Abstract
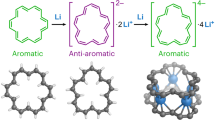
Aromaticity is a vital concept that governs the electronic properties of π-conjugated organic molecules and has long been restricted to 2D systems. The aromaticity in 3D π-conjugated molecules has been rarely studied. Here we report a fully conjugated diradicaloid molecular cage and its global aromaticity at different oxidation states. The neutral compound has an open-shell singlet ground state with a dominant 38π monocyclic conjugation pathway and follows the [4n + 2] Hückel aromaticity rule; the dication has a triplet ground state with a dominant 36π monocyclic conjugation pathway and satisfies [4n] Baird aromaticity; the tetracation is an open-shell singlet with 52 π-electrons that are delocalized along the 3D rigid framework, showing 3D global antiaromaticity; and the hexacation possesses D3 symmetry with 50 globally delocalized π-electrons, showing [6n + 2] 3D global aromaticity. Different types of aromaticity were therefore accessed in one molecular cage platform, depending on the symmetry, number of π-electrons and spin state.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information. Output files of the optimized geometries of c-T12, c-T122+, c-T124+, c-T126+ and c-T12′6+ in their respective ground states are available as Supplementary Data. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition nos. CCDC 1907315 (c-T12), 1907316 (c-T122+), 1907317 (c-T124+), 1907318 (c-T125+) and 1907319 (c-T126+). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Hückel, E. Quantentheoretische beiträge zum benzolproblem. Z. Phys. 70, 204–286 (1931).
Krygowski, T. M., Cyrañki, M. K., Czarnocki, Z., Häelinger, G. & Katritzky, A. R. Aromaticity: a theoretical concept of immense practical importance. Tetrahedron 56, 1783–1796 (2000).
Cyrański, M. K. Energetic aspects of cyclic π-electron delocalization: evaluation of the methods of estimating aromatic stabilization energies. Chem. Rev. 105, 3773–3811 (2005).
Breslow, R. Antiaromaticity. Acc. Chem. Res. 6, 393–398 (1973).
Voter, A. F. & Goddard, W. A. The generalized resonating valence bond description of cyclobutadiene. J. Am. Chem. Soc. 108, 2830–2837 (1986).
Tanaka, T. & Osuka, A. Chemistry of meso-aryl-substituted expanded porphyrins: aromaticity and molecular twist. Chem. Rev. 117, 2584–2640 (2017).
Stępień, M. & Latos-Grażyński, L. Aromaticity and tautomerism in porphyrins and porphyrinoids. Top. Heterocycl. Chem. 19, 82–153 (2008).
Peeks, M. D., Claridge, T. D. W. & Anderson, H. L. Aromatic and antiaromatic ring currents in a molecular nanoring. Nature 541, 200–203 (2017).
Toriumi, N., Muranaka, A., Kayahara, E., Yamago, S. & Uchiyama, M. In-plane aromaticity in cycloparaphenylene dications: a magnetic circular dichroism and theoretical study. J. Am. Chem. Soc. 137, 82–85 (2015).
Baird, N. C. Quantum organic photochemistry. II. Resonance and aromaticity in the lowest 3ππ* state of cyclic hydrocarbons. J. Am. Chem. Soc. 94, 4941–4948 (1972).
Sung, Y. M. et al. Reversal of Hückel (anti)aromaticity in the lowest triplet states of hexaphyrins and spectroscopic evidence for Baird’s rule. Nat. Chem. 7, 418–422 (2015).
Rosenberg, M. et al. Excited state aromaticity and antiaromaticity: opportunities for photophysical and photochemical rationalizations. Chem. Rev. 114, 5379–5425 (2014).
Saunders, M. et al. Unsubstituted cyclopentadienyl cation, a ground-state triplet. J. Am. Chem. Soc. 95, 3017 (1973).
Heilbronner, E. Hückel molecular orbitals of Möbius-type conformations of annulenes. Tetrahedron Lett. 29, 1923–1928 (1964).
Ajami, D., Oeckler, O., Simon, A. & Herges, R. Synthesis of a Möbius aromatic hydrocarbon. Nature 426, 819–821 (2003).
Yoon, Z. S., Osuka, A. & Kim, D. Möbius aromaticity and antiaromaticity in expanded porphyrins. Nat. Chem. 1, 113–122 (2009).
Stępień, M., Latos-Grażyński, L., Sprutta, N., Chwalisz, P. & Szterenberg, L. Expanded porphyrin with a split personality: a Hückel–Möbius aromaticity switch. Angew. Chem. Int. Ed. 46, 7869–7873 (2007).
