Abstract
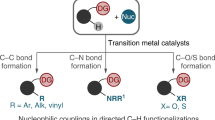
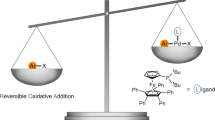
Transition metal-catalysed C–H functionalization and decarboxylative coupling are two of the most notable synthetic strategies developed in the past 30 years. Here, we connect these two reaction pathways using bases and a simple Pd-based catalyst system to promote a para-selective C–H functionalization reaction from benzylic electrophiles. Experimental and computational mechanistic studies suggest a pathway that involves an uncommon Pd-catalysed dearomatization of the benzyl moiety followed by a base-enabled rearomatization through a formal 1,5-hydrogen migration. This reaction complements ‘C–H activation’ strategies that convert inert C–H bonds into C–metal bonds prior to C–C bond formation. Instead, this reaction exploits an inverted sequence and promotes C–C bond formation prior to deprotonation. These studies provide an opportunity to develop general para-selective C–H functionalization reactions from benzylic electrophiles and show how new reactive modalities may be accessed with careful control of the reaction conditions.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the paper and its Supplementary Information files.
References
Johansson Seechurn, C. C. C., Kitching, M. O., Colacot, T. J. & Snieckus, V. Palladium-catalyzed cross-coupling: a historical contextual perspective to the 2010 Nobel prize. Angew. Chem. Int. Ed. 51, 5062–5085 (2012).
Labinger, J. A. & Bercaw, J. E. Understanding and exploiting C–H bond activation. Nature 417, 507–514 (2002).
Hartwig, J. F. Evolution of C–H bond functionalization from methane to methodology. J. Am. Chem. Soc. 138, 2–24 (2016).
Gensch, T., Hopkinson, M. N., Glorius, F. & Wencel-Delord, J. Mild metal-catalyzed C–H activation: examples and concepts. Chem. Soc. Rev. 45, 2900–2936 (2016).
Yamaguchi, J., Yamaguchi, A. D. & Itami, K. C–H bond functionalization: emerging synthetic tools for natural products and pharmaceuticals. Angew. Chem. Int. Ed. 51, 8960–9009 (2012).
Engle, K. M., Mei, T.-S., Wasa, M. & Yu, J.-Q. Weak coordination as a powerful means for developing broadly useful C–H functionalization reactions. Acc. Chem. Res. 45, 788–802 (2012).
Neufeldt, S. R. & Sanford, M. S. Controlling site selectivity in palladium-catalyzed C–H bond functionalization. Acc. Chem. Res. 45, 936–946 (2012).
Zhang, F. & Spring, D. R. Arene C–H functionalisation using a removable/modifiable or a traceless directing group strategy. Chem. Soc. Rev. 43, 6906–6919 (2014).
Yang, J. Transition metal catalyzed meta-C–H functionalization of aromatic compounds. Org. Biomol. Chem. 13, 1930–1941 (2015).
Dey, A., Agasti, S. & Maiti, D. Palladium catalysed meta-C–H functionalization reactions. Org. Biomol. Chem. 14, 5440–5453 (2016).
Ciana, C.-L., Phipps, R. J., Brandt, J. R., Meyer, F.-M. & Gaunt, M. J. A highly para-selective copper(ii)-catalyzed direct arylation of aniline and phenol derivatives. Angew. Chem. Int. Ed. 50, 458–462 (2011).
Wang, X., Leow, D. & Yu, J.-Q. Pd(ii)-catalyzed para-selective C–H arylation of monosubstituted arenes. J. Am. Chem. Soc. 133, 13864–13867 (2011).
Ball, L. T., Lloyd-Jones, G. C. & Russell, C. A. Gold-catalyzed direct arylation. Science 337, 1644–1648 (2012).
Wu, Z. et al. Palladium-catalyzed para-selective arylation of phenols with aryl iodides in water. Chem. Commun. 49, 7653–7655 (2013).
Yu, Z. et al. Highly site-selective direct C–H bond functionalization of phenols with α-aryl-α-diazoacetates and diazooxindoles via gold catalysis. J. Am. Chem. Soc. 136, 6904–6907 (2014).
Berzina, B., Sokolovs, I. & Suna, E. Copper-catalyzed para-selective C–H amination of electron-rich arenes. ACS Catal. 5, 7008–7014 (2015).
Marchetti, L., Kantak, A., Davis, R. & DeBoef, B. Regioselective gold-catalyzed oxidative C–N bond formation. Org. Lett. 17, 358–361 (2015).
Xu, H., Shang, M., Dai, H.-X. & Yu, J.-Q. Ligand-controlled para-selective C–H arylation of monosubstituted arenes. Org. Lett. 17, 3830–3833 (2015).
Yang, Z., Qiu, F.-C., Gao, J., Li, Z.-W. & Guan, B.-T. Palladium-catalyzed oxidative arylation of tertiary benzamides: para-selectivity of monosubstituted arenes. Org. Lett. 17, 4316–4319 (2015).
