Abstract
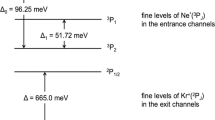
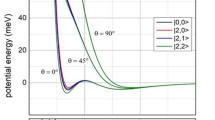
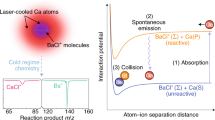
A prerequisite to gain a complete understanding of the most basic aspects of chemical reactions is the ability to perform experiments with complete control over the reactant degrees of freedom. By controlling these, details of a reaction mechanism can be investigated and ultimately manipulated. Here, we present a study of chemi-ionization—a fundamental energy-transfer reaction—under completely controlled conditions. The collision energy of the reagents was tuned from 0.02 K to 1,000 K, with the orientation of the excited Ne atom relative to Ar fully specified by an external magnetic field. Chemi-ionization of Ne(3P2) and Ar in these conditions enables a detailed investigation of how the reaction proceeds, and provides us with a means to control the branching ratio between the two possible reaction outcomes. The merged-beam experimental technique used here allows access to a low-energy regime in which the atoms dynamically reorient into a favourable configuration for reaction, irrespective of their initial orientations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Dulieu, O. & Osterwalder, A. Cold Chemistry. Molecular Scattering and Reactivity Near Absolute Zero (Royal Society of Chemistry, Cambridge, 2018).
Brouard, M., Parker, D. H. & van de Meerakker, S. Taming molecular collisions using electric and magnetic fields. Chem. Soc. Rev. 43, 7279–7294 (2014).
Onvlee, J. et al. Imaging quantum stereodynamics through Fraunhofer scattering of NO radicals with rare-gas atoms. Nat. Chem. 9, 226–233 (2017).
Henson, A. B., Gersten, S., Shagam, Y., Narevicius, J. & Narevicius, E. Observation of resonances in Penning ionization reactions at sub-kelvin temperatures in merged beams. Science 338, 234–238 (2012).
Jankunas, J., Bertsche, B., Jachymski, K., Hapka, M. & Osterwalder, A. Dynamics of gas phase Ne*+ NH3 and Ne*+ ND3 Penning ionisation at low temperatures. J. Chem. Phys. 140, 244302 (2014).
Dong, W., Mukherjee, N. & Zare, R. N. Optical preparation of H2 rovibrational levels with almost complete population transfer. J. Chem. Phys. 139, 074204 (2013).
Amarasinghe, C. & Suits, A. G. Intrabeam scattering for ultracold collisions. J. Phys. Chem. Lett. 8, 5153–5159 (2017).
Gustafsson, M. et al. Observing the stereodynamics of chemical reactions using randomly oriented molecular beams. J. Chem. Phys. 124, 241105 (2006).
Chefdeville, S. et al. Observation of partial wave resonances in low-energy O2–H2 inelastic collisions. Science 341, 1094–1096 (2013).
De Miranda, M. et al. Controlling the quantum stereodynamics of ultracold bimolecular reactions. Nat. Phys. 7, 502 (2011).
Stuhl, B. K., Hummon, M. T. & Ye, J. Cold state-selected molecular collisions and reactions. Ann. Rev. Phys. Chem. 65, 501–518 (2014).
Ospelkaus, S. et al. Quantum-state controlled chemical reactions of ultracold potassium–rubidium molecules. Science 327, 853–857 (2010).
Lavert-Ofir, E. et al. Observation of the isotope effect in sub-kelvin reactions. Nat. Chem. 6, 332–335 (2014).
Perreault, W. E., Mukherjee, N. & Zare, R. N. Quantum control of molecular collisions at 1 kelvin. Science 358, 356–359 (2017).
Wu, X. et al. A cryofuge for cold-collision experiments with slow polar molecules. Science 358, 645–648 (2017).
Nichols, B. et al. Steric effects and quantum interference in the inelastic scattering of NO(X)+Ar. Chem. Sci. 6, 2202–2210 (2015).
Barnwell, J., Loeser, J. & Herschbach, D. Angular correlations in chemical reactions. Statistical theory for four-vector correlations. J. Phys. Chem. 87, 2781–2786 (1983).
Pan, H., Wang, F., Czakó, G. & Liu, K. Direct mapping of the angle-dependent barrier to reaction for Cl + CHD3 using polarized scattering data. Nat. Chem. 9, 1175–1180 (2017).
Wang, F., Lin, J.-S. & Liu, K. Steric control of the reaction of CH stretch–excited CHD3 with chlorine atom. Science 331, 900–903 (2011).
Aoiz, F. J. et al. A new perspective: imaging the stereochemistry of molecular collisions. Phys. Chem. Chem. Phys. 17, 30210–30228 (2015).
Kasai, T., Stolte, S., Chandler, D. & González Ureña, A. Stereodynamics of chemical reactions. Eur. Phys. J. D 38, 1–2 (2006).
Siska, P. E. Molecular-beam studies of Penning ionization. Rev. Mod. Phys. 65, 337–412 (1993).
Gordon, S. D. S., Zou, J., Tanteri, S., Jankunas, J. & Osterwalder, A. Energy dependent stereodynamics of the Ne(3P2) +Ar reaction. Phys. Rev. Lett. 119, 053001 (2017).
Zou, J., Gordon, S. D. S., Tanteri, S. & Osterwalder, A. Stereodynamics of N(3P2) reacting with Ar, Kr, Xe, and N2. J. Chem. Phys. 148, 164310 (2018).
