Abstract
Non-covalently bound aromatic systems are ubiquitous and govern the physicochemical properties of various organic materials. They are important to many phenomena of biological and technological relevance, such as protein folding, base-pair stacking in nucleic acids, molecular recognition and self-assembly, DNA–drug interactions, crystal engineering and organic electronics. Nevertheless, their molecular dynamics and chemical reactivity, particularly in electronic excited states, are not fully understood. Here, we observe intermolecular Coulombic decay in benzene dimers, (C6H6)2—the simplest prototypes of noncovalent π–π interactions between aromatic systems. Intermolecular Coulombic decay is initiated by a carbon 2s vacancy state produced by electron-impact ionization and proceeds through ultrafast energy transfer between the benzene molecules. As a result, the dimer relaxes with the emission of a further low-energy electron (<10 eV) and a pair of C6H6+ cations undergoing Coulomb explosion. Coincident fragment-ion and electron momentum spectroscopy, accompanied by ab initio calculations, enables us to elucidate the dynamical details of this ultrafast relaxation process.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Source data are provided with this paper. The data supporting this study are also available from the corresponding author upon reasonable request.
References
Hunter, C. A. & Sanders, J. K. M. The nature of π–π interactions. J. Am. Chem. Soc. 112, 5525–5534 (1990).
Řezáĉ, J. & Hobza, P. Benchmark calculations of interaction energies in noncovalent complexes and their applications. Chem. Rev. 116, 5038–5071 (2016).
Kim, K. S., Tarakeshwar, P. & Lee, J. Y. Molecular clusters of π-systems: theoretical studies of structures, spectra, and origin of interaction energies. Chem. Rev. 100, 4145–4186 (2000).
Chen, T., Li, M. & Liu, J. π–π Stacking interaction: a nondestructive and facile means in material engineering for bioapplications. Cryst. Growth Des. 18, 2765–2783 (2018).
Mahadevi, A. S. & Sastry, G. N. Cooperativity in noncovalent interactions. Chem. Rev. 116, 2775–2825 (2016).
Burley, S. & Petsko, G. Aromatic-aromatic interaction: a mechanism of protein structure stabilization. Science 229, 23–28 (1985).
Hunter, C. A., Singh, J. & Thornton, J. M. π–π Interactions: the geometry and energetics of phenylalanine-phenylalanine interactions in proteins. J. Mol. Biol. 218, 837–846 (1991).
Yakovchuk, P., Protozanova, E. & Frank-Kamenetskii, M. D. Base-stacking and base-pairing contributions into thermal stability of the DNA double helix. Nucleic Acids Res. 34, 564–574 (2006).
Řezáĉ, J. & Hobza, P. On the nature of DNA-duplex stability. Chem. Eur. J. 13, 2983–2989 (2007).
Noller, H. F. RNA structure: reading the ribosome. Science 309, 1508–1514 (2005).
Meyer, E. A., Castellano, R. K. & Diederich, F. Interactions with aromatic rings in chemical and biological recognition. Angew. Chem. Int. Ed. 42, 1210–1250 (2003).
Philp, D. & Stoddart, J. F. Self-assembly in natural and unnatural systems. Angew. Chem. Int. Ed. 35, 1154–1196 (1996).
Hunter, C. A. Meldola lecture. The role of aromatic interactions in molecular recognition. Chem. Soc. Rev. 23, 101–109 (1994).
Vacas, T. et al. Role of aromatic rings in the molecular recognition of aminoglycoside antibiotics: implications for drug design. J. Am. Chem. Soc. 132, 12074–12090 (2010).
Frederick, C. A. et al. Structural comparison of anticancer drug-DNA complexes: adriamycin and daunomycin. Biochemistry 29, 2538–2549 (1990).
Desiraju, G. R. Crystal engineering: from molecule to crystal. J. Am. Chem. Soc. 135, 9952–9967 (2013).
Figueira-Duarte, T. M. & Müllen, K. Pyrene-based materials for organic electronics. Chem. Rev. 111, 7260–7314 (2011).
Pitoňák, M. et al. Benzene dimer: high-level wave function and density functional theory calculations. J. Chem. Theory Comput. 4, 1829–1834 (2008).