Sokolovs, I. & Suna, E. Para-selective Cu-catalyzed C–H aryloxylation of electron-rich arenes and heteroarenes. J. Org. Chem. 81, 371–379 (2016).
Ma, B. et al. Highly para-selective C−H alkylation of benzene derivatives with 2,2,2-trifluoroethyl α-aryl-α-diazoesters. Angew. Chem. Int. Ed. 56, 2749–2753 (2017).
Luan, Y.-X. et al. Amide-ligand-controlled highly para-selective arylation of monosubstituted simple arenes with arylboronic acids. J. Am. Chem. Soc. 139, 1786–1789 (2017).
Bag, S. et al. Remote para-C–H functionalization of arenes by a D-shaped biphenyl template-based assembly. J. Am. Chem. Soc. 137, 11888–11891 (2015).
Patra, T. et al. Palladium-catalyzed directed para C−H functionalization of phenols. Angew. Chem. Int. Ed. 55, 7751–7755 (2016).
Maji, A. et al. Experimental and computational exploration of para-selective silylation with a hydrogen-bonded template. Angew. Chem. Int. Ed. 56, 14903–14907 (2017).
Li, M. et al. Remote para-C–H acetoxylation of electron-deficient arenes. Org. Lett. 21, 540–544 (2019).
Nakao, Y., Yamada, Y., Kashihara, N. & Hiyama, T. Selective C-4 alkylation of pyridine by nickel/Lewis acid catalysis. J. Am. Chem. Soc. 132, 13666–13668 (2010).
Tsai, C.-C. et al. Bimetallic nickel aluminum mediated para-selective alkenylation of pyridine: direct observation of η2,η1-pyridine Ni(0)−Al(iii) intermediates prior to C−H bond activation. J. Am. Chem. Soc. 132, 11887–11889 (2010).
Saito, Y., Segawa, Y. & Itami, K. Para-C–H borylation of benzene derivatives by a bulky iridium catalyst. J. Am. Chem. Soc. 137, 5193–5198 (2015).
Okumura, S. et al. Para-selective alkylation of benzamides and aromatic ketones by cooperative nickel/aluminum catalysis. J. Am. Chem. Soc. 138, 14699–14704 (2016).
Berger, F. et al. Site-selective and versatile aromatic C−H functionalization by thianthrenation. Nature 567, 223–228 (2019).
Yang, M.-H., Hunt, J. R., Sharifi, N. & Altman, R. A. Palladium catalysis enables benzylation of α,α-difluoroketone enolates. Angew. Chem. Int. Ed. 55, 9080–9083 (2016).
Recio, I. I. I. A., Heinzman, J. D. & Tunge, J. A. Decarboxylative benzylation and arylation of nitriles. Chem. Commun. 48, 142–144 (2012).
Mendis, S. N. & Tunge, J. A. Decarboxylative dearomatization and mono-α-arylation of ketones. Chem. Commun. 52, 7695–7698 (2016).
Jaeger, C. W. & Kornblum, N. New type of substitution at a saturated carbon atom. J. Am. Chem. Soc. 94, 2545–2547 (1972).
Bao, M., Nakamura, H. & Yamamoto, Y. Facile allylative dearomatization catalyzed by palladium. J. Am. Chem. Soc. 123, 759–760 (2001).
Lu, S., Xu, Z., Bao, M. & Yamamoto, Y. Carbocycle synthesis through facile and efficient palladium-catalyzed allylative de-aromatization of naphthalene and phenanthrene allyl chlorides. Angew. Chem. Int. Ed. 47, 4366–4369 (2008).
Peng, B., Zhang, S., Yu, X., Feng, X. & Bao, M. Nucleophilic dearomatization of chloromethyl naphthalene derivatives via η3-benzylpalladium intermediates: a new strategy for catalytic dearomatization. Org. Lett. 13, 5402–5405 (2011).
Ueno, S., Komiya, S., Tanaka, T. & Kuwano, R. Intramolecular SN′-type aromatic substitution of benzylic carbonates at their para-position. Org. Lett. 14, 338–341 (2012).
Zhang, S., Wang, Y., Feng, X. & Bao, M. Palladium-catalyzed amination of chloromethylnaphthalene and chloromethylanthracene derivatives with various amines. J. Am. Chem. Soc. 134, 5492–5495 (2012).
Zhang, S., Yu, X., Feng, X., Yamamoto, Y. & Bao, M. Palladium-catalyzed regioselective allylation of five-membered heteroarenes with allyltributylstannane. Chem. Commun. 51, 3842–3845 (2015).
Arlow, S. I. & Hartwig, J. F. Synthesis, characterization, and reactivity of palladium fluoroenolate complexes. J. Am. Chem. Soc. 139, 16088–16091 (2017).
