Abstract
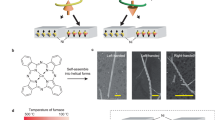
The generation of molecular chirality in the absence of any molecular chiral inductor is challenging and of fundamental interest for developing a better understanding of homochirality. Here, we show the manipulation of molecular chirality through control of the handedness of helical metal nanostructures (referred to as nanohelices) that are produced by glancing angle deposition onto a substrate that rotates in either a clockwise or counterclockwise direction. A prochiral molecule, 2-anthracenecarboxylic acid, is stereoselectively adsorbed on the metal nanohelices as enantiomorphous anti-head-to-head dimers. The dimers show either Si–Si or Re–Re facial stacking depending on the handedness of the nanohelices, which results in a specific enantiopreference during their photoinduced cyclodimerization: a left-handed nanohelix leads to the formation of (+)-cyclodimers, whereas a right-handed one gives (–)-cyclodimers. Density functional theory calculations, in good agreement with the experimental results, point to the enantioselectivity mainly arising from the selective spatial matching of either Si–Si or Re–Re facial stacking at the helical surface; it may also be influenced by chiroplasmonic effects.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Data supporting the findings of this study are available within this paper and its Supplementary Information, and are available from the corresponding author upon reasonable request.
References
Baiker, A. Crucial aspects in the design of chirally modified noble metal catalysts for asymmetric hydrogenation of activated ketones. Chem. Soc. Rev. 44, 7449–7464 (2015).
Vetica, F., Chauhan, P., Dochain, S. & Enders, D. Asymmetric organocatalytic methods for the synthesis of tetrahydropyrans and their application in total synthesis. Chem. Soc. Rev. 46, 1661–1674 (2017).
Xie, J. H., Zhu, S. F. & Zhou, Q. L. Transition metal-catalyzed enantioselective hydrogenation of enamines and imines. Chem. Rev. 111, 1713–1760 (2011).
Boersma, A. J., Megens, R. P., Feringa, B. L. & Roelfes, G. DNA-based asymmetric catalysis. Chem. Soc. Rev. 39, 2083–2092 (2010).
Yamamoto, T., Akai, Y. & Suginome, M. Chiral palladacycle catalysts generated on a single-handed helical polymer skeleton for asymmetric arylative ring opening of 1,4-epoxy-1,4-dihydronaphthalene. Angew. Chem. Int. Ed. 53, 12785–12788 (2014).
Le Bailly, B. A. F., Byrne, L. & Clayden, J. Refoldable foldamers: global conformational switching by deletion or insertion of a single hydrogen bond. Angew. Chem. Int. Ed. 55, 2132–2136 (2016).
Omosun, T. O. et al. Catalytic diversity in self-propagating peptide assemblies. Nat. Chem. 9, 805–809 (2017).
Iida, H., Iwahana, S., Mizoguchi, T. & Yashima, E. Main-chain optically active riboflavin polymer for asymmetric catalysis and its vapochromic behavior. J. Am. Chem. Soc. 134, 15103–15113 (2012).
Huck, N. P. M., Jager, W. F., de Lange, B. & Feringa, B. L. Dynamic control and amplification of molecular chirality by circular polarized light. Science 273, 1686–1688 (1996).
Feringa, B. L. & Van Delden, R. A. Absolute asymmetric synthesis: the origin, control, and amplification of chirality. Angew. Chem. Int. Ed. 38, 3419–3438 (1999).
Hazen, R. M. & Sholl, D. S. Chiral selection on inorganic crystalline surfaces. Nat. Mater. 2, 367–374 (2003).
Schwab, G. M., Rost, F. & Rudolph, L. Optisch asymmetrische Katalyse an Quarzkristallen. Kolloid-Zeitschrift 68, 157–165 (1934).
Schwab, G. M. & Rudolph, L. Katalytische Spaltung von Racematen durch Rechts- und Linksquarz. Naturwissenschaften 20, 363–364 (1932).
Horvath, J. D. & Gellman, A. J. Enantiospecific desorption of R- and S-propylene oxide from a chiral Cu(643) surface. J. Am. Chem. Soc. 123, 7953–7954 (2001).
Gellman, A. J. Chiral Surfaces: accomplishments and challenges. ACS Nano 4, 5–10 (2010).
Pour, S. O. et al. Through-space transfer of chiral information mediated by a plasmonic nanomaterial. Nat. Chem. 7, 591–596 (2015).
Morrow, S. M., Bissette, A. J. & Fletcher, S. P. Transmission of chirality through space and across length scales. Nat. Nanotechnol. 12, 410–419 (2017).
Robbie, K., Brett, M. J. & Lakhtakia, A. Chiral sculptured thin films. Nature 384, 616 (1996).
