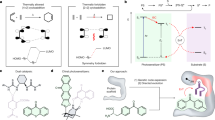
Abstract
Photosensitizers, which harness light energy to upgrade weak reductants to strong reductants, are pivotal components of the natural and artificial photosynthesis machineries. However, it has proved difficult to enhance and expand their functions through genetic engineering. Here we report a genetically encoded, 27 kDa photosensitizer protein (PSP), which facilitates the rational design of miniature photocatalytic CO2-reducing enzymes. Visible light drives PSP efficiently into a long-lived triplet excited state (PSP*), which reacts rapidly with reduced nicotinamide adenine dinucleotide to generate a super-reducing radical (PSP•), which is strong enough to reduce many CO2-reducing catalysts. We determined the three-dimensional structure of PSP• at 1.8 Å resolution by X-ray crystallography. Genetic engineering enabled the site-specific attachment of a nickel–terpyridine complex and the modular optimization of the photochemical properties of PSP, the chromophore/catalytic centre distance and the catalytic centre microenvironment, which culminated in a miniature photocatalytic CO2-reducing enzyme that has a CO2/CO conversion quantum efficiency of 2.6%.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Synthetic and experimental procedures, spectroscopic and mass spectrometric data, expression, purification and crystallization for X-ray diffraction as well as additional experiments are provided in the Supplementary Information. All other data are available from the authors upon request. Protein structures have been deposited to the Protein Data Bank under accession numbers 5YR2 and 5YR3.
References
Liu, J. et al. Metalloproteins containing cytochrome, iron–sulfur or copper redox centers. Chem. Rev. 114, 4366–4469 (2014).
Marshall, N. M. et al. Rationally tuning the reduction potential of a single cupredoxin beyond the natural range. Nature 462, 113–116 (2009).
Blankenship, R. E. et al. Comparing photosynthetic and photovoltaic efficiencies and recognizing the potential for improvement. Science 332, 805–809 (2011).
Romero, E., Novoderezhkin, V. I. & van Grondelle, R. Quantum design of photosynthesis for bio-inspired solar-energy conversion. Nature 543, 355–365 (2017).
Appel, A. M. et al. Frontiers, opportunities, and challenges in biochemical and chemical catalysis of CO2 fixation. Chem. Rev. 113, 6621–6658 (2013).
Sakimoto, K. K., Wong, A. B. & Yang, P. D. Self-photosensitization of nonphotosynthetic bacteria for solar-to-chemical production. Science 351, 74–77 (2016).
Liu, C., Colon, B. C., Ziesack, M., Silver, P. A. & Nocera, D. G. Water splitting-biosynthetic system with CO2 reduction efficiencies exceeding photosynthesis. Science 352, 1210–1213 (2016).
Schuchmann, K. & Muller, V. Direct and reversible hydrogenation of CO2 to formate by a bacterial carbon dioxide reductase. Science 342, 1382–1385 (2013).
Ort, D. R. et al. Redesigning photosynthesis to sustainably meet global food and bioenergy demand. Proc. Natl Acad. Sci. USA 112, 8529–8536 (2015).
Lewis, N. S. & Nocera, D. G. Powering the planet: chemical challenges in solar energy utilization. Proc. Natl Acad. Sci. USA 103, 15729–15735 (2006).
Gray, H. B. Powering the planet with solar fuel. Nat. Chem. 1, 7–12 (2009).
Rao, H., Schmidt, L. C. S., Bonin, J. & Robert, M. Visible-light-driven methane formation from CO2 with a molecular iron catalyst. Nature 548, 74–77 (2017).
Giepmans, B. N. G., Adams, S. R., Ellisman, M. H. & Tsien, R. Y. The fluorescent toolbox for assessing protein location and function. Science 312, 217–224 (2006).
Chattoraj, M., King, B. A., Bublitz, G. U. & Boxer, S. G. Ultra-fast excited state dynamics in green fluorescent protein: multiple states and proton transfer. Proc. Natl Acad. Sci. USA 93, 8362–8367 (1996).
Pearson, A. D. et al. Trapping a transition state in a computationally designed protein bottle. Science 347, 863–867 (2015).
Wang, L., Xie, J. & Schultz, P. G. Expanding the genetic code. Annu. Rev. Biophys. Biomol. 35, 225–249 (2006).
Breslow, R. et al. Remote oxidation of steroids by photolysis of attached benzophenone groups. J. Am. Chem. Soc. 95, 3251–3262 (1973).
Breslow, R., Kitabatake, S. & Rothbard, J. Photoreactions of charged benzophenone with amphiphiles in micelles and multicomponent aggregates as conformational probes. J. Am. Chem. Soc. 100, 8156–8160 (1978).
