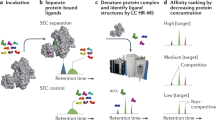
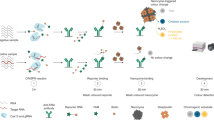
Abstract
Biomarker discovery is essential for the understanding, diagnosis, targeted therapy and prognosis assessment of malignant diseases. However, it remains a huge challenge due to the lack of sensitive methods to identify disease-specific rare molecules. Here we present MORAC, molecular recognition based on affinity and catalysis, which enables the effective identification of candidate biomarkers with low abundance. MORAC relies on a class of DNAzymes, each cleaving a sole RNA linkage embedded in their DNA chain upon specifically sensing a complex system with no prior knowledge of the system’s molecular content. We show that signal amplification from catalysis ensures the DNAzymes high sensitivity (for target probing); meanwhile, a simple RNA-to-DNA mutation can shut down their RNA cleavage ability and turn them into a pure affinity tool (for target pulldown). Using MORAC, we identify previously unknown, low-abundance candidate biomarkers with clear clinical value, including apolipoprotein L6 in breast cancer and seryl-tRNA synthetase 1 in polyps preceding colon cancer.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in the main text or the supplementary materials. Requests for resources and reagents should be directed to the corresponding authors. Source data are provided with this paper.
References
Crosby, D. et al. Early detection of cancer. Science 375, eaay9040 (2022).
Cilento, E. M. et al. Mass spectrometry: a platform for biomarker discovery and validation for Alzheimer’s and Parkinson’s diseases. J. Neurochem. 151, 397–416 (2019).
Kim, D. et al. Proteomics analysis reveals differential pattern of widespread protein expression and novel role of histidine-rich glycoprotein and lipopolysaccharide-binding protein in rheumatoid arthritis. Int. J. Biol. Macromol. 109, 704–710 (2018).
Thomas, S., Hao, L., Ricke, W. A. & Li, L. Biomarker discovery in mass spectrometry-based urinary proteomics. Proteomics Clin. Appl. 10, 358–370 (2016).
Keshishian, H., Addona, T., Burgess, M., Kuhn, E. & Carr, S. A. Quantitative, multiplexed assays for low abundance proteins in plasma by targeted mass spectrometry and stable isotope dilution. Mol. Cell. Proteomics 6, 2212–2229 (2007).
Drabovich, A. P. & Diamandis, E. P. Combinatorial peptide libraries facilitate development of multiple reaction monitoring assays for low-abundance proteins. J. Proteome Res. 9, 1236–1245 (2010).
Wikstrand, C. J. & Bigner, D. D. Expression of human fetal brain antigens by human tumors of neuroectodermal origin as defined by monoclonal antibodies. Cancer Res. 42, 267–275 (1982).
Kobayashi, M., Katayama, H., Fahrmann, J. F. & Hanash, S. M. Development of autoantibody signatures for common cancers. Semin. Immunol. 47, 101388 (2020).
Daniels, D. A., Chen, H., Hicke, B. J., Swiderek, K. M. & Gold, L. A tenascin-C aptamer identified by tumor cell SELEX: systematic evolution of ligands by exponential enrichment. Proc. Natl Acad. Sci. USA 100, 15416–15421 (2003).
Sefah, K., Shangguan, D., Xiong, X., O’Donoghue, M. B. & Tan, W. Development of DNA aptamers using cell-SELEX. Nat. Protoc. 5, 1169–1185 (2010).
Xiong, H. et al. Cancer protein biomarker discovery based on nucleic acid aptamers. Int. J. Biol. Macromol. 132, 190–202 (2019).
Ray, P., Rialon-Guevara, K. L., Veras, E., Sullenger, B. A. & White, R. R. Comparing human pancreatic cell secretomes by in vitro aptamer selection identifies cyclophilin B as a candidate pancreatic cancer biomarker. J. Clin. Invest. 122, 1734–1741 (2012).
Breaker, R. R. & Joyce, G. F. A DNA enzyme that cleaves RNA. Chem. Biol. 1, 223–229 (1994).