Lee, E. C. et al. Understanding of assembly phenomena by aromatic–aromatic interactions: benzene dimer and the substituted systems. J. Phys. Chem. A 111, 3446–3457 (2007).
Sinnokrot, M. O. & Sherrill, C. D. High-accuracy quantum mechanical studies of π–π interactions in benzene dimers. J. Phys. Chem. A 110, 10656–10668 (2006).
Podeszwa, R., Bukowski, R. & Szalewicz, K. Potential energy surface for the benzene dimer and perturbational analysis of π–π interactions. J. Phys. Chem. A 110, 10345–10354 (2006).
Fu, C.-F. & Tian, S. X. A comparative study for molecular dynamics simulations of liquid benzene. J. Chem. Theory Comput. 7, 2240–2252 (2011).
Pieniazek, P. A., Krylov, A. I. & Bradforth, S. E. Electronic structure of the benzene dimer cation. J. Chem. Phys. 127, 044317 (2007).
Sisourat, N., Kazandjian, S. & Miteva, T. Probing conformers of benzene dimer with intermolecular Coulombic decay spectroscopy. J. Phys. Chem. A 121, 45–50 (2017).
Deleuze, M. S., Francois, J.-P. & Kryachko, E. S. The fate of dicationic states in molecular clusters of benzene and related compounds. J. Am. Chem. Soc. 127, 16824–16834 (2005).
Kryachko, E. S. Dicationic states of benzene dimer: benzene dimer cation and benzene dication parenthood patterns. Int. J. Quantum Chem. 107, 2741–2755 (2007).
Cederbaum, L. S., Zobeley, J. & Tarantelli, F. Giant intermolecular decay and fragmentation of clusters. Phys. Rev. Lett. 79, 4778–4781 (1997).
Hergenhahn, U. Interatomic and intermolecular Coulombic decay: the early years. J. Electron Spectros. Relat. Phenomena 184, 78–90 (2011).
Kuleff, A. I. & Cederbaum, L. S. Tracing ultrafast interatomic electronic decay processes in real time and space. Phys. Rev. Lett. 98, 083201 (2007).
Schnorr, K. et al. Time-resolved measurement of interatomic Coulombic decay in Ne2. Phys. Rev. Lett. 111, 093402 (2013).
Trinter, F. et al. Evolution of interatomic Coulombic decay in the time domain. Phys. Rev. Lett. 111, 093401 (2013).
Sisourat, N. et al. Ultralong-range energy transfer by interatomic Coulombic decay in an extreme quantum system. Nat. Phys. 6, 508–511 (2010).
Havermeier, T. et al. Interatomic Coulombic decay following photoionization of the helium dimer: observation of vibrational structure. Phys. Rev. Lett. 104, 133401 (2010).
Marburger, S., Kugeler, O., Hergenhahn, U. & Möller, T. Experimental evidence for interatomic Coulombic decay in Ne clusters. Phys. Rev. Lett. 90, 203401 (2003).
Jahnke, T. et al. Experimental observation of interatomic Coulombic decay in neon dimers. Phys. Rev. Lett. 93, 163401 (2004).
Trinter, F. et al. Resonant Auger decay driving intermolecular Coulombic decay in molecular dimers. Nature 505, 664–666 (2014).
O’Keeffe, P. et al. The role of the partner atom and resonant excitation energy in interatomic Coulombic decay in rare gas dimers. J. Phys. Chem. Lett. 4, 1797–1801 (2013).
Iskandar, W. et al. Interatomic Coulombic decay as a new source of low energy electrons in slow ion-dimer collisions. Phys. Rev. Lett. 114, 033201 (2015).
Nagaya, K. et al. Interatomic Coulombic decay cascades in multiply excited neon clusters. Nat. Commun. 7, 13477 (2016).
LaForge, A. C. et al. Highly efficient double ionization of mixed alkali dimers by intermolecular Coulombic decay. Nat. Phys. 15, 247–250 (2019).
Obaid, R. et al. Intermolecular Coulombic decay in endohedral fullerene at the 4d → 4f resonance. Phys. Rev. Lett. 124, 113002 (2020).
Iskandar, W. et al. Tracing intermolecular Coulombic decay of carbon-dioxide dimers and oxygen dimers after valence photoionization. Phys. Rev. A 99, 043414 (2019).
