Abstract
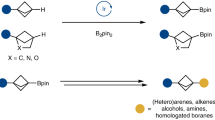
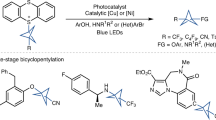
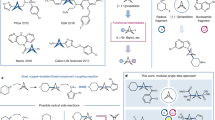
Strained bicyclic substructures are increasingly relevant in medicinal chemistry discovery research because of their role as bioisosteres. Over the last decade, the successful use of bicyclo[1.1.1]pentane (BCP) as a para-disubstituted benzene replacement has made it a highly valuable pharmacophore. However, various challenges, including limited and lengthy access to useful BCP building blocks, are hampering early discovery research. Here we report a single-step transition-metal-free multi-component approach to synthetically versatile BCP boronates. Radicals derived from commonly available carboxylic acids and organohalides perform additions onto [1.1.1]propellane to afford BCP radicals, which then engage in polarity-matched borylation. A wide array of alkyl-, aryl- and alkenyl-functionalized BCP boronates were easily prepared. Late-stage functionalization performed on natural products and approved drugs proceeded with good efficiency to generate the corresponding BCP conjugates. Various photoredox transformations forging C–C and C–N bonds were demonstrated by taking advantage of BCP trifluoroborate salts derived from the BCP boronates.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data supporting the findings of this study are available in this Article and its Supplementary Information. Crystallographic data for the structure 4ar reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under the deposition number CCDC 2105768. Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
van de Waterbeemd, H. & Gifford, E. ADMET in silico modelling: towards prediction paradise? Nat. Rev. Drug Discov. 2, 192–204 (2003).
Honorio, K. M., Moda, T. L. & Andricopulo, A. D. Pharmacokinetic properties and in silico ADME modeling in drug discovery. Med. Chem. 9, 163–176 (2013).
Lovering, F., Bikker, J. & Humblet, C. Escape from flatland: increasing saturation as an approach to improving clinical success. J. Med. Chem. 52, 6752–6756 (2009).
Ishikawa, M. & Hashimoto, Y. Improvement in aqueous solubility in small molecule drug discovery programs by disruption of molecular planarity and symmetry. J. Med. Chem. 54, 1539–1554 (2011).
Meanwell, N. A. Synopsis of some recent tactical application of bioisosteres in drug design. J. Med. Chem. 54, 2529–2591 (2011).
Lovering, F. Escape from flatland 2: complexity and promiscuity. Med. Chem. Commun. 4, 515–519 (2013).
Walker, M. A. Novel tactics for designing water-soluble molecules in drug discovery. Expert Opin. Drug Discov. 9, 1421–1433 (2014).
Ritchie, T. J. & Macdonald, S. J. F. The impact of aromatic ring count on compound developability – are too many aromatic rings a liability in drug design? Drug Discov. Today 14, 1011–1020 (2009).
Auberson, Y. P. et al. Improving nonspecific binding and solubility: bicycloalkyl groups and cubanes as para-phenyl bioisosteres. ChemMedChem 12, 590–598 (2017).
Mykhailiuk, P. K. Saturated bioisosteres of benzene: where to go next? Org. Biomol. Chem. 17, 2839–2849 (2019).
Stepan, A. F. et al. Application of the bicyclo[1.1.1]pentane motif as a nonclassical phenyl ring bioisostere in the design of a potent and orally active γ-secretase inhibitor. J. Med. Chem. 55, 3414–3424 (2012).
Wiberg, K. B. & Walker, F. H. [1.1.1]Propellane. J. Am. Chem. Soc. 104, 5239–5240 (1982).
Wiberg, K. B. Small ring propellanes. Chem. Rev. 89, 975–983 (1989).
Kanazawa, J. & Uchiyama, M. Recent advances in the synthetic chemistry of bicyclo[1.1.1]pentane. Synlett 30, 1–11 (2019).
Pramanik, M. M. D., Qian, H., Xiao, W.-J. & Chen, J.-R. Photoinduced strategies towards strained molecules. Org. Chem. Front. 7, 2531–2537 (2020).
Makarov, I. S., Brocklehurst, C. E., Karaghiosoff, K., Koch, G. & Knochel, P. Synthesis of bicyclo[1.1.1]pentane bioisosteres of internal alkynes and para-disubstituted benzenes from [1.1.1]propellane. Angew. Chem. Int. Ed. 56, 12774–12777 (2017).
Schwärzer, K., Zipse, H., Karaghiosoff, K. & Knochel, P. Highly regioselective addition of allylic zinc halides and various zinc enolates to [1.1.1]propellane. Angew. Chem. Int. Ed. 59, 20235–20241 (2020).
Kanazawa, J., Maeda, K. & Uchiyama, M. Radical multicomponent carboamination of [1.1.1]propellane. J. Am. Chem. Soc. 139, 17791–17794 (2017).
Kondo, M. et al. Silaboration of [1.1.1]propellane: a storable feedstock for bicyclo[1.1.1]pentane derivatives. Angew. Chem. Int. Ed. 59, 1970–1974 (2020).
