Abstract
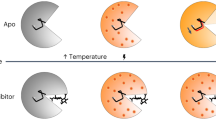
Correlated motions of proteins are critical to function, but these features are difficult to resolve using traditional structure determination techniques. Time-resolved X-ray methods hold promise for addressing this challenge, but have relied on the exploitation of exotic protein photoactivity, and are therefore not generalizable. Temperature jumps, through thermal excitation of the solvent, have been utilized to study protein dynamics using spectroscopic techniques, but their implementation in X-ray scattering experiments has been limited. Here, we perform temperature-jump small- and wide-angle X-ray scattering measurements on a dynamic enzyme, cyclophilin A, demonstrating that these experiments are able to capture functional intramolecular protein dynamics on the microsecond timescale. We show that cyclophilin A displays rich dynamics following a temperature jump, and use the resulting time-resolved signal to assess the kinetics of conformational changes. Two relaxation processes are resolved: a fast process is related to surface loop motions, and a slower process is related to motions in the core of the protein that are critical for catalytic turnover.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Scattering data are deposited at NIH Figshare (https://doi.org/10.35092/yhjc.9177143). Additional information and files are available from the corresponding author upon reasonable request.
Code availability
All Python scripts used for analysis of integrated X-ray scattering curves are publicly available from GitHub (https://github.com/fraser-lab/solution_scattering). A code release checkpoint containing the exact scripts used in this work is available via Zenodo (https://doi.org/10.5281/zenodo.3355707).
References
Henzler-Wildman, K. & Kern, D. Dynamic personalities of proteins. Nature 450, 964–972 (2007).
Van den Bedem, H. & Fraser, J. S. Integrative, dynamic structural biology at atomic resolution—it’s about time. Nat. Methods 12, 307–318 (2015).
Bottaro, S. & Lindorff-Larsen, K. Biophysical experiments and biomolecular simulations: a perfect match? Science 361, 355–360 (2018).
Schmidt, M. Time-resolved macromolecular crystallography at modern X-ray sources. Methods Mol. Biol. 1607, 273–294 (2017).
Neutze, R. & Moffat, K. Time-resolved structural studies at synchrotrons and X-ray free electron lasers: opportunities and challenges. Curr. Opin. Struct. Biol. 22, 651–659 (2012).
Schotte, F. et al. Watching a signaling protein function in real time via 100-ps time-resolved Laue crystallography. Proc. Natl Acad. Sci. USA 109, 19256–19261 (2012).
Cho, H. S. et al. Picosecond photobiology: watching a signaling protein function in real time via time-resolved small- and wide-angle X-ray scattering. J. Am. Chem. Soc. 138, 8815–8823 (2016).
Schlichting, I. & Miao, J. Emerging opportunities in structural biology with X-ray free-electron lasers. Curr. Opin. Struct. Biol. 22, 613–626 (2012).
Arnlund, D. et al. Visualizing a protein quake with time-resolved X-ray scattering at a free-electron laser. Nat. Methods 11, 923–926 (2014).
Berntsson, O. et al. Sequential conformational transitions and α-helical supercoiling regulate a sensor histidine kinase. Nat. Commun. 8, 284 (2017).
Takala, H. et al. Signal amplification and transduction in phytochrome photosensors. Nature 509, 245–248 (2014).
Brinkmann, L. U. L. & Hub, J. S. Ultrafast anisotropic protein quake propagation after CO photodissociation in myoglobin. Proc. Natl Acad. Sci. USA 113, 10565–10570 (2016).
Barends, T. R. M. et al. Direct observation of ultrafast collective motions in CO myoglobin upon ligand dissociation. Science 350, 445–450 (2015).
Coquelle, N. et al. Chromophore twisting in the excited state of a photoswitchable fluorescent protein captured by time-resolved serial femtosecond crystallography. Nat. Chem. 10, 31–37 (2018).
Pande, K. et al. Femtosecond structural dynamics drives the trans/cis isomerization in photoactive yellow protein. Science 352, 725–729 (2016).
Kern, J. et al. Structures of the intermediates of Kok’s photosynthetic water oxidation clock. Nature 563, 421–425 (2018).
Nogly, P. et al. Retinal isomerization in bacteriorhodopsin captured by a femtosecond X-ray laser. Science 361, eaat0094 (2018).
Malmerberg, E. et al. Time-resolved WAXS reveals accelerated conformational changes in iodoretinal-substituted proteorhodopsin. Biophys. J. 101, 1345–1353 (2011).
Hekstra, D. R. et al. Electric-field-stimulated protein mechanics. Nature 540, 400–405 (2016).
Schlichting, I. et al. Time-resolved X-ray crystallographic study of the conformational change in Ha-Ras p21 protein on GTP hydrolysis. Nature 345, 309–315 (1990).
