. Science 327, 454–458 (2010).
Behie, A. M., Kutz, S. & Pavelka, M. S. Cascading effects of climate change: do hurricane‐damaged forests increase risk of exposure to parasites? Biotropica 46, 25–31 (2014).
Fardi, S., Sauther, M. L., Cuozzo, F. P., Jacky, I. A. & Bernstein, R. M. The effect of extreme weather events on hair cortisol and body weight in a wild ring‐tailed lemur population (Lemur catta) in southwestern Madagascar. Am. J. Primatol. 80, e22731 (2018).
Ameca y Juárez, E. I., Ellis, E. A. & Rodríguez-Luna, E. Quantifying the severity of hurricanes on extinction probabilities of a primate population: insights into “Island” extirpations. Am. J. Primatol. 77, 786–800 (2015).
Dai, A. Drought under global warming: a review. Wiley Interdiscip. Rev. Clim. Change 2, 45–65 (2011).
Sheffield, J., Wood, E. F. & Roderick, M. L. Little change in global drought over the past 60 years. Nature 491, 435–438 (2012).
Dai, A. Increasing drought under global warming in observations and models. Nat. Clim. Change 3, 52–58 (2013).
Trenberth, K. E. et al. Global warming and changes in drought. Nat. Clim. Change 4, 17–22 (2014).
Carnicer, J. et al. Widespread crown condition decline, food web disruption, and amplified tree mortality with increased climate change-type drought. Proc. Natl Acad. Sci. USA 108, 1474–1478 (2011).
Ingram, J. C. & Dawson, T. P. Climate change impacts and vegetation response on the island of Madagascar. Philos. Trans. R. Soc. Lond. A 363, 55–59 (2005).
Harper, G. J., Steininger, M. K., Tucker, C. J., Juhn, D. & Hawkins, F. Fifty years of deforestation and forest fragmentation in Madagascar. Environ. Conserv. 34, 325–333 (2007).
Schwitzer, C. et al. Averting lemur extinctions amid Madagascar’s political crisis. Science 343, 842–843 (2014).
Woodruff, D. S. Biogeography and conservation in Southeast Asia: how 2.7 million years of repeated environmental fluctuations affect today’s patterns and the future of the remaining refugial-phase biodiversity. Biodivers. Conserv. 19, 919–941 (2010).
Vijay, V., Pimm, S. L., Jenkins, C. N. & Smith, S. J. The impacts of oil palm on recent deforestation and biodiversity loss. PLoS ONE 11, e0159668 (2016).
Swarna Nantha, H. & Tisdell, C. The orangutan–oil palm conflict: economic constraints and opportunities for conservation. Biodivers. Conserv. 18, 487–502 (2009).
Meijaard, E. et al. Quantifying killing of orangutans and human–orangutan conflict in Kalimantan, Indonesia. PLoS ONE 6, e27491 (2011).
Maron, M., McAlpine, C. A., Watson, J. E. M., Maxwell, S. & Barnard, P. Climate-induced resource bottlenecks exacerbate species vulnerability: a review. Divers. Distrib. 21, 731–743 (2015).
Dalerum, F. Identifying the role of conservation biology for solving the environmental crisis. Ambio 43, 839–846 (2014).
Ghil, M. et al. Extreme events: dynamics, statistics and prediction. Nonlin. Process. Geophys. 18, 295–350 (2011).
Seneviratne, S. et al. in Managing the risks of extreme events and disasters to advance climate change adaptation: IPCC Special Report (eds Field, C. B. et al.) (IPCC, Cambridge Univ. Press, 2012).
Cardillo, M. et al. The predictability of extinction: biological and external correlates of decline in mammals. Proc. R. Soc. Lond. B 275, 1441–1448 (2008).
Cowlishaw, G., Pettifor, R. A. & Isaac, N. J. High variability in patterns of population decline: the importance of local processes in species extinctions. Proc. R. Soc. Lond. B 276, 63–69 (2008).
Böhm, M. et al. Hot and bothered: using trait-based approaches to assess climate change vulnerability in reptiles. Biol. Conserv. 204, 32–41 (2016).