Caputo, D. F. J. et al. Synthesis and applications of highly functionalized 1-halo-3-substituted bicyclo[1.1.1]pentanes. Chem. Sci. 9, 5295–5300 (2018).
Nugent, J. et al. A general route to bicyclo[1.1.1]pentanes through photoredox catalysis. ACS Catal. 9, 9568–9574 (2019).
Yu, S., Jing, C., Noble, A. & Aggarwal, V. K. 1,3-Difunctionalizations of [1.1.1]propellane via 1,2-metallate rearrangements of boronate complexes. Angew. Chem. Int. Ed. 59, 3917–3921 (2020).
Kim, J. H., Ruffoni, A., Al-Faiyz, Y. S. S., Sheikh, N. S. & Leonori, D. Divergent strain-release amino-functionalization of [1.1.1]propellane with electrophilic nitrogen-radicals. Angew. Chem. Int. Ed. 59, 8225–8231 (2020).
Zhang, X. et al. Copper-mediated synthesis of drug-like bicyclopentanes. Nature 580, 220–226 (2020).
Shelp, R. A. et al. Strain-release 2-azaallyl anion addition/borylation of [1.1.1]propellane: synthesis and functionalization of benzylamine bicyclo[1.1.1]pentyl boronates. Chem. Sci. 12, 7066–7072 (2021).
Shin, S., Lee, S., Choi, W., Kim, N. & Hong, S. Visible-light-induced 1,3-aminopyridylation of [1.1.1]propellane with N-aminopyridinium salts. Angew. Chem. Int. Ed. 60, 7873–7879 (2021).
Wu, Z., Xu, Y., Zhang, H., Wu, X. & Zhu, C. Radical-mediated sulfonyl alkynylation, allylation, and cyanation of propellane. Chem. Commun. 57, 6066–6069 (2021).
VanHeyst, M. D. et al. Continuous flow-enabled synthesis of bench-stable bicyclo[1.1.1]pentane trifluoroborate salts and their utilization in metallaphotoredox cross-couplings. Org. Lett. 22, 1648–1654 (2020).
Merchant, R. R. & Lopez, J. A. A general C(sp3)–C(sp3) cross-coupling of benzyl sulfonylhydrazones with alkyl boronic acids. Org. Lett. 22, 2271–2275 (2020).
Zarate, C. et al. Development of scalable routes to 1-bicyclo[1.1.1]pentylpyrazoles. Org. Process Res. Dev. 25, 642–647 (2021).
Yang, Y. et al. Practical and modular construction of C(sp3)-rich alkyl boron compounds. J. Am. Chem. Soc. 143, 471–480 (2021).
Ripenko, V., Vysochyn, D., Klymov, I., Zhersh, S. & Mykhailiuk, P. K. Large-scale synthesis and modifications of bicyclo[1.1.1]pentane-1,3-dicarboxylic acid (BCP). J. Org. Chem. 86, 14061–14068 (2021).
Yang, Y. et al. An intramolecular coupling approach to alkyl bioisosteres for the synthesis of multisubstituted bicycloalkyl boronates. Nat. Chem. 13, 950–955 (2021).
Fawcett, A. et al. Photoinduced decarboxylative borylation of carboxylic acids. Science 357, 283–286 (2017).
Friese, F. W. & Studer, A. New avenues for C–B bond formation via radical intermediates. Chem. Sci. 10, 8503–8518 (2019).
Levin, M. D., Kaszynski, P. & Michl, J. Bicyclo[1.1.1]pentanes, [n]staffanes, [1.1.1]propellanes, and tricyclo[2.1.0.02,5]pentanes. Chem. Rev. 100, 169–234 (2000).
Jin, S. et al. Visible light-induced borylation of C–O, C–N, and C–X bonds. J. Am. Chem. Soc. 142, 1603–1613 (2020).
Liu, Q. et al. Transition-metal-free borylation of alkyl iodides via a radical mechanism. Org. Lett. 21, 6597–6602 (2019).
Cheng, Y., Mück-Lichtenfeld, C. & Studer, A. Transition metal-free 1,2-carboboration of unactivated alkenes. J. Am. Chem. Soc. 140, 6221–6225 (2018).
Toriyama, F. et al. Redox-active esters in Fe-catalyzed C–C coupling. J. Am. Chem. Soc. 138, 11132–11135 (2016).
Qin, T. et al. A general alkyl-alkyl cross-coupling enabled by redox-active esters and alkylzinc reagents. Science 352, 801–805 (2016).
Fawcett, A. New Developments in Organoboron Incorporation, Homologation and Functionalization. PhD thesis, Bristol Univ. (2018).
Hu, D., Wang, L. & Li, P. Decarboxylative borylation of aliphatic esters under visible-light photoredox conditions. Org. Lett. 19, 2770–2773 (2017).
