Abstract
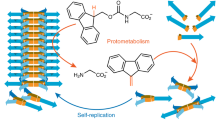
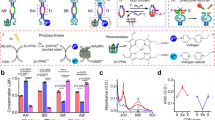
Establishing how life can emerge from inanimate matter is among the grand challenges of contemporary science. Chemical systems that capture life’s essential characteristics—replication, metabolism and compartmentalization—offer a route to understanding this momentous process. The synthesis of life, whether based on canonical biomolecules or fully synthetic molecules, requires the functional integration of these three characteristics. Here we show how a system of fully synthetic self-replicating molecules, on recruiting a cofactor, acquires the ability to transform thiols in its environment into disulfide precursors from which the molecules can replicate. The binding of replicator and cofactor enhances the activity of the latter in oxidizing thiols into disulfides through photoredox catalysis and thereby accelerates replication by increasing the availability of the disulfide precursors. This positive feedback marks the emergence of light-driven protometabolism in a system that bears no resemblance to canonical biochemistry and constitutes a major step towards the highly challenging aim of creating a new and completely synthetic form of life.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The UPLC data generated and analysed in this Article is included in its Supplementary Information in the form of integrated peak areas and exported traces of representative chromatograms. All other chromatograms are stored locally on their native format and are available on request. All other data generated or analysed during this study are included in this published Article and its Supplementary Information. Source data are provided with this paper.
References
Sutherland, J. D. Opinion: studies on the origin of life—the end of the beginning. Nat. Rev. Chem. 1, 1–8 (2017).
Szostak, J. W., Bartel, D. P. & Luisi, P. L. Synthesizing life. Nature 409, 387–390 (2001).
Benner, S. A., Ricardo, A. & Carrigan, M. A. Is there a common chemical model for life in the universe? Curr. Opin. Chem. Biol. 8, 672–689 (2004).
Gayon, J., Malaterre, C., Morange, M., Raulin-Cerceau, F. & Tirard, S. Defining life: conference proceedings. Orig. Life Evol. Biosph. 40, 119–120 (2010).
Ruiz-Mirazo, K., Briones, C. & De La Escosura, A. Prebiotic systems chemistry: new perspectives for the origins of life. Chem. Rev. 114, 285–366 (2014).
Ganti, T. in The Principles of Life 78–83 (Oxford Univ. Press, 2003).
Pascal, R., Pross, A. & Sutherland, J. D. Towards an evolutionary theory of the origin of life based on kinetics and thermodynamics. Open Biol. 3, 1–9 (2013).
Pross, A. The driving force for life’s emergence: kinetic and thermodynamic considerations. J. Theor. Biol. 220, 393–406 (2003).
Wagner, N., Pross, A. & Tannenbaum, E. Selection advantage of metabolic over non-metabolic replicators: a kinetic analysis. BioSystems 99, 126–129 (2010).
Pross, A. How can a chemical system act purposefully? Bridging between life and non-life. J. Phys. Org. Chem. 21, 724–730 (2008).
Hardy, M. D. et al. Self-reproducing catalyst drives repeated phospholipid synthesis and membrane growth. Proc. Natl Acad. Sci. USA 112, 8187–8192 (2015).
Gardner, P. M., Winzer, K. & Davis, B. G. Sugar synthesis in a protocellular model leads to a cell signalling response in bacteria. Nat. Chem. 1, 377–383 (2009).
Adamala, K. P. & Szostak, J. W. Nonenzymatic template-directed RNA synthesis inside model protocells. Science 1593, 283–289 (2003).
Ichihashi, N. et al. Darwinian evolution in a translation-coupled RNA replication system within a cell-like compartment. Nat. Commun. 4, 1–7 (2013).
Könnyu, B. & Czárán, T. The evolution of enzyme specificity in the metabolic replicator model of prebiotic evolution. PLoS One https://doi.org/10.1371/journal.pone.0020931 (2011).