Schlosser, K. & Li, Y. Biologically inspired synthetic enzymes made from DNA. Chem. Biol. 16, 311–322 (2009).
Silverman, S. K. DNA as a versatile chemical component for catalysis, encoding, and stereocontrol. Angew. Chem. Int. Ed. 49, 7180–7201 (2010).
Aguirre, S. D., Ali, M. M., Salena, B. J. & Li, Y. A sensitive DNA enzyme-based fluorescent assay for bacterial detection. Biomolecules 3, 563–577 (2013).
Ali, M. M., Aguirre, S. D., Lazim, H. & Li, Y. Fluorogenic DNAzyme probes as bacterial indicators. Angew. Chem. Int. Ed. 50, 3751–3754 (2011).
Shen, Z. et al. A catalytic DNA activated by a specific strain of bacterial pathogen. Angew. Chem. Int. Ed. 55, 2431–2434 (2016).
Geng, X. et al. Selective and sensitive detection of chronic myeloid leukemia using fluorogenic DNAzyme probes. Anal. Chim. Acta 1123, 28–35 (2020).
He, S. et al. Highly specific recognition of breast tumors by an RNA-cleaving fluorogenic DNAzyme probe. Anal. Chem. 87, 569–577 (2015).
Smith, E. E. & Malik, H. S. The apolipoprotein L family of programmed cell death and immunity genes rapidly evolved in primates at discrete sites of host-pathogen interactions. Genome Res. 19, 850–858 (2009).
Vanhamme, L. et al. Apolipoprotein L-I is the trypanosome lytic factor of human serum. Nature 422, 83–87 (2003).
Pérez-Morga, D. et al. Apolipoprotein L-I promotes trypanosome lysis by forming pores in lysosomal membranes. Science 309, 469–472 (2005).
Wang, M. et al. PaxDb, a database of protein abundance averages across all three domains of life. Mol. Cell. Proteomics 11, 492–500 (2012).
Andy Hu, C.-A., Zhaorigetu, S., Davidson, W. S. & Laskey, W. ApoL6: a novel biomarker of apoptotic activity in evolving ST-segment myocardial infarction in man. J. Integr. Cardiol. Open Access https://doi.org/10.31487/j.JICOA.2020.04.10 (2020).
Murphy, I. et al. ApoL6 induces dichotomous cell death phenotype involving both apoptosis and necroptosis in cancer cells. Clin. Oncol. Res. https://doi.org/10.31487/j.COR.2020.07.12 (2020).
Wu, S. Z. et al. A single-cell and spatially resolved atlas of human breast cancers. Nat. Genet. 53, 1334–1347 (2021).
Mertins, P. G. et al. Proteogenomics connects somatic mutations to signalling in breast cancer. Nature 534, 55–62 (2016).
Krug, K. et al. Proteogenomic landscape of breast cancer tumorigenesis and targeted therapy. Cell 183, 1436–1456 (2020).
Gaudet, R. G. et al. A human apolipoprotein L with detergent-like activity kills intracellular pathogens. Science 373, eabf8113 (2021).
How to combine chromatography techniques to purify an untagged or native protein. Cytiva https://www.cytivalifesciences.com/en/us/Solutions/Protein-Research/Knowledge-center/Protein-purification-methods/How-to-combine-chromatography-techniques/Untagged-protein-purification-protocols (2021).
Fukui, H., Hanaoka, R. & Kawahara, A. Noncanonical activity of seryl-tRNA synthetase is involved in vascular development. Circ. Res. 104, 1253–1259 (2009).
Herzog, W., Müller, K., Huisken, J. & Stainier, D. Y. R. Genetic evidence for a noncanonical function of seryl-tRNA synthetase in vascular development. Circ. Res. 104, 1260–1266 (2009).
Xu, X. et al. Unique domain appended to vertebrate tRNA synthetase is essential for vascular development. Nat. Commun. 3, 681 (2012).
Imashimizu, M., Takahashi, M., Amano, R. & Nakamura, Y. Single-round isolation of diverse RNA aptamers from a random sequence pool. Biol. Methods Protoc. 3, bpy004 (2018).