Robbie, K., Broer, D. J. & Brett, M. J. Chiral nematic order in liquid crystals imposed by an engineered inorganic nanostructure. Nature 399, 764–766 (1999).
Petit-Garrido, N., Claret, J., Ignes-Mullol, J. & Sagues, F. Stirring competes with chemical induction in chiral selection of soft matter aggregates. Nat. Commun. 3, 1001 (2012).
Ribo, J. M., Crusats, J., Sagues, F., Claret, J. & Rubires, R. Chiral sign induction by vortices during the formation of mesophases in stirred solutions. Science 292, 2063–2066 (2001).
Mark, A. G., Gibbs, J. G., Lee, T. C. & Fischer, P. Hybrid nanocolloids with programmed three-dimensional shape and material composition. Nat. Mater. 12, 802–807 (2013).
Larsen, G. K., He, Y. Z., Wang, J. & Zhao, Y. P. Scalable fabrication of composite Ti/Ag plasmonic helices: controlling morphology and optical activity by tailoring material properties. Adv. Opt. Mater. 2, 245–249 (2014).
Caridad, J. M. et al. Effective wavelength scaling of and damping in plasmonic helical antennae. ACS Photonics 2, 675–679 (2015).
Hawkeye, M. M. & Brett, M. J. Glancing angle deposition: fabrication, properties, and applications of micro- and nanostructured thin films. J. Vac. Sci. Technol. A 25, 1317–1335 (2007).
Jen, Y. J., Huang, Y. J., Liu, W. C. & Lin, Y. W. Densely packed aluminum-silver nanohelices as an ultra-thin perfect light absorber. Sci. Rep. 7, 39791 (2017).
Sobhkhiz, N. & Moshaii, A. Broadband improvement of light absorption properties of α-Fe2O3 thin-film by silver helical nanostructures. Plasmonics 10, 1243–1253 (2015).
Deng, J. H., Fu, J. X., Ng, J. & Huang, Z. F. Tailorable chiroptical activity of metallic nanospiral arrays. Nanoscale 8, 4504–4510 (2016).
Liu, J. J., Yang, L. & Huang, Z. F. Chiroptically active plasmonic nanoparticles having hidden helicity and reversible aqueous solvent effect on chiroptical activity. Small 12, 5902–5909 (2016).
Liu, J. J., Yang, L., Zhang, H., Wang, J. F. & Huang, Z. F. Ultraviolet–visible chiroptical activity of aluminum nanostructures. Small 13, 1701112 (2017).
Gibbs, J. G. et al. Nanohelices by shadow growth. Nanoscale 6, 9457–9466 (2014).
Jeong, H. H., Mark, A. G. & Fischer, P. Magnesium plasmonics for UV applications and chiral sensing. Chem. Commun. 52, 12179–12182 (2016).
Bai, F. et al. Two chiroptical modes of silver nanospirals. Nanotechnology 27, 115703 (2016).
Ke, C. F. et al. Catalytic enantiodifferentiating photocyclodimerization of 2-anthracenecarboxylic acid mediated by a non-sensitizing chiral metallosupramolecular host. Angew. Chem. Int. Ed. 48, 6675–6677 (2009).
Yang, C. & Inoue, Y. Supramolecular photochirogenesis. Chem. Soc. Rev. 43, 4123–4143 (2014).
Kawanami, Y. et al. Supramolecular photochirogenesis with a higher-order complex: highly accelerated exclusively head-to-head photocyclodimerization of 2-anthracenecarboxylic acid via 2:2 complexation with prolinol. J. Am. Chem. Soc. 138, 12187–12201 (2016).
Wang, Q. et al. Wavelength-controlled supramolecular photocyclodimerization of anthracenecarboxylate mediated by γ-cyclodextrins. Chem. Commun. 47, 6849–6851 (2011).
Yang, C., Nakamura, A., Wada, T. & Inoue, Y. Enantiodifferentiating photocyclodimerization of 2-anthracenecarboxylic acid mediated by γ-cyclodextrins with a flexible or rigid cap. Org. Lett. 8, 3005–3008 (2006).
Dawn, A., Fujita, N., Haraguchi, S., Sada, K. & Shinkai, S. An organogel system can control the stereochemical course of anthracene photodimerization. Chem. Commun. 16, 2100–2102 (2009).
Ishida, Y. et al. Two-component liquid crystals as chiral reaction media: highly enantioselective photodimerization of an anthracene derivative driven by the ordered microenvironment. Angew. Chem. Int. Ed. 47, 8241–8245 (2008).
Yang, C. et al. A remarkable stereoselectivity switching upon solid-state versus solution-phase enantiodifferentiating photocyclodimerization of 2-anthracenecarboxylic acid mediated by native and 3,6-anhydro-γ-cyclodextrins. Tetrahedron Lett. 48, 4357–4360 (2007).
