Abstract
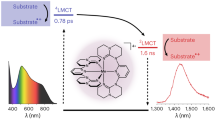
Advances in chemical control of the photophysical properties of transition-metal complexes are revolutionizing a wide range of technologies, particularly photocatalysis and light-emitting diodes, but they rely heavily on molecules containing precious metals such as ruthenium and iridium. Although the application of earth-abundant ‘early’ transition metals in photosensitizers is clearly advantageous, a detailed understanding of excited states with ligand-to-metal charge transfer (LMCT) character is paramount to account for their distinct electron configurations. Here we report an air- and moisture-stable, visible light-absorbing Zr(iv) photosensitizer, Zr(MesPDPPh)2, where [MesPDPPh]2− is the doubly deprotonated form of [2,6-bis(5-(2,4,6-trimethylphenyl)-3-phenyl-1H-pyrrol-2-yl)pyridine]. This molecule has an exceptionally long-lived triplet LMCT excited state (τ = 350 μs), featuring highly efficient photoluminescence emission (Ф = 0.45) due to thermally activated delayed fluorescence emanating from the higher-lying singlet configuration with significant LMCT contributions. Zr(MesPDPPh)2 engages in numerous photoredox catalytic processes and triplet energy transfer. Our investigation provides a blueprint for future photosensitizer development featuring early transition metals and excited states with significant LMCT contributions.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Crystallographic data for the structure reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition no. CCDC 1922700 (Zr(MesPDPPh)2). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All remaining data are available in the main text or the Supplementary Information.
References
Nazeeruddin, M. K., Baranoff, E. & Grätzel, M. Dye-sensitized solar cells: a brief overview. Sol. Energy 85, 1172–1178 (2011).
Hagfeldt, A., Boschloo, G., Sun, L., Kloo, L. & Pettersson, H. Dye-sensitized solar cells. Chem. Rev. 110, 6595–6663 (2010).
Ashford, D. L. et al. Molecular chromophore–catalyst assemblies for solar fuel applications. Chem. Rev. 115, 13006–13049 (2015).
Xuan, J. & Xiao, W.-J. Visible-light photoredox catalysis. Angew. Chem. Int. Ed. 51, 6828–6838 (2012).
Tucker, J. W. & Stephenson, C. R. J. Shining light on photoredox catalysis: theory and synthetic applications. J. Org. Chem. 77, 1617–1622 (2012).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Monro, S. et al. Transition metal complexes and photodynamic therapy from a tumor-centered approach: challenges, opportunities, and highlights from the development of TLD1433. Chem. Rev. 119, 797–828 (2019).
Yersin, H., Rausch, A. F., Czerwieniec, R., Hofbeck, T. & Fischer, T. The triplet state of organo-transition metal compounds. Triplet harvesting and singlet harvesting for efficient OLEDs. Coord. Chem. Rev. 255, 2622–2652 (2011).
Xu, H. et al. Recent progress in metal–organic complexes for optoelectronic applications. Chem. Soc. Rev. 43, 3259–3302 (2014).
Dias, F. B., Penfold, T. J., Berberan-Santos, M. N. & Monkman, A. P. Photophysics of thermally activated delayed fluorescence in organic molecules. Methods Appl. Fluoresc. 5, 012001 (2017).
Li, G., Zhu, Z.-Q., Chen, Q. & Li, J. Metal complex based delayed fluorescence materials. Org. Electron. 69, 135–152 (2019).
Uoyama, H., Goushi, K., Shizu, K., Nomura, H. & Adachi, C. Highly efficient organic light-emitting diodes from delayed fluorescence. Nature 492, 234–238 (2012).
Penfold, T. J., Dias, F. B. & Monkman, A. P. The theory of thermally activated delayed fluorescence for organic light emitting diodes. Chem. Commun. 54, 3926–3935 (2018).
Kirchhoff, J. R. et al. Temperature dependence of luminescence from Cu(NN)2 + systems in fluid solution. Evidence for the participation of two excited states. Inorg. Chem. 22, 2380–2384 (1983).
Peltier, J. L. et al. Eliminating nonradiative decay in Cu(i) emitters: 99% quantum efficiency and microsecond lifetime. Science 363, 601–606 (2019).
Czerwieniec, R., Leitl, M. J., Homeier, H. H. H. & Yersin, H. Cu(i) complexes—thermally activated delayed fluorescence. Photophysical approach and material design. Coord. Chem. Rev. 325, 2–28 (2016).