Jahnke, T. et al. Interatomic and intermolecular Coulombic decay. Chem. Rev. 120, 11295–11369 (2020).
Jahnke, T. et al. Ultrafast energy transfer between water molecules. Nat. Phys. 6, 139–142 (2010).
Mucke, M. et al. A hitherto unrecognized source of low-energy electrons in water. Nat. Phys. 6, 143–146 (2010).
Thürmer, S. et al. On the nature and origin of dicationic, charge-separated species formed in liquid water on X-ray irradiation. Nat. Chem. 5, 590–596 (2013).
Ren, X. et al. Experimental evidence for ultrafast intermolecular relaxation processes in hydrated biomolecules. Nat. Phys. 14, 1062–1066 (2018).
Stoychev, S. D., Kuleff, A. I. & Cederbaum, L. S. Intermolecular Coulombic decay in small biochemically relevant hydrogen-bonded systems. J. Am. Chem. Soc. 133, 6817–6824 (2011).
Weikert, H.-G. & Cederbaum, L. On the satellite structure accompanying the ionization of benzene. Chem. Phys. Lett. 237, 1–6 (1995).
Baltzer, P. et al. An experimental and theoretical study of the valence shell photoelectron spectrum of the benzene molecule. Chem. Phys. 224, 95–119 (1997).
Eland, J. H. Spectra of the dications of benzene, naphthalene and azulene. Chem. Phys. 345, 82–86 (2008).
Pimblott, S. M. & LaVerne, J. A. Production of low-energy electrons by ionizing radiation. Radiat. Phys. Chem. 76, 1244–1247 (2007).
Huels, M. A., Boudaïffa, B., Cloutier, P., Hunting, D. & Sanche, L. Single, double, and multiple double strand breaks induced in DNA by 3–100 eV electrons. J. Am. Chem. Soc. 125, 4467–4477 (2003).
Alizadeh, E., Orlando, T. M. & Sanche, L. Biomolecular damage induced by ionizing radiation: the direct and indirect effects of low-energy electrons on DNA. Ann. Rev. Phys. Chem. 66, 379–398 (2015).
Ullrich, J. et al. Recoil-ion and electron momentum spectroscopy: reaction-microscopes. Rep. Prog. Phys. 66, 1463 (2003).
Ren, X., Jabbour Al Maalouf, E., Dorn, A. & Denifl, S. Direct evidence of two interatomic relaxation mechanisms in argon dimers ionized by electron impact. Nat. Commun. 7, 11093 (2016).
Zhou, J. et al. Real-time observation of ultrafast molecular rotation in weakly bound dimers. Phys. Rev. Res. 3, 023050 (2021).
Vendrell, O., Stoychev, S. D. & Cederbaum, L. S. Generation of highly damaging H2O+ radicals by inner valence shell ionization of water. ChemPhysChem 11, 1006–1009 (2010).
Iachello, F. & Oss, S. Stretching vibrations of benzene in the algebraic model. Chem. Phys. Lett. 187, 500–505 (1991).
Feng, R., Cooper, G. & Brion, C. Dipole (e,e+ion) spectroscopic studies of benzene: absolute oscillator strengths for molecular and dissociative photoionization in the VUV and soft X-ray regions. J. Electron Spectrosc. Relat. Phenomena 123, 211–223 (2002).
Řezáĉ, J. & Hobza, P. Benzene dimer: dynamic structure and thermodynamics derived from on-the-fly ab initio DFT-D molecular dynamic simulations. J. Chem. Theory Comput. 4, 1835–1840 (2008).
Erlekam, U., Frankowski, M., Meijer, G. & von Helden, G. An experimental value for the B1u C–H stretch mode in benzene. J. Chem. Phys. 124, 171101 (2006).
Felker, P. M., Maxton, P. M. & Schaeffer, M. W. Nonlinear Raman studies of weakly bound complexes and clusters in molecular beams. Chem. Rev. 94, 1787–1805 (1994).
Gokhberg, K., Kopelke, S., Kryzhevoi, N. V., Kolorenč, P. & Cederbaum, L. S. Dependence of interatomic decay widths on the symmetry of the decaying state: analytical expressions and ab initio results. Phys. Rev. A 81, 013417 (2010).
Galbraith, M. C. E. et al. Few femtosecond passage of conical intersections in the benzene cation. Nat. Commun. 8, 1018 (2017).
