Abstract
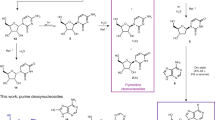
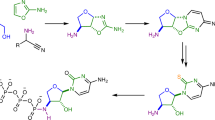
Breakthroughs in the study of the origin of life have demonstrated how some of the building blocks essential to biology could have been formed under various primordial scenarios, and could therefore have contributed to the chemical evolution of life. Missing building blocks are then sometimes inferred to be products of primitive biosynthesis, which can stretch the limits of plausibility. Here, we demonstrate the synthesis of 2′-deoxy-2-thiouridine, and subsequently 2′-deoxyadenosine and 2-deoxyribose, under prebiotic conditions. 2′-Deoxy-2-thiouridine is produced by photoreduction of 2,2′-anhydro-2-thiouridine, which is in turn formed by phosphorylation of 2-thiouridine—an intermediate of prebiotic RNA synthesis. 2′-Deoxy-2-thiouridine is an effective deoxyribosylating agent and may have functioned as such in either abiotic or proto-enzyme-catalysed pathways to DNA, as demonstrated by its conversion to 2′-deoxyadenosine by reaction with adenine, and 2-deoxyribose by hydrolysis. An alternative prebiotic phosphorylation of 2-thiouridine leads to the formation of its 5′-phosphate, showing that hypotheses in which 2-thiouridine was a key component of early RNA sequences are within the bounds of synthetic credibility.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rao, Y. S., Thimmappayya, B. & Cherayil, J. D. Thionucleotides in the transfer ribonucleic acids of Pseudomonas aeruginosa and Escherichia coli. Biochem. J. 128, 67 (1972).
Larsen, A. T., Fahrenbach, A. C., Sheng, J., Pian, J. & Szostak, J. W. Thermodynamic insights into 2-thiouridine-enhanced RNA hybridization. Nucleic Acids Res. 43, 7675–7687 (2015).
Heuberger, B. D., Pal, A., Del Frate, F., Topkar, V. V. & Szostak, J. W. Replacing uridine with 2-thiouridine enhances the rate and fidelity of nonenzymatic RNA primer extension. J. Am. Chem. Soc. 137, 2769–2775 (2015).
Patel, B. H., Percivalle, C., Ritson, D. J., Duffy, C. D. & Sutherland, J. D. Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism. Nat. Chem. 7, 301–307 (2015).
Xu, J. et al. A prebiotically plausible synthesis of pyrimidine beta-ribonucleosides and their phosphate derivatives involving photoanomerization. Nat. Chem. 9, 303–309 (2017).
Verlander, M. S., Lohrmann, R. & Orgel, L. E. Catalysts for the self-polymerization of adenosine cyclic 2′,3′-phosphate. J. Mol. Evol. 2, 303–316 (1973).
Schoffstall, A. M. Prebiotic phosphorylation of nucleosides in formamide. Orig. Life 7, 399–412 (1976).
Lohrmann, R. & Orgel, L. E. Urea–inorganic phosphate mixtures as prebiotic phosphorylating agents. Science 171, 490–494 (1971).
Li, L. et al. Enhanced nonenzymatic RNA copying with 2-aminoimidazole activated nucleotides. J. Am. Chem. Soc. 139, 1810–1813 (2017).
Ueda, T. & Shibuya, S. Synthesis of sulfur-bridged uracil anhydronucleosides. Chem. Pharm. Bull. 18, 1076–1078 (1970).
Hampton, A. & Nichol, A. Nucleotides. V. Purine ribonucleoside 2′,3′-cyclic carbonates. Preparation and use for the synthesis of 5′-monosubstituted nucleosides. Biochemistry 5, 2076–2082 (1966).
Russell, A. F., Prystasz, M., Hamamura, E. K., Verheyden, J. P. & Moffatt, J. G. Reactions of 2-acyloxyisobutyryl halides with nucleosides. V. Reactions with cytidine and its derivatives. J. Org. Chem. 39, 2182–2186 (1974).
Gibard, C., Bhowmik, S., Karki, M., Kim, E. K. & Krishnamurthy, R. Phosphorylation, oligomerization and self-assembly in water under potential prebiotic conditions. Nat. Chem. 10, 212–217 (2018).
Belenʹkiĭ, L. I. Chemistry of Organosulfur Compounds: General Problems (Ellis Horwood, New York, 1990).
Larock, R. C. Comprehensive Organic Transformations: A Guide to Functional Group Preparations 3rd edn (Wiley, New York, 2018).
Reichard, P. From RNA to DNA, why so many ribonucleotide reductases? Science 260, 1773–1777 (1993).
Bradbury, E. & Nagyvary, J. Preparation of 2′-thio-2′-deoxycytidine 2′-3′-phosphorothioate. Nucleic Acids Res. 3, 2437–2442 (1976).
Patel, A. D., Schrier, W. H. & Nagyvary, J. Synthesis and properties of 2′-deoxy-2′-thiocytidine. J. Org. Chem. 45, 4830–4834 (1980).