Stoddard, B. L., Cohen, B. E., Brubaker, M., Mesecar, A. D. & Koshland, D. E. Jr Millisecond Laue structures of an enzyme-product complex using photocaged substrate analogs. Nat. Struct. Biol. 5, 891–897 (1998).
Josts, I. et al. Photocage-initiated time-resolved solution X-ray scattering investigation of protein dimerization. IUCrJ 5, 667–672 (2018).
Rimmerman, D. et al. Revealing fast structural dynamics in pH-responsive peptides with time-resolved X-ray scattering. J. Phys. Chem. B 123, 2016–2021 (2019).
Olmos, J. L. Jr et al. Enzyme intermediates captured ‘on the fly’ by mix-and-inject serial crystallography. BMC Biol. 16, 59 (2018).
Rimmerman, D. et al. Direct observation of insulin association dynamics with time-resolved X-ray scattering. J. Phys. Chem. Lett. 8, 4413–4418 (2017).
Rimmerman, D. et al. Insulin hexamer dissociation dynamics revealed by photoinduced T-jumps and time-resolved X-ray solution scattering. Photochem. Photobiol. Sci. 17, 874–882 (2018).
Cho, H. S. et al. Dynamics of quaternary structure transitions in R-state carbonmonoxyhemoglobin are unveiled in time-resolved X-ray scattering patterns following a temperature jump. J. Phys. Chem. B 122, 11488–11496 (2018).
Frauenfelder, H., Fenimore, P. W. & Young, R. D. Protein dynamics and function: insights from the energy landscape and solvent slaving. IUBMB Life 59, 506–512 (2007).
Fenimore, P. W., Frauenfelder, H., McMahon, B. H. & Parak, F. G. Slaving: solvent fluctuations dominate protein dynamics and functions. Proc. Natl Acad. Sci. USA 99, 16047–16051 (2002).
Wang, J. & El-Sayed, M. A. Temperature jump-induced secondary structural change of the membrane protein bacteriorhodopsin in the premelting temperature region: a nanosecond time-resolved Fourier transform infrared study. Biophys. J. 76, 2777–2783 (1999).
Wang, T., Lau, W. L., DeGrado, W. F. & Gai, F. T-jump infrared study of the folding mechanism of coiled-coil GCN4-p1. Biophys. J. 89, 4180–4187 (2005).
Akasaka, K., Naito, A. & Nakatani, H. Temperature-jump NMR study of protein folding: ribonuclease A at low pH. J. Biomol. NMR 1, 65–70 (1991).
Gillespie, B. et al. NMR and temperature-jump measurements of de novo designed proteins demonstrate rapid folding in the absence of explicit selection for kinetics. J. Mol. Biol. 330, 813–819 (2003).
Yamasaki, K. et al. Real-time NMR monitoring of protein-folding kinetics by a recycle flow system for temperature jump. Anal. Chem. 85, 9439–9443 (2013).
Meadows, C. W., Balakrishnan, G., Kier, B. L., Spiro, T. G. & Klinman, J. P. Temperature-jump fluorescence provides evidence for fully reversible microsecond dynamics in a thermophilic alcohol dehydrogenase. J. Am. Chem. Soc. 137, 10060–10063 (2015).
Vaughn, M. B., Zhang, J., Spiro, T. G., Dyer, R. B. & Klinman, J. P. Activity-related microsecond dynamics revealed by temperature-jump Förster resonance energy transfer measurements on thermophilic alcohol dehydrogenase. J. Am. Chem. Soc. 140, 900–903 (2018).
Hori, T. et al. The initial step of the thermal unfolding of 3-isopropylmalate dehydrogenase detected by the temperature-jump Laue method. Protein Eng. 13, 527–533 (2000).
Eisenmesser, E. Z. et al. Intrinsic dynamics of an enzyme underlies catalysis. Nature 438, 117–121 (2005).
Caines, M. E. C. et al. Diverse HIV viruses are targeted by a conformationally dynamic antiviral. Nat. Struct. Mol. Biol. 19, 411–416 (2012).
Price, A. J. et al. Active site remodeling switches HIV specificity of antiretroviral TRIMCyp. Nat. Struct. Mol. Biol. 16, 1036–1042 (2009).
Virgen, C. A., Kratovac, Z., Bieniasz, P. D. & Hatziioannou, T. Independent genesis of chimeric TRIM5-cyclophilin proteins in two primate species. Proc. Natl Acad. Sci. USA 105, 3563–3568 (2008).
Wilson, S. J. et al. Independent evolution of an antiviral TRIMCyp in rhesus macaques. Proc. Natl Acad. Sci. USA 105, 3557–3562 (2008).
Keedy, D. A. et al. Mapping the conformational landscape of a dynamic enzyme by multitemperature and XFEL crystallography.eLife 4, e07574 (2015).