Ren, X. et al. An (e, 2e + ion) study of low-energy electron-impact ionization and fragmentation of tetrahydrofuran with high mass and energy resolutions. J. Chem. Phys. 141, 134314 (2014).
Frisch, M. J. et al. Gaussian 16 revision A.03 (Gaussian, 2016).
Acknowledgements
This work was jointly supported by the National Natural Science Foundation of China under grants no. 11974272 (X.R., Z.X., J.Z.) and no. 11774281 (X.R., J.Z.) and the Deutsche Forschungsgemeinschaft under project no. RE 2966/5-1 (X.R., A.D.). E.W. acknowledges a fellowship from the Alexander von Humboldt Foundation. J.Z. is grateful for support from the China Scholarship Council.
Author information
Authors and Affiliations
Contributions
X.R. and A.D. conceived and supervised the project. X.R. performed the experiments and analysed the data. X.R., J.Z. and E.W. carried out the molecular dynamics simulations. N.S. performed the energetic calculations. X.R. and A.D. wrote the first draft of the manuscript. All authors including T.Y., Z.X. and T.P. contributed to the interpretation of the data and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Chemistry thanks Elke Fasshauer and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
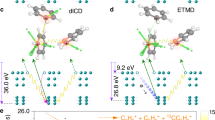
Extended Data Fig. 1 Calculated kinetic energy spectra for C6H6+ + C6H6+ Coulomb explosion process.
From top to bottom rows, the spectra correspond to the results for T-shape (a-c), PD (d-f), TT (g-i) and S (j-l) conformers of the benzene dimer, respectively. Left column: Total kinetic energy of all atoms (black lines) and the COM kinetic energy of the exploding C6H6+ + C6H6+ ion pair (red lines) as a function of propagation time; Middle column: The initial Coulomb energy calculated from the COM distance at t = 0 fs; Right column: Energy difference between the initial Coulomb energy and the KER.
Extended Data Fig. 2 Measured ion TOF spectra for different targets.
The spectra show the measurements for a mixture of C6H6 and C6D6 (M), the pure C6H6 (H) and C6D6 (D) targets and the sum of D + H (S) result. The numbered ion mass peaks are (1) (C6H2,3)+, (2) (C6H4)+, (3) (C6H5)+, (4) (C6H6)+, (5) (13CC5H6)+, (6) (13C2C4H6)+, (7) (C6D5)+, (8) (C6D5H)+, (9) (C6D6)+, (10) (13CC5D6)+ and 11 (13C2C4D6)+. The M-S difference spectrum is obtained to estimate the possible contribution of a fusion pathway.
Supplementary information
Supplementary Information
Supplementary Figs. 1–7, Table 1, Discussion Sections I–IV and references.
Supplementary Data 1
Atomic coordinates of the optimized computational models, and the initial and final configurations for molecular dynamics trajectories.
Supplementary Data 2
Statistical source data for Supplementary Figs. 1, 2 and 4–7.
Source data
Source Data Fig. 2
Statistical source data for Fig. 2a,b.
Source Data Fig. 3
Statistical source data for Fig. 3a–e.
Source Data Fig. 4
Statistical source data for Fig. 4a–c.
Source Data Fig. 5
Statistical source data for Fig. 5a,b.
Source Data Extended Data Fig. 1
Statistical source data for Extended Data Fig. 1.
Source Data Extended Data Fig. 2
Statistical source data for Extended Data Fig. 2.
Rights and permissions
About this article
Cite this article
Ren, X., Zhou, J., Wang, E. et al. Ultrafast energy transfer between π-stacked aromatic rings upon inner-valence ionization. Nat. Chem. 14, 232–238 (2022). https://doi.org/10.1038/s41557-021-00838-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00838-4
This article is cited by
-
Prebiotic chemical origin of biomolecular complementarity
Communications Chemistry (2023)
-
Directly imaging excited state-resolved transient structures of water induced by valence and inner-shell ionisation
Nature Communications (2023)
-
Triple ionization and fragmentation of benzene trimers following ultrafast intermolecular Coulombic decay
Nature Communications (2022)
-
Ambient-light-induced intermolecular Coulombic decay in unbound pyridine monomers
Nature Chemistry (2022)