Stępień, M., Sprutta, N. & Latos-Grażyński, L. Figure eights, Möbius bands, and more: conformation and aromaticity of porphyrinoids. Angew. Chem. Int. Ed. 50, 4288–4340 (2011).
Hirsch, A., Chen, Z. & Jiao, H. Spherical aromaticity in I h symmetrical fullerenes: the 2(N+1)2 rule. Angew. Chem. Int. Ed. 39, 3915–3917 (2000).
Bühl, M. & Hirsch, A. Spherical aromaticity of fullerenes. Chem. Rev. 101, 1153–1183 (2001).
Chen, Z. & King, R. B. Spherical aromaticity: recent work on fullerenes, polyhedral boranes, and related structures. Chem. Rev. 105, 3613–3642 (2005).
Chen, Z., Jiao, H., Hirsch, A. & Schleyer, Pv. R. Spherical homoaromaticity. Angew. Chem. Int. Ed. 41, 4309–4312 (2002).
Corminboeuf, C., Schleyer, P. v. R. & Warner, P. Are antiaromatic rings stacked face-to-face aromatic? Org. Lett. 9, 3263–3266 (2007).
Nozawa, R. et al. Stacked antiaromatic porphyrins. Nat. Commun. 7, 13620 (2016).
Högberg, H.-E., Thulin, B. & Wennerström, O. Bicyclophanehexaene, a new case cyclophane from a sixfold Wittig reaction. Tetrahedron Lett. 18, 931–934 (1977).
Wu, Z., Lee, S. & Moore, J. S. Synthesis of three-dimensional nanoscaffolding. J. Am. Chem. Soc. 114, 8730–8732 (1992).
Kayahara, E. et al. Synthesis and physical properties of a ball-like three-dimensional π-conjugated molecule. Nat. Commun. 4, 2694 (2013).
Matsui, K., Segawa, Y. & Itami, K. All-benzene carbon nanocages: size-selective synthesis, photophysical properties, and crystal structure. J. Am. Chem. Soc. 136, 16452–16458 (2014).
Song, J., Aratani, N., Shinokubo, H. & Osuka, A. A porphyrin nanobarrel that encapsulates C60. J. Am. Chem. Soc. 132, 16356–16357 (2010).
Zhang, C., Wang, Q., Long, H. & Zhang, W. A highly C70 selective shape-persistent rectangular prism constructed through one-step alkyne metathesis. J. Am. Chem. Soc. 133, 20995–21001 (2011).
Ke, X.-S. et al. Three-dimensional fully conjugated carbaporphyrin cage. J. Am. Chem. Soc. 140, 16455–16459 (2018).
Oh, J. et al. Unraveling excited-singlet-state aromaticity via vibrational Analysis. Chem 3, 870–880 (2017).
Soya, T. et al. Internally 2,5‐thienylene‐bridged [46]decaphyrin: (annuleno)annulene network consisting of Möbius aromatic thia[28]hexaphyrins and strong Hückel aromaticity of its protonated form. Angew. Chem. Int. Ed. 56, 3232–3236 (2017).
Cha, W.-Y. et al. Bicyclic Baird-type aromaticity. Nat. Chem. 9, 1243–1248 (2017).
Das, S. et al. Fully fused quinoidal/aromatic carbazole macrocycles with poly-radical characters. J. Am. Chem. Soc. 138, 7782–7790 (2016).
Lu, X. et al. Fluorenyl based macrocyclic polyradicaloids. J. J. Am. Chem. Soc. 139, 13173–13183 (2017).
Liu, C. et al. Macrocyclic polyradicaloids with unusual super-ring structure and global aromaticity. Chem 4, 1586–1595 (2018).
Liu, C., Ni, Y., Lu, X., Li, G. & Wu, J. Global aromaticity in macrocyclic polyradicaloids: Hückel’s rule or Baird’s rule? Acc. Chem. Res. 52, 2309–2321 (2019).
Nakano, M. et al. Second hyperpolarizability of zethrenes. Comp. Lett. 3, 333–338 (2007).
Kamada, K. et al. Singlet diradical character from experiment. J. Phys. Chem. Lett. 1, 937–940 (2010).
Katritzky, A. R., Jug, K. & Oniciu, D. C. Quantitative measures of aromaticity for mono-, bi-, and tricyclic penta- and hexaatomic heteroaromatic ring systems and their interrelationships. Chem. Rev. 101, 1421–1449 (2001).
Schleyer, Pv. R., Jiao, H., Goldfuss, B. & Freeman, P. K. Aromaticity and antiaromaticity in five‐membered C4H4X ring systems: “classical” and “magnetic” concepts may not be “orthogonal”. Angew. Chem. Int. Ed. Engl. 34, 337–340 (1995).