Fraser, J. S. et al. Hidden alternative structures of proline isomerase essential for catalysis. Nature 462, 669–673 (2009).
Otten, R. et al. Rescue of conformational dynamics in enzyme catalysis by directed evolution. Nat. Commun. 9, 1314 (2018).
Clark, G. N. I., Hura, G. L., Teixeira, J., Soper, A. K. & Head-Gordon, T. Small-angle scattering and the structure of ambient liquid water. Proc. Natl Acad. Sci. USA 107, 14003–14007 (2010).
Kjær, K. S. et al. Introducing a standard method for experimental determination of the solvent response in laser pump, X-ray probe time-resolved wide-angle X-ray scattering experiments on systems in solution. Phys. Chem. Chem. Phys. 15, 15003–15016 (2013).
Cammarata, M. et al. Impulsive solvent heating probed by picosecond X-ray diffraction. J. Chem. Phys. 124, 124504 (2006).
Gruebele, M., Sabelko, J., Ballew, R. & Ervin, J. Laser temperature jump induced protein refolding. Acc. Chem. Res. 31, 699–707 (1998).
Levantino, M. et al. Ultrafast myoglobin structural dynamics observed with an X-ray free-electron laser. Nat. Commun. 6, 6772 (2015).
DiCiccio, T. J. & Efron, B. Bootstrap confidence intervals. Stat. Sci. 11, 189–212 (1996).
Smith, C. A. et al. Population shuffling of protein conformations. Angew. Chem. Int. Ed. Engl. 54, 207–210 (2015).
Wapeesittipan, P., Mey, A., Walkinshaw, M. & Michel, J. Allosteric effects in catalytic impaired variants of the enzyme cyclophilin A may be explained by changes in nano-microsecond time scale motions. Commun. Chem. 2, 41 (2019).
Chi, C. N. et al. A structural ensemble for the enzyme cyclophilin reveals an orchestrated mode of action at atomic resolution. Angew. Chem. Int. Ed. Engl. 54, 11657–11661 (2015).
Kim, J. G. et al. Cooperative protein structural dynamics of homodimeric hemoglobin linked to water cluster at subunit interface revealed by time-resolved X-ray solution scattering. Struct. Dynam. 3, 023610 (2016).
Colombo, M. F., Rau, D. C. & Parsegian, V. A. Protein solvation in allosteric regulation: a water effect on hemoglobin. Science 256, 655–659 (1992).
Salvay, A. G., Grigera, J. R. & Colombo, M. F. The role of hydration on the mechanism of allosteric regulation: in situ measurements of the oxygen-linked kinetics of water binding to hemoglobin. Biophys. J. 84, 564–570 (2003).
Royer, W. E. Jr, Pardanani, A., Gibson, Q. H., Peterson, E. S. & Friedman, J. M. Ordered water molecules as key allosteric mediators in a cooperative dimeric hemoglobin. Proc. Natl Acad. Sci. USA 93, 14526–14531 (1996).
Fenwick, R. B., Oyen, D., Dyson, H. J. & Wright, P. E. Slow dynamics of tryptophan–water networks in proteins. J. Am. Chem. Soc. 140, 675–682 (2018).
Grossman, M. et al. Correlated structural kinetics and retarded solvent dynamics at the metalloprotease active site. Nat. Struct. Mol. Biol. 18, 1102–1108 (2011).
Decaneto, E. et al. Solvent water interactions within the active site of the membrane type I matrix metalloproteinase. Phys. Chem. Chem. Phys. 19, 30316–30331 (2017).
Leidner, F., Kurt Yilmaz, N., Paulsen, J., Muller, Y. A. & Schiffer, C. A. Hydration structure and dynamics of inhibitor-bound HIV-1 protease. J. Chem. Theory Comput. 14, 2784–2796 (2018).
Guha, S. et al. Slow solvation dynamics at the active site of an enzyme: implications for catalysis. Biochemistry 44, 8940–8947 (2005).
Dahanayake, J. N. & Mitchell-Koch, K. R. Entropy connects water structure and dynamics in protein hydration layer. Phys. Chem. Chem. Phys. 20, 14765–14777 (2018).
Wand, A. J. & Sharp, K. A. Measuring entropy in molecular recognition by proteins. Annu. Rev. Biophys. 47, 41–61 (2018).
Caro, J. A. et al. Entropy in molecular recognition by proteins. Proc. Natl Acad. Sci. USA 114, 6563–6568 (2017).
Gavrilov, Y., Leuchter, J. D. & Levy, Y. On the coupling between the dynamics of protein and water. Phys. Chem. Chem. Phys. 19, 8243–8257 (2017).
Conti Nibali, V., D’Angelo, G., Paciaroni, A., Tobias, D. J. & Tarek, M. On the coupling between the collective dynamics of proteins and their hydration water. J. Phys. Chem. Lett. 5, 1181–1186 (2014).