Arango, C. A., Shapiro, M. & Brumer, P. Cold atomic collisions: coherent control of Penning and associative ionization. Phys. Rev. Lett. 97, 193202 (2006).
Arango, C. A., Shapiro, M. & Brumer, P. Coherent control of collision processes: Penning versus associative ionization. J. Chem. Phys. 125, 094315 (2006).
Illenberger, E. & Niehaus, A. Velocity dependence of total Penning ionization cross sections. Z. Phys. B 20, 33–41 (1975).
Bussert, W. et al. Ionizing collisions of laser-excited rare gas atoms. J. Phys. Colloq. 46, C1–199 (1985).
Niehaus, A. Penning ionization. Ber. Bunsenges. Phys. Chem. 77, 632–640 (1973).
Falcinelli, S., Rosi, M., Pirani, F., Stranges, D. & Vecchiocattivi, F. Measurements of ionization cross sections by molecular beam experiments: information content on the imaginary part of the optical potential. J. Phys. Chem. A 120, 5169–5174 (2016).
Hausamann, D. & Morgner, H. The heteronuclear rare gas ions: a simple model for the determination of the potential curves. Mol. Phys. 54, 1085 (1985).
Op De Beek, S. S., Driessen, J. & Kokkelmans, S. Ionization widths for Ne(3l)–Ar systems (l = s,p): application to ionization and intramultiplet mixing cross sections. Phys. Rev. A. 56, 2792–2805 (1997).
Aguilar-Navarro, A., Brunetti, B., Rosi, S., Vecchiocattivi, F. & Volpi, G. G. Velocity dependence of the cross section for Penning and associative ionization of argon atoms by metastable neon atoms. J. Chem. Phys. 82, 773–779 (1985).
Miller, W. H. Theory of Penning ionization. I. Atoms. J. Chem. Phys. 52, 3563–3572 (1970).
Zare, R. N. Angular Momentum: Understanding Spatial Aspects in Chemistry and Physics (Wiley-Interscience, New York, 1988).
Verheijen, M. J. & Beijerinck, H. C. W. State selected total Penning ionisation cross sections for the systems Ne*(3P0, 3P2)+ Ar, Kr, Xe and N2 in the energy range 0.06 < E 0 (eV) < 8.0. Chem. Phys. 102, 255–273 (1986).
Baudon, J. et al. Polarization effects in metastable neon atom (Ne(3P2)) on ground state neon atom collision at thermal energy. Chem. Phys. 145, 153–161 (1990).
Osterwalder, A. Merged neutral beams. EPJ Tech. Instrum. 2, 10 (2015).
Shagam, Y. & Narevicius, E. Sub-kelvin collision temperatures in merged neutral beams by correlation in phase-space. J. Phys. Chem. C 117, 22454–22461 (2013).
Even, U. The Even–Lavie valve as a source for high intensity supersonic beam. EPJ Tech. Instrum. 2, 17 (2015).
Driessen, J. P. J., Op De Beek, S. S., Somers, L. M. T., Beijerinck, H. C. W. & Verhaar, B. J. Autoionization widths for Ne(3s)–Ar and Ne(3p)–Ar collisions. Phys. Rev. A. 44, 167–185 (1991).
Gregor, R. & Siska, P. Differential elastic scattering of Ne(3s 3P2,0) by Ar, Kr, and Xe: Optical potentials and their orbital interpretation. J. Chem. Phys. 74, 1078–1092 (1981).
van de Meerakker, S. Y. T., Bethlem, H. L., Vanhaecke, N. & Meijer, G. Manipulation and control of molecular beams. Chem. Rev. 112, 4828–4878 (2012).
Watanabe, D., Ohoyama, H., Matsumura, T. & Kasai, T. Characterization of an oriented metastable atom source based on a magnetic hexapole. Eur. Phys. J. D 38, 219–223 (2006).
Jankunas, J., Reisyan, K. S., Rakitzis, T. P. & Osterwalder, A. Oriented O(3P2), Ne(3P2), and He(3S1) atoms emerging from a bent magnetic guide. Mol. Phys. 114, 245–252 (2016).
Acknowledgements
This work is funded by EPFL and the Swiss Science Foundation (project number 200021_165975). J.J.O. and P.B. acknowledge the support by Natural Sciences and Engineering Research Council Canada.
Author information
Authors and Affiliations
Contributions
The experiment was conceived by A.O. and S.D.S.G. The experiment and data analysis were performed by S.D.S.G. The potential surfaces and observables were calculated by J.J.O. and P.B. All the authors contributed to the interpretation of the data and discussion of the results. The manuscript was written by S.D.S.G., A.O. and J.J.O. with additional comments and contributions from all the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Supplementary Figures 1–6
Rights and permissions
About this article
Cite this article
Gordon, S.D.S., Omiste, J.J., Zou, J. et al. Quantum-state-controlled channel branching in cold Ne(3P2)+Ar chemi-ionization. Nature Chem 10, 1190–1195 (2018). https://doi.org/10.1038/s41557-018-0152-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-018-0152-2
This article is cited by
-
The topology of the reaction stereo-dynamics in chemi-ionizations
Communications Chemistry (2023)
-
Intermolecular interactions and the weakly bound precursor states of elementary physicochemical processes
Rendiconti Lincei. Scienze Fisiche e Naturali (2023)
-
Ultimate control in chemistry
Nature Chemistry (2021)
-
Nuclear spin conservation enables state-to-state control of ultracold molecular reactions
Nature Chemistry (2021)
-
Towards chemistry at absolute zero
Nature Reviews Chemistry (2021)