Chown, S. L. Temporal biodiversity change in transformed landscapes: a southern African perspective. Philos. Trans. R. Soc. Lond. B 365, 3729–3742 (2010).
Encyclopedia of Life (EOL, accessed 20 January 2017); http://www.eol.org.
Jones, K. E. et al. PanTHERIA: a species-level database of life history, ecology, and geography of extant and recently extinct mammals. Ecology 90, 2648–2648 (2009).
Myers, P. et al. The Animal Diversity Web (Univ. Michigan, accessed 10 January 2017); http://animaldiversity.org
Purvis, A., Gittleman, J. L., Cowlishaw, G. & Mace, G. M. Predicting extinction risk in declining species. Proc. R. Soc. Lond. B 267, 1947–1952 (2000).
Davies, R. G. et al. Human impacts and the global distribution of extinction risk. Proc. R. Soc. Lond. B 273, 2127–2133 (2006).
Woodward, G. et al. Climate change impacts in multispecies systems: drought alters food web size structure in a field experiment. Philos. Trans. R. Soc. Lond. B 367, 2990–2997 (2012).
Carbone, C., Cowlishaw, G., Isaac, N. J. & Rowcliffe, J. M. How far do animals go? Determinants of day range in mammals. Am. Nat. 165, 290–297 (2004).
Stenseth, N. C. & Lidicker, W. Z. Z. J. in Animal Dispersal: Small Mammals as a Model (eds Stenseth, N. C. & Lidicker Jr, W. Z. Z.) 4–20 (Chapman & Hall, 1992).
Cowlishaw, G. & Dunbar, R. I. Primate Conservation Biology (Univ. of Chicago Press, 2000).
Campos, F. A. & Fedigan, L. M. Behavioral adaptations to heat stress and water scarcity in white-faced capuchins (Cebus capucinus) in Santa Rosa National Park, Costa Rica. Am. J. Phys. Anthropol. 138, 101–111 (2009).
Aristizabal, J. F., Lévêque, L., Chapman, C. A. & Serio-Silva, J. C. Impacts of temperature on behaviour of the Mexican endangered black howler monkey Alouatta pigra Lawrence, 1933 (Primates: Atelidae) in a fragmented landscape. Acta Zool. Bulg. 70, 377–382 (2018).
Levy, O., Dayan, T., Porter, W. P. and Kronfeld-Schor, N. Time and ecological resilience: can diurnal animals compensate for climate change by shifting to nocturnal activity? Ecol. Monogr. 89, e01334 (2018).
Russell, G. J., Brooks, T. M., McKinney, M. M. & Anderson, C. G. Present and future taxonomic selectivity in bird and mammal extinctions. Conserv. Biol. 12, 1365–1376 (1998).
Cardillo, M., Mace, G. M., Gittleman, J. L. & Purvis, A. Latent extinction risk and the future battlegrounds of mammal conservation. Proc. Natl Acad. Sci. USA 103, 4157–4161 (2006).
Gould, L., Sussman, R. W. & Sauther, M. L. Natural disasters and primate populations: the effects of a 2-year drought on a naturally occurring population of ring-tailed lemurs (Lemur catta) in southwestern Madagascar. Int. J. Primatol. 20, 69–84 (1999).
Arroyo-Rodríguez, V., Dias, P. A. D. & Cristóbal Cristóbal-Azkarate, J. in Perspectivas en Primatología Mexicana 103–116 (Universidad Juárez Autónoma de Tabasco, 2011).
Soulè, Michael E. in Genetics and Conservation (eds Schonewald-Cox, C. M. et al.) 111–124 (Benjamin Cummings, 1983).
Fimbel, C. Ecological correlates of species success in modified habitats may be disturbance-specific and site-specific—the primates of Tiwai Island. Conserv. Biol. 8, 106–113 (1994).
Rowland, L. et al. Shock and stabilisation following long‐term drought in tropical forest from 15 years of litterfall dynamics. J. Ecol. 106, 1673–1682 (2018).
Sánchez-Galván, I. R., Díaz-Castelazo, C. & Rico-Gray, V. Effect of Hurricane Karl on a plant–ant network occurring in coastal Veracruz, Mexico. J. Trop. Ecol. 28, 603–609 (2012).