Hub, J. S. Interpreting solution X-ray scattering data using molecular simulations. Curr. Opin. Struct. Biol. 49, 18–26 (2018).
Svergun, D. I. et al. Protein hydration in solution: experimental observation by X-ray and neutron scattering. Proc. Natl Acad. Sci. USA 95, 2267–2272 (1998).
Virtanen, J. J., Makowski, L., Sosnick, T. R. & Freed, K. F. Modeling the hydration layer around proteins: applications to small- and wide-angle X-ray scattering. Biophys. J. 101, 2061–2069 (2011).
Henriques, J., Arleth, L., Lindorff-Larsen, K. & Skepö, M. On the calculation of SAXS profiles of folded and intrinsically disordered proteins from computer simulations. J. Mol. Biol. 430, 2521–2539 (2018).
Dellus-Gur, E. et al. Negative epistasis and evolvability in TEM-1 β-lactamase—the thin line between an enzyme’s conformational freedom and disorder. J. Mol. Biol. 427, 2396–2409 (2015).
Biel, J. T., Thompson, M. C., Cunningham, C. N., Corn, J. E. & Fraser, J. S. Flexibility and design: conformational heterogeneity along the evolutionary trajectory of a redesigned ubiquitin. Structure 25, 739–749.e3 (2017).
Fischer, M., Coleman, R. G., Fraser, J. S. & Shoichet, B. K. Incorporation of protein flexibility and conformational energy penalties in docking screens to improve ligand discovery. Nat. Chem. 6, 575–583 (2014).
Fischer, M., Shoichet, B. K. & Fraser, J. S. One crystal, two temperatures: cryocooling penalties alter ligand binding to transient protein sites. ChemBioChem 16, 1560–1564 (2015).
Keedy, D. A. et al. An expanded allosteric network in PTP1B by multitemperature crystallography, fragment screening, and covalent tethering. eLife 7, e36307 (2018).
Fraser, J. S. et al. Accessing protein conformational ensembles using room-temperature X-ray crystallography. Proc. Natl Acad. Sci. USA 108, 16247–16252 (2011).
Van den Bedem, H., Bhabha, G., Yang, K., Wright, P. E. & Fraser, J. S. Automated identification of functional dynamic contact networks from X-ray crystallography. Nat. Methods 10, 896–902 (2013).
Acknowledgements
We thank R. Ranganathan, J. Holton, G. Hura and D. Elnatan for helpful discussions, and the staff at the BioCARS beamline at the Advanced Photon Source (I. Kosheleva, R. Henning, A. DiChiara and V. Srajer) for assistance. This work was supported by the NSF (STC-1231306), the NIH (GM123159 and GM124149), a Packard Fellowship from the David and Lucile Packard Foundation, the UC Office of the President Laboratory Fees Research Program LFR-17-476732 (to J.S.F.), the Intramural Research Program of the National Institute of Diabetes and Digestive and Kidney Diseases (to P.A.) and a Ruth L. Kirschstein National Research Service Award (F32 HL129989 to M.C.T.). Use of the Advanced Photon Source was supported by the US Department of Energy, Basic Energy Sciences, Office of Science, under contract number DE-AC02-06CH11357. Use of the BioCARS Sector 14 was also supported by the NIH National Institute of General Medical Sciences (grant R24GM111072). The time-resolved setup at Sector 14 was funded in part through a collaboration with P.A. (NIH/NIDDK).
Author information
Authors and Affiliations
Contributions
M.C.T., P.A. and J.S.F. conceived and designed the experiments. M.C.T., B.A.B., A.M.W., H.S.C., F.S., D.M.C.S. and P.A. performed the experiments. M.C.T., B.A.B. and A.M.W. analysed the data. M.C.T., B.A.B., A.M.W., H.S.C., F.S. and P.A. contributed materials/analysis tools. M.C.T. and J.S.F. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–8, Supplementary Table 1 and Supplementary Data, Methods and References.
Rights and permissions
About this article
Cite this article
Thompson, M.C., Barad, B.A., Wolff, A.M. et al. Temperature-jump solution X-ray scattering reveals distinct motions in a dynamic enzyme. Nat. Chem. 11, 1058–1066 (2019). https://doi.org/10.1038/s41557-019-0329-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0329-3
This article is cited by
-
Activation and friction in enzymatic loop opening and closing dynamics
Nature Communications (2024)
-
Visualization of protein motions using temperature-jump crystallography
Nature Chemistry (2023)
-
Single-pulse terahertz spectroscopy monitoring sub-millisecond time dynamics at a rate of 50 kHz
Nature Communications (2023)
-
Mapping protein dynamics at high spatial resolution with temperature-jump X-ray crystallography
Nature Chemistry (2023)