Schleyer, Pv. R., Maerker, C., Dransfeld, A., Jiao, H. & Hommes, N. J. Rv. E. Nucleus-independent chemical shifts: a simple and efficient aromaticity probe. J. Am. Chem. Soc. 118, 6317–6318 (1996).
Gopalakrishna, T. Y., Reddy, J. S. & Anand, V. G. An amphoteric switch to aromatic and antiaromatic states of a neutral air-stable 25π radical. Angew. Chem. Int. Ed. 53, 10984–10987 (2014).
Zeng, Z. et al. Pro-aromatic and antiaromatic π-conjugated molecules: an irresistible wish to be diradicals. Chem. Soc. Rev. 44, 6578–6596 (2015).
Kubo, T. Recent progress in quinoidal singlet biradical molecules. Chem. Lett. 44, 111–122 (2015).
Gopalakrishna, T. Y., Zeng, W., Lu, X. & Wu, J. From open-shell singlet diradicaloids to polyradicaloids. Chem. Commun. 54, 2186–2199 (2018).
Bleaney, B. & Bowers, K. D. Anomalous paramagnetism of copper acetate. Proc. R. Soc. Lond. A 214, 451–465 (1952).
Choi, C. H. & Kertesz, M. Bond length alternation and aromaticity in large annulenes. J. Chem. Phy. 108, 6681––66688 (1998).
Geuenich, D., Hess, K., Köhler, F. & Herges, R. Anisotropy of the induced current density (ACID), a general method to quantify and visualize electronic delocalization. Chem. Rev. 105, 3758–3772 (2005).
Valiev, R. R., Fliegl, H. & Sundholm, D. Bicycloaromaticity and Baird-type bicycloaromaticity of dithienothiophene-bridged [34]octaphyrins. Phys. Chem. Chem. Phys. 20, 17705–17713 (2018).
Chen, Z., Wannere, C. S., Corminboeuf, C., Puchta, R. & Schleyer, Pv. R. Nucleus-independent chemical shifts (NICS) as an aromaticity criterion. Chem. Rev. 105, 3842–3888 (2005).
Humphrey, W., Dalke, A. & Schulten, K. VMD-visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Shin, J.-Y. et al. Aromaticity and photophysical properties of various topology-controlled expanded porphyrins. Chem. Soc. Rev. 39, 2751–2767 (2010).
Acknowledgements
J.W. acknowledges financial support from the MOE Tier 3 programme (grant no. MOE2014-T3-1-004) and NRF Investigatorship (grant no. NRF-NRFI05-2019-0005). We thank S. Tobias and H. Ott from Brucker for their support on X-ray diffraction data collection. We also thank Z. Chen in the University of Puerto Rico for his helpful discussion.
Author information
Authors and Affiliations
Contributions
J.W. and Y.N. conceived the project. Y.N. synthesized the compounds and collected the spectral data. T.Y.G. performed theoretical calculations. Y.N., T.S.H., H.P. and J.D. did magnetic measurements and analysis. Y.N., Y.H. and T.T. did the X-ray analysis. T.K. and D.K. did the transient absorption spectral measurement and analysis. All authors participated in the manuscript writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Experimental methods, synthetic procedures and characterization data. Details of theoretical calculations and X-ray crystallographic analysis.
Crystallographic data
CIF for compound c-T12; CCDC reference: 1907315.
Crystallographic data
CIF for compound c-T122+; CCDC reference: 1907316.
Crystallographic data
CIF for compound c-T124+; CCDC reference: 1907317.
Crystallographic data
CIF for compound c-T125+; CCDC reference: 1907318.
Crystallographic data
CIF for compound c-T126+; CCDC reference: 1907319.
Computational data
Output files of the optimized geometries of c-T12, c-T122+, c-T124+, c-T126+ and c-T12′6+ in their respective ground states. Note that c-T12′6+ is an analogue of c-T126+ in which the Mes groups are replaced by hydrogen atoms.
Rights and permissions
About this article
Cite this article
Ni, Y., Gopalakrishna, T.Y., Phan, H. et al. 3D global aromaticity in a fully conjugated diradicaloid cage at different oxidation states. Nat. Chem. 12, 242–248 (2020). https://doi.org/10.1038/s41557-019-0399-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0399-2
This article is cited by
-
Clustering-triggered phosphorescence of nonconventional luminophores
Science China Chemistry (2023)
-
3D and 2D aromatic units behave like oil and water in the case of benzocarborane derivatives
Nature Communications (2022)
-
Design of an open-shell nitrogen-centered diradicaloid with tunable stimuli-responsive electronic properties
Communications Chemistry (2022)
-
Isolation of a triplet benzene dianion
Nature Chemistry (2021)
-
Cyclic polyacetylene
Nature Chemistry (2021)