Tutin, C. E., Ham, R. M., White, L. J. & Harrison, M. J. The primate community of the Lopé Reserve, Gabon: diets, responses to fruits scarcity and effects on biomass. Am. J. Primatol. 42, 1–24 (1997).
Behie, A. M., Pavelka, M. S., Hartwell, K., Champion, J. & Notman, H. in Primate Research and Conservation in the Anthropocene, Vol. 82 (eds Behie, A. M. et al.) Ch. 13 (Cambridge Univ. Press, 2019).
Struhsaker, T. T. A recensus of vervet monkeys in the Masai–Amboseli Game Reserve, Kenya. Ecology 54, 930–932 (1973).
Berenstain, L. Responses of long-tailed macaques to drought and fire in eastern Borneo: a preliminary report. Biotropica 18, 257–262 (1986).
Bicca-Marques, J. C., Muhle, C. B., Prates, H. M., Oliveira, S. G. & Calegaro-Marques, C. Habitat impoverishment and egg predation by Alouatta caraya. Int. J. Primatol. 30, 743–748 (2009).
Brown, A. D. & Zunino, G. E. Dietary variability in Cebus apella in extreme habitats: evidence for adaptability. Folia Primatol. 54, 187–195 (1990).
IUCN. Habitats Classification Scheme, v.3.1 (IUCN, accessed February 2017); https://www.iucnredlist.org/resources/habitat-classification-scheme.
Fisher, D. O., Blomberg, S. P. & Owens, I. P. F. Extrinsic versus intrinsic factors in the decline and extinction of Australian marsupials. Proc. R. Soc. Lond. B 270, 1801–1808 (2003).
Brashares, J. S. Ecological, behavioural and life-history correlates of mammal extinctions in West Africa. Conserv. Biol. 17, 733–743 (2003).
Harcourt, A. H., Coppeto, S. A. & Parks, S. A. Rarity, specialization and extinction in primates. J. Biogeogr. 29, 445–456 (2002).
Pavelka, M. S., Brusselers, O. T., Nowak, D. & Behie, A. M. Population reduction and social disorganization in Alouatta pigra following a hurricane. Int. J. Primatol. 24, 1037–1055 (2003).
Campbell, C. J. et al. Terrestrial behavior of Ateles spp. Int. J. Primatol. 26, 1039–1051 (2005).
Harcourt, A. H. in Behavioral Ecology and Conservation (ed. Caro, T. M.) 56–79 (Oxford Univ. Press, 1998).
Schloss, C. A., Nuñez, T. A. & Lawler, J. J. Dispersal will limit ability of mammals to track climate change in the Western Hemisphere. Proc. Natl Acad. Sci. USA 109, 8606–8611 (2012).
Travis, J. M. et al. Dispersal and species’ responses to climate change. Oikos 122, 1532–1540 (2013).
Whitmee, S. & Orme, C. D. L. Predicting dispersal distance in mammals: a trait‐based approach. J. Anim. Ecol. 82, 211–221 (2013).
Schaffner, C. M., Rebecchini, L., Ramos-Fernandez, G., Vick, L. G. & Aureli, F. Spider monkeys (Ateles geoffroyi yucatenensis) cope with the negative consequences of hurricanes through changes in diet, activity budget, and fission–fusion dynamics. Int. J. Primatol. 33, 922–936 (2012).
Wilson, D. E. & Reeder, D. M. (eds) Mammal Species of the World: A Taxonomic and Geographic Reference, Vol. 1 (JHU Press, 2005).
Presence, Seasonal and Origin Attributes for Species Ranges (IUCN, accessed 5 January 2017); https://nc.iucnredlist.org/redlist/resources/files/1539614211-Mapping_attribute_codes_v1.16_2018.pdf
Roskov Y. et al. Catalogue of Life: 2017 Annual Checklist (Species 2000, ITIS, accessed 7 January 2017); www.catalogueoflife.org/annual-checklist/2017
Fan, P. et al. Description of a new species of Hoolock gibbon (Primates: Hylobatidae) based on integrative taxonomy. Am. J. Primatol. 79, e22631 (2017).