Schaferling, M., Dregely, D., Hentschel, M. & Giessen, H. Tailoring enhanced optical chirality: design principles for chiral plasmonic nanostructures. Phys. Rev. X 2, 031010 (2012).
Birks, J. B. Photophysics of Aromatic Molecules (Wiley, 1970).
Shoeib, T., Rodriquez, C. F., Siu, K. W. M. & Hopkinson, A. C. A comparison of copper(i) and silver(i) complexes of glycine, diglycine and triglycine. Phys. Chem. Chem. Phys. 3, 853–861 (2001).
Herron, J. A., Scaranto, J., Ferrin, P., Li, S. & Mavrikakis, M. Trends in formic acid decomposition on model transition metal surfaces: a density functional theory study. ACS Catal. 4, 4434–4445 (2014).
Wakai, A., Fukasawa, H., Yang, C., Mori, T. & Inoue, Y. Theoretical and experimental investigations of circular dichroism and absolute configuration determination of chiral anthracene photodimers. J. Am. Chem. Soc. 134, 4990–4997 (2012).
Nakamura, A. & Inoue, Y. Supramolecular catalysis of the enantiodifferentiating [4 + 4] photocyclodimerization of 2-anthracenecarboxylate by γ-cyclodextrin. J. Am. Chem. Soc. 125, 966–972 (2003).
Ernst, K. H. Molecular chirality in surface science. Surf. Sci. 613, 1–5 (2013).
Persson, I. & Nilsson, K. B. Coordination chemistry of the solvated silver(i) ion in the oxygen donor solvents water, dimethyl sulfoxide, and N,N′-dimethylpropyleneurea. Inorg. Chem. 45, 7428–7434 (2006).
Tang, Y. Q. & Cohen, A. E. Optical chirality and its interaction with matter. Phys. Rev. Lett. 104, 163901 (2010).
Tang, Y. Q. & Cohen, A. E. Enhanced enantioselectivity in excitation of chiral molecules by superchiral light. Science 332, 333–336 (2011).
Inoue, Y. Asymmetric photochemical reactions in solution. Chem. Rev. 92, 741–770 (1992).
Acknowledgements
The authors thank U. Mirsaidov and P. Nandi for technical support with HRTEM (National University of Singapore), G. Fu (Xiamen University, China) and Y. Yang (Soochow University, China) for discussions, H. Zhang (Chinese University of Hong Kong) for technical support with TEM, W. Wu (IAM, HKBU) for technical support with XRD and XPS, and financial support from NSFC/91856127/21871194/21971169/21473149/21572142/21372165, the National Key Research and Development Program of China (No. 2017YFA0505903), GRF/12200118, FRG2/17-18/058 (HKBU), FRG2/16-17/013 (HKBU) and HKBU8/CRF/11E (GLAD). G.J.X. and Y.G.W. acknowledge financial support from SUSTech and computational resource support from the Center for Computational Science and Engineering (SUSTech). C.Y. thanks the Comprehensive Training Platform of the Specialized Laboratory, College of Chemistry for financial support and P. Wu of the Analytical & Testing Center, Sichuan University, for analytical support.
Author information
Authors and Affiliations
Contributions
Z.F.H. and C.Y. conceived and designed the study; J.J.L. performed GLAD and material characterization; X.Q.W. and W.H.W performed photocyclodimerization and product analysis; G.J.X. and Y.G.W. performed theoretical simulations of AC photocyclodimerization; J.H.D. and P.S. performed the numerical simulation of optical chirality; Z.F.H., C.Y., Y.G.W. and J.J.C. composed the manuscript. All the authors reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Characterization data, computational data, Supplementary Tables 1–3, Figs. 1–23 and refs. 1–9.
Rights and permissions
About this article
Cite this article
Wei, X., Liu, J., Xia, GJ. et al. Enantioselective photoinduced cyclodimerization of a prochiral anthracene derivative adsorbed on helical metal nanostructures. Nat. Chem. 12, 551–559 (2020). https://doi.org/10.1038/s41557-020-0453-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-020-0453-0
This article is cited by
-
Emerging trends in chiral inorganic nanomaterials for enantioselective catalysis
Nature Communications (2024)
-
Controlling the broadband enhanced light chirality with L-shaped dielectric metamaterials
Nature Communications (2024)
-
Regulation of biological processes by intrinsically chiral engineered materials
Nature Reviews Materials (2023)
-
Bioinspired chiral inorganic nanomaterials
Nature Reviews Bioengineering (2023)
-
A new sensitization strategy for achieving organic RTP in aqueous solution: tunable RTP and UC emission in supramolecular TTA-UC systems
Science China Chemistry (2023)