Polites, V. C., Badir, S. O., Keess, S., Jolit, A. & Molander, G. A. Nickel-catalyzed decarboxylative cross-coupling of bicyclo[1.1.1]pentyl radicals enabled by electron donor–acceptor complex photoactivation. Org. Lett. 23, 4828–4833 (2021).
Primer, D. N. & Molander, G. A. Enabling the cross-coupling of tertiary organoboron nucleophiles through radical-mediated alkyl transfer. J. Am. Chem. Soc. 139, 9847–9850 (2017).
Suzuki, J., Tanigawa, M., Inagi, S. & Fuchigami, T. Electrochemical oxidation of organotrifluoroborate compounds. ChemElectroChem 3, 2078–2083 (2016).
Pickford, H. D. et al. Twofold radical-based synthesis of N,C-difunctionalized bicyclo[1.1.1]pentanes. J. Am. Chem. Soc. 143, 9729–9736 (2021).
Matsui, J. K., Primer, D. N. & Molander, G. A. Metal-free C–H alkylation of heteroarenes with alkyltrifluoroborates: a general protocol for 1°, 2° and 3° alkylation. Chem. Sci. 8, 3512–3522 (2017).
West, M. J., Fyfe, J. W. B., Vantourout, J. C. & Watson, A. J. B. Mechanistic development and recent applications of the Chan–Lam amination. Chem. Rev. 119, 12491–12523 (2019).
Lam, P. Y. S. in Synthetic Methods in Drug Discovery Vol. 1 (eds Blakemore, D., Doyle, P. & Fobian, Y.) 242–273 (The Royal Society of Chemistry, 2016).
Rossi, S. A., Shimkin, K. W., Xu, Q., Mori-Quiroz, L. M. & Watson, D. A. Selective formation of secondary amides via the copper-catalyzed cross-coupling of alkylboronic acids with primary amides. Org. Lett. 15, 2314–2317 (2013).
Lavagnino, M. N., Liang, T. & MacMillan, D. W. C. HARC as an open-shell strategy to bypass oxidative addition in Ullmann–Goldberg couplings. Proc. Natl Acad. Sci. USA 117, 21058–21064 (2020).
Campbell, M. W., Compton, J. S., Kelly, C. B. & Molander, G. A. Three-component olefin dicarbofunctionalization enabled by nickel/photoredox dual catalysis. J. Am. Chem. Soc. 141, 20069–20078 (2019).
Yoon, U. C. & Mariano, P. S. The synthetic potential of phthalimide SET photochemistry. Acc. Chem. Res. 34, 523–533 (2001).
Cheng, Y., Mück-Lichtenfeld, C. & Studer, A. Metal-free radical borylation of alkyl and aryl iodides. Angew. Chem. Int. Ed. 57, 16832–16836 (2018).
Acknowledgements
We are grateful for financial support provided by NIGMS (R35 GM 131680 to G.A.M.). Financial support for this research was provided in part by AbbVie. AbbVie participated in the interpretation of data, review and approval of the publication. The NSF Major Research Instrumentation Program (award NSF CHE-1827457), the NIH supplements awards 3R01GM118510-03S1 and 3R01GM087605-06S1, as well as the Vagelos Institute for Energy Science and Technology supported the purchase of the NMR instruments used in this study. We thank C. W. Ross III (UPenn) for mass spectral data and M. R. Gau (UPenn) for X-ray crystallography data. We thank Frontier for donations of B2pin2, BASF for donations of B2(OH)4, Kessil for donations of LED lamps and Merck for donations of informer sets.
Author information
Authors and Affiliations
Contributions
W.D., E.Y.-P., L.L. and A.B. performed the laboratory experiments. W.D. and G.A.M. conceived the project. W.D., A.J. and G.A.M. directed the investigations. W.D., E.Y.-P., L.L. and G.A.M. analysed the experimental data and prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Edward Anderson and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
General information, experimental procedures, extended optimizations, synthesis and characterization data, mechanistic studies, discussion, X-ray crystallographic data, references, NMR spectra, Supplementary Scheme 1, Figs. 1–16 and Tables 1–13.
Supplementary Data 1
Crystallographic data for compound 4ar; CCDC reference 2105768.
Rights and permissions
About this article
Cite this article
Dong, W., Yen-Pon, E., Li, L. et al. Exploiting the sp2 character of bicyclo[1.1.1]pentyl radicals in the transition-metal-free multi-component difunctionalization of [1.1.1]propellane. Nat. Chem. 14, 1068–1077 (2022). https://doi.org/10.1038/s41557-022-00979-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-022-00979-0
This article is cited by
-
C−F bond activation enables synthesis of aryl difluoromethyl bicyclopentanes as benzophenone-type bioisosteres
Nature Communications (2024)
-
Programmable late-stage functionalization of bridge-substituted bicyclo[1.1.1]pentane bis-boronates
Nature Chemistry (2024)
-
O-, N- and C-bicyclopentylation using thianthrenium reagents
Nature Synthesis (2023)
-
2-Oxabicyclo[2.1.1]hexanes as saturated bioisosteres of the ortho-substituted phenyl ring
Nature Chemistry (2023)