Wenger, O. S. Photoactive complexes with earth-abundant metals. J. Am. Chem. Soc. 140, 13522–13533 (2018).
Harlang, T. C. B. et al. Iron sensitizer converts light to electrons with 92% yield. Nat. Chem. 7, 883–889 (2015).
Chábera, P. et al. A low-spin Fe(iii) complex with 100 ps ligand-to-metal charge transfer photoluminescence. Nature 543, 695–699 (2017).
Kjær, K. S. et al. Luminescence and reactivity of a charge-transfer excited iron complex with nanosecond lifetime. Science 363, 249–253 (2019).
Lazorski, M. S. & Castellano, F. N. Advances in the light conversion properties of Cu(i)-based photosensitizers. Polyhedron 82, 57–70 (2014).
Zhang, Y., Schulz, M., Wächtler, M., Karnahl, M. & Dietzek, B. Heteroleptic diimine–diphosphine Cu(i) complexes as an alternative towards noble-metal based photosensitizers: design strategies, photophysical properties and perspective applications. Coord. Chem. Rev. 356, 127–146 (2018).
Arias-Rotondo, D. M. & McCusker, J. K. The photophysics of photoredox catalysis: a roadmap for catalyst design. Chem. Soc. Rev. 45, 5803–5820 (2016).
Pfennig, B. W., Thompson, M. E. & Bocarsly, A. B. A new class of room temperature luminescent organometallic complexes: luminescence and photophysical properties of permethylscandocene chloride in fluid solution. J. Am. Chem. Soc. 111, 8947–8948 (1989).
Paulson, S., Sullivan, B. P. & Caspar, J. V. Luminescent ligand-to-metal charge-transfer excited states based on pentamethylcyclopentadienyl complexes of tantalum. J. Am. Chem. Soc. 114, 6905–6906 (1992).
Heinselman, K. S. & Hopkins, M. D. Luminescence properties of d 0 metal–imido compounds. J. Am. Chem. Soc. 117, 12340–12341 (1995).
Loukova, G. V., Huhn, W., Vasiliev, V. P. & Smirnov, V. A. Ligand-to-metal charge transfer excited states with unprecedented luminescence yield in fluid solution. J. Phys. Chem. A 111, 4117–4121 (2007).
Romain, C. et al. Redox and luminescent properties of robust and air-stable N-heterocyclic carbene group 4 metal complexes. Inorg. Chem. 53, 7371–7376 (2014).
Loukova, G. V. & Smirnov, V. A. Phosphorescent ligand-to-metal charge-transfer excited states in the group IVB metallocene triad. Chem. Phys. Lett. 329, 437–442 (2000).
Gazi, S. et al. Selective photocatalytic C–C bond cleavage under ambient conditions with earth abundant vanadium complexes. Chem. Sci. 6, 7130–7142 (2015).
Zhang, Y., Petersen, J. L. & Milsmann, C. A luminescent zirconium(iv) complex as a molecular photosensitizer for visible light photoredox catalysis. J. Am. Chem. Soc. 138, 13115–13118 (2016).
Zhang, Y., Lee, T. S., Petersen, J. L. & Milsmann, C. A zirconium photosensitizer with a long-lived excited state: mechanistic insight into photo-induced single electron transfer. J. Am. Chem. Soc. 140, 5934–5947 (2018).
Zhang, Y., Petersen, J. L. & Milsmann, C. Photochemical C–C bond formation in luminescent zirconium complexes with CNN pincer ligands. Organometallics 37, 4488–4499 (2018).
Zhang, Y., Akhmedov, N. G., Petersen, J. L. & Milsmann, C. Photoluminescence of seven-coordinate zirconium and hafnium complexes with 2,2′-pyridylpyrrolide ligands. Chem. Eur. J. 25, 3042–3052 (2019).
Chan, K.-T. et al. Strongly luminescent tungsten emitters with emission quantum yields of up to 84%: TADF and high-efficiency molecular tungsten OLEDs. Angew. Chem. Int. Ed. 58, 14896–14900 (2019).
Zhang, L. L.-M. et al. Core-dependent properties of copper nanoclusters: valence-pure nanoclusters as NIR TADF emitters and mixed-valence ones as semiconductors. Chem. Sci. 10, 10122–10128 (2019).
Hakey, B. M., Darmon, J. M., Zhang, Y., Petersen, L. & Milsmann, C. Synthesis and electronic structure of neutral square-planar high-spin iron(ii) complexes supported by a dianionic pincer ligand. Inorg. Chem. 58, 1252–1266 (2019).