Czárán, T., Könnyu, B. & Szathmáry, E. Metabolically coupled replicator systems: overview of an RNA-world model concept of prebiotic evolution on mineral surfaces. J. Theor. Biol. 381, 39–54 (2015).
Arsène, S., Ameta, S., Lehman, N., Griffiths, A. D. & Nghe, P. Coupled catabolism and anabolism in autocatalytic RNA sets. Nucleic Acids Res. 46, 9660–9666 (2018).
Kamioka, S., Ajami, D. & Rebek, J. Autocatalysis and organocatalysis with synthetic structures. Proc. Natl Acad. Sci. USA 107, 541–544 (2010).
Colomb-Delsuc, M., Mattia, E., Sadownik, J. W. & Otto, S. Exponential self-replication enabled through a fibre elongation/breakage mechanism. Nat. Commun. 6, 1–7 (2015).
Carnall, J. M. A. et al. Mechanosensitive self-replication driven by self-organization. Science 327, 1502–1507 (2010).
Caetano-Anollés, G., Kim, K. M. & Caetano-Anollés, D. The phylogenomic roots of modern biochemistry: origins of proteins, cofactors and protein biosynthesis. J. Mol. Evol. 74, 1–34 (2012).
Wachtershauser, G. Evolution of the first metabolic cycles. Proc. Natl Acad. Sci. USA 87, 200–204 (1990).
Croce, R. & Van Amerongen, H. Natural strategies for photosynthetic light harvesting. Nat. Chem. Biol. 10, 492–501 (2014).
Tankam, T., Poochampa, K., Vilaivan, T., Sukwattanasinitt, M. & Wacharasindhu, S. Organocatalytic visible light induced S–S bond formation for oxidative coupling of thiols to disulfides. Tetrahedron 72, 788–793 (2016).
Frederix, P. W. J. M. et al. Structural and spectroscopic properties of assemblies of self-replicating peptide macrocycles. ACS Nano 11, 7858–7868 (2017).
Würthner, F., Kaiser, T. E. & Saha-Möller, C. R. J-Aggregates: from serendipitous discovery to supramolecular engineering of functional dye materials. Angew. Chem. Int. Ed. 50, 3376–3410 (2011).
Bilski, P., Holt, R. N. & Chignell, C. F. Premicellar aggregates of Rose Bengal with cationic and zwitterionic surfactants. J. Photochem. Photobiol. A Chem 110, 67–74 (1997).
Krasnovsky, A. A. Photoluminescence of singlet oxygen in pigment solutions. Photochem. Photobiol. 29, 29–36 (1979).
Redmond, R. W. & Gamlin, J. N. A compilation of singlet oxygen yields from biologically relevant molecules. Photochem. Photobiol. 70, 391–475 (1999).
Komatsu, T., Wang, R. M., Zunszain, P. A., Curry, S. & Tsuchida, E. Photosensitized reduction of water to hydrogen using human serum albumin complexed with zinc-protoporphyrin IX. J. Am. Chem. Soc. 128, 16297–16301 (2006).
Lindig, B. A., Rodgers, M. A. J. & Schaaplc, A. P. Determination of the lifetime of singlet oxygen in D2O using 9,10-anthracenedipropionic acid, a water-soluble probe. J. Am. Chem. Soc. 102, 5590–5593 (1980).
Malakoutikhah, M. et al. Uncovering the selection criteria for the emergence of multi-building-block replicators from dynamic combinatorial libraries. J. Am. Chem. Soc. 135, 18406–18417 (2013).
Ottelé, J., Hussain, A. S., Mayer, C. & Otto, S. Chance emergence of catalytic activity and promiscuity in a self-replicator. Nat. Catal. https://doi.org/10.1038/s41929-020-0463-8 (2020).
Stuart, M. C. A., van de Pas, J. C. & Engberts, J. B. F. N. The use of nile red to monitor the aggregation behavior in ternary surfactant-water-organic solvent systems. J. Phys. Org. Chem. 18, 929–934 (2005).