Bordwell, F. G. Equilibrium acidities in dimethyl sulfoxide solution. Acc. Chem. Res. 21, 456–463 (1988).
Trost, B. M., Xu, J. & Schmidt, T. Palladium-catalyzed decarboxylative asymmetric allylic alkylation of enol carbonates. J. Am. Chem. Soc. 131, 18343–18357 (2009).
Weaver, J. D., Recio, A., Grenning, A. J. & Tunge, J. A. Transition metal-catalyzed decarboxylative allylation and benzylation reactions. Chem. Rev. 111, 1846–1913 (2011).
Xie, H., Zhang, H. & Lin, Z. DFT studies on the palladium-catalyzed dearomatization reaction between chloromethylnaphthalene and the cyclic amine morpholine. Organometallics 32, 2336–2343 (2013).
Boga, C., Del Vecchio, E., Forlani, L. & Tozzi, S. Evidence of reversibility in azo-coupling reactions between 1,3,5-tris(N,N-dialkylamino)benzenes and arenediazonium salts. J. Org. Chem. 72, 8741–8747 (2007).
Forlani, L., Boga, C., Del Vecchio, E., Ngobo, A.-L. T. D. & Tozzi, S. Reactions of Wheland complexes: base catalysis in re-aromatization reaction of σ complexes obtained from 1,3,5-tris(N,N-dialkylamino)benzene and arenediazonium salts. J. Phys. Org. Chem. 20, 201–205 (2007).
Simmons, E. M. & Hartwig, J. F. On the interpretation of deuterium kinetic isotope effects in C–H bond functionalizations by transition-metal complexes. Angew. Chem. Int. Ed. 51, 3066–3072 (2012).
Ma, S., Villa, G., Thuy-Boun, P. S., Homs, A. & Yu, J.-Q. Palladium-catalyzed ortho-selective C–H deuteration of arenes: evidence for superior reactivity of weakly coordinated palladacycles. Angew. Chem. Int. Ed. 53, 734–737 (2014).
Ariafard, A. & Lin, Z. DFT studies on the mechanism of allylative dearomatization catalyzed by palladium. J. Am. Chem. Soc. 128, 13010–13016 (2006).
Acknowledgements
We thank the donors of the Herman Frasch Foundation for Chemical Research (701-HF12), the National Science Foundation (NSF, CHE-1455163) and the National Institute of General Medical Sciences (R35 GM124661) for supporting this work. NMR instrumentation was provided by NIH (S10OD016360, S10RR024664 and P20GM103418) and NSF Grants (9977422 and 0320648). P.H.-Y.C. acknowledges financial support from the Bert and Emelyn Christensen Professorship and the Vicki & Patrick F. Stone family. P.H.-Y.C., T.F., M.A.G. and A.C.B. acknowledge the NSF (CHE-1352663) and the computing infrastructure in part provided by the NSF Phase-2 CCI, Center for Sustainable Materials Chemistry (NSF, CHE-1102637). T.F. acknowledges the Summer Fellowship Award from the department of Chemistry at Oregon State University.
Author information
Authors and Affiliations
Contributions
F.d.A. and M.-H.Y. contributed equally. R.A.A. and M.-H.Y. created the project. R.A.A., M.-H.Y., F.d.A., M.S. and S.K. designed the experiments. M.-H.Y., M.S. and S.K. optimized the reaction conditions. M.-H.Y., F.d.A., M.S. and S.K. explored the substrate scope. F.d.A. designed and conducted the mechanistic experiments. R.A.A. supervised the synthetic and mechanistic portions of the experimental work. T.F. used density functional theory (DFT) to compute the key transition states and intermediates in the different proposed mechanisms, which ultimately led to pinpointing the operative pathway; A.C.B. performed the initial DFT computations; M.A.G. performed energy refinements at various levels of theory to verify that the DFT results were in line with experimental results. P.H.-Y.C. supervised the computational aspect of the work and also contributed to the DFT energy refinements. All the authors contributed to the writing and editing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Experimental details for synthetic chemistry, including: general synthetic information and procedures, data for reaction optimization, characterization of substrates and products, data supporting mechanistic investigations. Computational details, including: complete authorship of Gaussian 09 and Gaussian 16, computational procedure, reaction coordinate diagram with higher-in-energy intermediates, exploration of para-selectivity of arylation process, reaction coordinate diagram with dimethyl substrate, coordinates for relevant computed structures. NMR spectra supporting the characterization of substrates and products and mechanistic studies
Rights and permissions
About this article
Cite this article
de Azambuja, F., Yang, MH., Feoktistova, T. et al. Connecting remote C–H bond functionalization and decarboxylative coupling using simple amines. Nat. Chem. 12, 489–496 (2020). https://doi.org/10.1038/s41557-020-0428-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-020-0428-1
This article is cited by
-
α-C(sp3)-H Arylation of Cyclic Carbonyl Compounds
Natural Products and Bioprospecting (2021)