Global Risk Data Platform: Tropical Cyclones Windspeed Buffers 1970–2015 (UNEP/GRID, accessed 10 February 2017); http://preview.grid.unep.ch.
Schott, T. et al. in The Saffir–Simpson Hurricane Wind Scale, 1–4 (NOAA/National Weather Service, 2012)
Pacifici, M. et al. Generation length for mammals. Nat. Conserv. 5, 89 (2013).
O’Grady, J. J., Reed, D. H., Brook, B. W. & Frankham, R. Extinction risk scales better to generations than to years. Anim. Conserv. 11, 442–451 (2008).
Schneider, U. et al. Evaluating the hydrological cycle over land using the newly-corrected precipitation climatology from the Global Precipitation Climatology Centre (GPCC). Atmosphere 8, 52 (2017).
WMO Standardized Precipitation Index User Guide (World Meteorological Organization, 2012).
McKee, T., Doesken, N. & Kleist, J. The relationship of drought frequency and duration to time scales. In Proc. 8th Conf. Appl. Climatol. 179–183 (American Meteorological Society, 1993).
Pavelka, M. S. & Chapman, C. A. in New Perspectives in the Study of Mesoamerican Primates 143–163 (Springer, 2006)
Wunderle, J. M. Jr, Lodge, D. J. & Waide, R. B. Short-term effects of Hurricane Gilbert on terrestrial bird populations on Jamaica. Auk 109, 148–166 (1992).
Alencar, A., Nepstad, D. & Diaz, M. C. V. Forest understory fire in the Brazilian Amazon in ENSO and non-ENSO years: area burned and committed carbon emissions. Earth Interact. 10, 1–17 (2006).
AghaKouchak, A., Cheng, L., Mazdiyasni, O. & Farahmand, A. Global warming and changes in risk of concurrent climate extremes: insights from the 2014 California drought. Geophys. Res. Lett. 41, 8847–8852 (2014).
Lau, W. K. & Kim, K.-M. The 2010 Pakistan flood and Russian heat wave: teleconnection of hydrometeorological extremes. J. Hydrometeorol. 13, 392–403 (2012).
Graham, N. A. J. et al. Extinction vulnerability of coral reef fishes. Ecol. Lett. 14, 341–348 (2011).
Thomas, C. D. et al. A framework for assessing threats and benefits to species responding to climate change. Methods Ecol. Evol. 2, 125–142 (2011).
Author information
Authors and Affiliations
Contributions
L.Z. and E.I.A. conceived and designed the study. G.M.M., G.C., N.P. and W.F. contributed in the design of the vulnerability framework proposed by L.Z. and E.I.A. L.Z., E.I.A. and G.C. reviewed and collected data. L.Z. analysed data and all authors contributed greatly to the discussion of results. L.Z. wrote the initial draft of this manuscript and all authors contributed on improvements of the manuscript and agreed the final version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Nature Climate Change thanks Amanda Korstjens and other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–4 and Supplementary Figures 1–3.
Supplementary Data 1
Scores of the traits used for assessing susceptibility to cyclones and droughts.
Supplementary Data 2
Vulnerability and threatened categories in IUCN Red List of assessed primate taxa.
Rights and permissions
About this article
Cite this article
Zhang, L., Ameca, E.I., Cowlishaw, G. et al. Global assessment of primate vulnerability to extreme climatic events. Nat. Clim. Chang. 9, 554–561 (2019). https://doi.org/10.1038/s41558-019-0508-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41558-019-0508-7
This article is cited by
-
Spatial–temporal evolution characteristics and influence factors of extreme precipitation indices based on bias-corrected and gauge-measured precipitation data in the main river basins of China, 1980–2020
Theoretical and Applied Climatology (2024)
-
Water scooping: tool use by a wild bonobo (Pan paniscus) at LuiKotale, a case report
Primates (2024)
-
Investigating monthly geopotential height changes and mid-latitude Northern Hemisphere westerlies
Theoretical and Applied Climatology (2024)
-
Animal-borne sensors as a biologically informed lens on a changing climate
Nature Climate Change (2023)
-
A conceptual framework for assessing behavioral flexibility of species in response to extreme climatic events
Scientific Reports (2023)