Acknowledgements
We thank B. M. Matysiak for performing mass spectrometry measurements. This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Sklodowska-Curie grant agreement (no. 642192) and was supported by the ERC (AdG 741774), NWO (VICI grant) and the Dutch Ministry of Education, Culture and Science (Gravitation programme 024.001.035). K. L. acknowledges support from Simons Foundation (award no. 553330) and Marie Skłodowska-Curie grant (no. 786350).
Author information
Authors and Affiliations
Contributions
S.O., G.M.S. and K.L. conceived the experiments. G.M.S. performed the experiments related to rose bengal. K.L. performed the experiments related to porphyrins. W.R.B. performed the experiments related to IR luminescence of singlet oxygen. G.M.S, K.L. and S.O. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
Authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Absorption spectra of 3 recorded in reduced scattering conditions.
Visible absorption spectra of 3 (10 μM) in buffer (black), and in the presence of 13/14 (1.0 mM in 1; blue) and 16 (1.0 mM in 1; red). In order to show that the increased absorbance of 3 in presence of 16 is not related to scattering, this effect was minimized by recording these spectra in a different spectrophotometer (see Materials), using a wider slit (4 nm), a shorter light path (4.5 mm), and placing the cuvette immediately in front of the detector.
Extended Data Fig. 2 Non-irradiated controls for the emergence of 16 in presence of 2 and 3.
Evolution over time of the concentrations of 1 and 16 in non-irradiated libraries made from 1 and 2 (a) or 1 and 3 (b). The top halves of the graphs show the concentration of 1 (black squares) and 16 (red squares, in units of 1). The bottom halves of the graphs represent the oxidation (black circles) and replication (red circles) rates in the system, calculated by numerical differentiation of the curves in the top halves. Both libraries were prepared exactly as their equivalents in Fig. 4a,b, but they were kept in the dark.
Extended Data Fig. 3 Non-irradiated controls for the emergence of 16 in absence of photosensitizers.
Evolution over time of the concentrations of 1 and 16 in non-irradiated libraries prepared from 1, with no dyes added, in the same conditions as in Fig. 4a (left panel) or Fig. 4b (right panel). The top halves of the graphs show the concentration of 1 (black squares) and 16 (red squares, in units of 1). The bottom halves of the graphs represent the oxidation (black circles) and replication (red circles) rates in the system, calculated by numerical differentiation of the curves in the top halves.
Supplementary information
Supplementary Information
Supplementary Figs. 1–17, Tables 1 and 2.
Supplementary Data
Source Data for Supplementary Figs. 1–11,13–15 and Supplementary Table 2.
Source data
Source Data Fig. 2
Raw spectral data.
Source Data Fig. 3
Integrated peak areas of 1 in chromatograms, and calculation of oxidation rates from them.
Source Data Fig. 4
Integrated peak areas of 1 and 16, and calculation of concentrations and rates from them.
Source Data Extended Data Fig. 1
Raw spectral data.
Source Data Extended Data Fig. 2
Integrated peak areas of 1 and 16, and calculation of concentrations and rates from them.
Source Data Extended Data Fig. 3
Integrated peak areas of 1 and 16, and calculation of concentrations and rates from them.
Rights and permissions
About this article
Cite this article
Monreal Santiago, G., Liu, K., Browne, W.R. et al. Emergence of light-driven protometabolism on recruitment of a photocatalytic cofactor by a self-replicator. Nat. Chem. 12, 603–607 (2020). https://doi.org/10.1038/s41557-020-0494-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-020-0494-4
This article is cited by
-
Light-driven eco-evolutionary dynamics in a synthetic replicator system
Nature Chemistry (2024)
-
Photoswitchable gating of non-equilibrium enzymatic feedback in chemically communicating polymersome nanoreactors
Nature Chemistry (2023)
-
The rise of intelligent matter
Nature (2021)
-
Combining catalysis and replication
Nature Chemistry (2020)
-
From self-replication to replicator systems en route to de novo life
Nature Reviews Chemistry (2020)