Wolman, Y. (ed.) Origin of life. In Proc. 3rd ISSOL Meeting and 6th ICOL Meeting 201–208 (D. Reidel, 1981)..
Divakar, K. J., Mottoh, A., Reese, C. B. & Sanghvi, Y. S. Approaches to the synthesis of 2′-thio analogues of pyrimidine ribosides. J. Chem. Soc. Perkin Trans. 1 0, 969–974 (1990).
Powner, M. W., Zheng, S. L. & Szostak, J. W. Multicomponent assembly of proposed DNA precursors in water. J. Am. Chem. Soc. 134, 13889–13895 (2012).
Powner, M. W., Gerland, B. & Sutherland, J. D. Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions. Nature 459, 239–242 (2009).
Ritson, D. J. & Sutherland, J. D. Conversion of biosynthetic precursors of RNA to those of DNA by photoredox chemistry. J. Mol. Evol. 78, 245–250 (2014).
Rimmer, P. B. et al. The origin of RNA precursors on exoplanets. Sci. Adv. 4, eaar3302 (2018).
Kuimelis, R. G., Hope, H. & Nambiar, K. P. A stereoselective synthesis of α- and β-2′-deoxy-2-thiouridine. Nucleos. Nucleot. Nucl. 12, 737–755 (1993).
Johnson, R., Reese, C. B. & Peizhuo, Z. Lability of glycosidic linkages of 2′-thio-ribonucleosides. Tetrahedron 51, 5093–5098 (1995).
Ritson, D. J. & Sutherland, J. D. Synthesis of aldehydic ribonucleotide and amino acid precursors by photoredox chemistry. Angew. Chem. Int. Ed. 52, 5845–5847 (2013).
Sauer, M. C., Crowell, R. A. & Shkrob, I. A. Electron photodetachment from aqueous anions. 1. Quantum yields for generation of hydrated electron by 193 and 248 nm laser photoexcitation of miscellaneous inorganic anions. J. Phys. Chem. A 108, 5490–5502 (2004).
Ritson, D. & Sutherland, J. D. Prebiotic synthesis of simple sugars by photoredox systems chemistry. Nat. Chem. 4, 895–899 (2012).
Unrau, P. J. & Bartel, D. P. RNA-catalysed nucleotide synthesis. Nature 395, 260–263 (1998).
Chapple, K. E., Bartel, D. P. & Unrau, P. J. Combinatorial minimization and secondary structure determination of a nucleotide synthase ribozyme. RNA 9, 1208–1220 (2003).
Lau, M. W., Cadieux, K. E. & Unrau, P. J. Isolation of fast purine nucleotide synthase ribozymes. J. Am. Chem. Soc. 126, 15686–15693 (2004).
Miyaki, M., Saito, A. & Shimizu, B. N→N alkyl and glycosyl migrations of purines and pyrimidines. IV. Trans-glycosylation from pyrimidines to purines. (A novel synthetic method of purine nucleosides and nucleotides). Chem. Pharm. Bull. 18, 2459–2468 (1970).
Vorbrüggen, H. & Ruh-Pohlenz, C. Handbook of Nucleoside Synthesis (Wiley, New York, 2001).
Nam, I., Nam, H. G. & Zare, R. N. Abiotic synthesis of purine and pyrimidine ribonucleosides in aqueous microdroplets. Proc. Natl Acad. Sci. USA 115, 36–40 (2018).
Fuller, W. D., Orgel, L. E. & Sanchez, R. A. Studies in prebiotic synthesis: VII. Solid-state synthesis of purine nucleosides. J. Mol. Evol. 1, 249–257 (1972).
Acknowledgements
This work was supported by the Medical Research Council (number MC_UP_A024_1009) and grants from the Simons Foundation (number 290362 to J.D.S. and number 327124 to R.K.). We acknowledge P. B. Rimmer for discussions on the photochemical reactor setup.
Author information
Authors and Affiliations
Contributions
J.D.S. and R.K. supervised the experimental research. J.X., N.J.G. and C.G. performed the experiments. All of the authors co-wrote the paper and assembled the Supplementary Information.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
All experimental procedures, characterization data (NMR, MS, UV and HPLC) for compounds and additional data referenced in the manuscript.
Rights and permissions
About this article
Cite this article
Xu, J., Green, N.J., Gibard, C. et al. Prebiotic phosphorylation of 2-thiouridine provides either nucleotides or DNA building blocks via photoreduction. Nat. Chem. 11, 457–462 (2019). https://doi.org/10.1038/s41557-019-0225-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-019-0225-x
This article is cited by
-
Biogeochemical explanations for the world’s most phosphate-rich lake, an origin-of-life analog
Communications Earth & Environment (2024)
-
Combined effects of copper and cadmium exposure on ovarian function and structure in Nile Tilapia (Oreochromis niloticus)
Ecotoxicology (2024)
-
Physical non-equilibria for prebiotic nucleic acid chemistry
Nature Reviews Physics (2023)
-
Synthesis and applications of cyclonucleosides: an update (2010–2023)
Molecular Diversity (2023)
-
2,6-diaminopurine promotes repair of DNA lesions under prebiotic conditions
Nature Communications (2021)