Vernitskaya, T. V. & Efimov, O. N. Polypyrrole: a conducting polymer; its synthesis, properties and applications. Russ. Chem. Rev. 66, 443–457 (1997).
Herr, P., Glaser, F., Büldt, L. A., Larsen, C. B. & Wenger, O. S. Long-lived, strongly emissive, and highly reducing excited states in Mo(0) complexes with chelating isocyanides. J. Am. Chem. Soc. 141, 14394–14402 (2019).
Lees, A. J. The luminescence rigidochromic effect exhibited by organometallic complexes: rationale and applications. Comments Inorg. Chem. 17, 319–346 (1995).
Brown, A. M., McCusker, C. E. & McCusker, J. K. Spectroelectrochemical identification of charge-transfer excited states in transition metal-based polypyridyl complexes. Dalton Trans. 43, 17635–17646 (2014).
Hammond, G. S. et al. Mechanisms of photochemical reactions in solution. XXII. Photochemical cis–trans isomerization. J. Am. Chem. Soc. 86, 3197–3217 (1964).
Larsen, C. B. & Wenger, O. S. Photoredox catalysis with metal complexes made from earth-abundant elements. Chem. Eur. J. 24, 2039–2058 (2018).
Hockin, B. M., Li, C., Robertson, N. & Zysman-Colman, E. Photoredox catalysts based on earth-abundant metal complexes. Catal. Sci. Technol. 9, 889–915 (2019).
Kalyanasundaram, K. Photophysics, photochemistry and solar energy conversion with tris(bipyridyl)ruthenium(ii) and its analogues. Coord. Chem. Rev. 46, 159–244 (1982).
Connelly, N. G. & Geiger, W. E. Chemical redox agents for organometallic chemistry. Chem. Rev. 96, 877–910 (1996).
Acknowledgements
C.M., Y.Z. and D.C.L. acknowledge West Virginia University and the National Science Foundation (CHE-1752738) for financial support. This work used X-ray crystallography (CHE-1336071) and NMR (CHE-1228336) equipment funded by the National Science Foundation. The WVU High Performance Computing facilities are funded by the National Science Foundation EPSCoR Research Infrastructure Improvement Cooperative Agreement no. 1003907, the state of West Virginia (WVEPSCoR via the Higher Education Policy Commission), the WVU Research Corporation and faculty investments. The temperature-dependent static and time-resolved photoluminescence experiments performed at NC State (F.N.C. and J.M.F.) were supported by the US Department of Energy, Office of Science, Office of Basic Energy Sciences, under award no. DE-SC0011979. G.D.S and T.L. acknowledge the Division of Chemical Sciences, Geosciences and Biosciences, Office of Basic Energy Sciences, of the US Department of Energy through grant no. DE-SC0015429.
Author information
Authors and Affiliations
Contributions
Y.Z. synthesized and characterized the compound, performed electrochemical measurements, collected steady-state absorption and emission spectra, conducted the majority of photoredox catalytic reactions, and obtained and analysed all computational data. T.S.L. collected and analysed the TA spectroscopic data. J.M.F. conducted the temperature-dependent emission studies and analysed the corresponding data. D.C.L. performed redox titrations and additional photoredox catalytic reactions. J.L.P. determined the crystal structure. G.D.S., F.N.C. and C.M. directed the project and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, characterization and computational results. Figs. 1–39, Tables 1–4 and refs. 1–29.
XYZ coordinates
Cartesian coordinates for optimized structures from DFT calculations.
Crystallographic data
Crystallographic data for Zr(MesPDPPh)2; CCDC reference 1922700.
Rights and permissions
About this article
Cite this article
Zhang, Y., Lee, T.S., Favale, J.M. et al. Delayed fluorescence from a zirconium(iv) photosensitizer with ligand-to-metal charge-transfer excited states. Nat. Chem. 12, 345–352 (2020). https://doi.org/10.1038/s41557-020-0430-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-020-0430-7
This article is cited by
-
Triplet–triplet annihilation photon upconversion-mediated photochemical reactions
Nature Reviews Chemistry (2024)
-
Photoredox-active Cr(0) luminophores featuring photophysical properties competitive with Ru(II) and Os(II) complexes
Nature Chemistry (2023)
-
Spin–vibronic coherence drives singlet–triplet conversion
Nature (2023)
-
The ligand-to-metal charge transfer excited state of [Re(dmpe)3]2+
Photosynthesis Research (2022)
-
Manganese(i) complexes with metal-to-ligand charge transfer luminescence and photoreactivity
Nature Chemistry (2021)