Nagtegaal, I. D. et al. The 2019 WHO classification of tumours of the digestive system. Histopathology 76, 182–188 (2020).
Zhou, W., Zhang, Y., Ding, J. & Liu, J. In vitro selection in serum: RNA-cleaving DNAzymes for measuring Ca2+ and Mg2+. ACS Sens. 1, 600–606 (2016).
Gootenberg, J. S. et al. Nucleic acid detection with CRISPR-Cas13a/C2c2. Science 356, 438–442 (2017).
Chen, J. S. et al. CRISPR-Cas12a target binding unleashes indiscriminate single-stranded DNase activity. Science 360, 436–439 (2018).
Gootenberg, J. S. et al. Multiplexed and portable nucleic acid detection platform with Cas13, Cas12a, and Csm6. Science 360, 439–444 (2018).
Zhu, X. et al. Efficacy and mechanism of the combination of PARP and CDK4/6 inhibitors in the treatment of triple-negative breast cancer. J. Exp. Clin. Cancer Res. 40, 122–140 (2021).
Acknowledgements
We acknowledge funding by the National Key Research and Development Program of China (2020YFA0908901 to H.G.), the National Natural Science Foundation of China (82121002 and 91859104 to H.G.; 21991134 and T2188102 to C.F.), the Program of Shanghai Academic Research Leader (22XD1421500 to H.G.) and the New Cornerstone Science Foundation (to C.F.). The funders had no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Q.H., Z.T., A.Y. and L.-P.G. conducted the selection, target identification and experimental validation of the target. Q.S., X.D., P.W., W.Z., X.G., D.S. and T.F. conducted the experimental validation of the target. X.-Y.L. and A.Y. collected clinical specimens. D.Y., J.L. and C.F. analysed the data. Y.-S.Z. and Y.-Z.J. supervised the project. H.G. developed the initial concept, supervised the project, interpreted the data and wrote the manuscript. All authors participated in the discussions and reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Three Chinese patents have been filed, with a status of initiative for examination as to substance. Q.H., Z.T., L.-P.G., X.-Y.L., T.F., Y.-Z.J. and H.G. declare the following competing interests: patent no. CN202110364792.0 was applied for by Fudan University Shanghai Cancer Center, covering the identified DNAzyme probes that selectively sense MDA-MB-231 cells in this study; and patent no. CN202110361337.5 was also applied for by Fudan University Shanghai Cancer Center, covering the method to identify the potential biomarker (APOL6) of breast cancer based on the DNAzyme probes in this study. Z.T., A.Y., Q.S., D.S., Y.-S.Z. and H.G. declare the following competing interests: patent no. CN202111198079.X was applied for by Zhongshan Hospital, Fudan University, covering the findings of the DNAzyme probes that selectively sense LGIN/HGIN specimens and the probes’ utility in biomarker discovery in this study. X.D., P.W., W.Z., X.G., D.Y., C.F. and J.L. declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Feng Li, Chao Liang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–26, Materials and Methods and unprocessed gels and micrographs for Supplementary Figs. 3–6, 8, 10, 11, 14, 16, 18–21, 23 and 26.
Supplementary Data 1
Supplementary Tables 1–4.
Supplementary Video 1
Vesicle-like movement of APOL6–Emerald in MDA-MB-231 cells.
Supplementary Data 2
Statistical source data for Supplementary Figs. 2, 4, 5, 7, 15–17, 21, 22, 24 and 26.
Source data
Source Data Fig. 2
Unprocessed gels and western blots.
Source Data Fig. 3
Unprocessed micrographs.
Source Data Fig. 4
Unprocessed gels and micrographs.
Source Data Fig. 5
Unprocessed micrographs.
Source Data Figs. 2–5
Statistical source data for Figs. 2–5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, Q., Tong, Z., Yalikong, A. et al. DNAzyme-based faithful probing and pulldown to identify candidate biomarkers of low abundance. Nat. Chem. 16, 122–131 (2024). https://doi.org/10.1038/s41557-023-01328-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-023-01328-5
This article is cited by
-
A non-FRET DNA reporter that changes fluorescence colour upon nuclease digestion
Nature Nanotechnology (2024)