Braun, A. M. et al. Photochemical processes of benzophenone in microheterogeneous systems. J. Am. Chem. Soc. 103, 7312–7316 (1981).
Turro, N. J., Aikawa, M. & Gould, I. R. The laser vs the lamp—a novel laser-induced adiabatic reaction and luminescence of benzophenone. J. Am. Chem. Soc. 104, 856–858 (1982).
Kauer, J. C., Erickson-Viitanen, S., Wolfe, H. R.Jr. & DeGrado, W. F. p-Benzoyl-l-phenylalanine, a new photoreactive amino acid. Photolabeling of calmodulin with a synthetic calmodulin-binding peptide. J. Biol. Chem. 261, 10695–10700 (1986).
Connelly, N. G. & Geiger, W. E. Chemical redox agents for organometallic chemistry. Chem. Rev. 96, 877–910 (1996).
Hammes-Schiffer, S. & Stuchebrukhov, A. A. Theory of coupled electron and proton transfer reactions. Chem. Rev. 110, 6939–6960 (2010).
Li, H. & Zhang, M. T. Tuning excited-state reactivity by proton-coupled electron transfer. Angew. Chem. Int. Ed. 55, 13132–13136 (2016).
Rebelein, J. G., Stiebritz, M. T., Lee, C. C. & Hu, Y. Activation and reduction of carbon dioxide by nitrogenase iron proteins. Nat. Chem. Biol. 13, 147–149 (2017).
Dorman, G. & Prestwich, G. D. Benzophenone photophores in biochemistry. Biochemistry 33, 5661–5673 (1994).
Kuehnel, M. F., Orchard, K. L., Dalle, K. E. & Reisner, E. Selective photocatalytic CO2 reduction in water through anchoring of a molecular Ni catalyst on CdS nanocrystals. J. Am. Chem. Soc. 139, 7217–7223 (2017).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Shih, C. et al. Tryptophan-accelerated electron flow through proteins. Science 320, 1760–1762 (2008).
Song, W. J. & Tezcan, F. A. A designed supramolecular protein assembly with in vivo enzymatic activity. Science 346, 1525–1528 (2014).
Hsia, Y. et al. Design of a hyperstable 60-subunit protein icosahedron. Nature 540, 136–139 (2016).
Kan, S. B. J., Lewis, R. D., Chen, K. & Arnold, F. H. Directed evolution of cytochrome C for carbon–silicon bond formation: bringing silicon to life. Science 354, 1048–1051 (2016).
Blomberg, R. et al. Precision is essential for efficient catalysis in an evolved Kemp eliminase. Nature 503, 418–421 (2013).
Jeschek, M. et al. Directed evolution of artificial metalloenzymes for in vivo metathesis. Nature 537, 661–665 (2016).
Dydio, P. et al. An artificial metalloenzyme with the kinetics of native enzymes. Science 354, 102–106 (2016).
Joh, N. H. et al. De novo design of a transmembrane Zn2+-transporting four-helix bundle. Science 346, 1520–1524 (2014).
Acknowledgements
We are grateful for the financial support from the National Key Research and Development Program of China under awards 2017YFA0503704, 2016YFA0501502 and 2015CB856203; the National Science Foundation of China under awards 21750003, 91527302, U1632133, 31628004, 21473237, 31628004 and U1732264; Key Research Program of Frontier Sciences, CAS, grant numbers QYZDB-SSW-SMC032 and QYZDJ-SSW-SMC018; Tianjin Science and Technology grant 15PTCYSY00020; and Sanming Project of Medicine in Shenzhen (number SZSM201811092). We thank S. S. Zang for help with NMR spectra determination; Z. Xie for protein mass spectrometry; C. X. Zhang for ESR experiments; L. Xia for CV experiments; C. Wang and D. S. Liu for TEM experiments; J. H. Li for CD experiments and J. L. Jie for help with transient absorption spectroscopy experiments.
Author information
Authors and Affiliations
Contributions
J.Y.W. conceived the study and designed the experiments. X.H.L. performed most of the experiments, and together with J.Y.W. wrote the manuscript. F.Y.K., L.W. and D.D.Z. performed protein purification, crystallization and X-ray diffraction. C.H. and Z.X. synthesized small molecules and performed enzyme activity assays. Y.L., W.M.G. and Y.H.M. inspired the work and helped to revise the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Methods, Supplementary Figures, Supplementary Tables and Supplementary References
Rights and permissions
About this article
Cite this article
Liu, X., Kang, F., Hu, C. et al. A genetically encoded photosensitizer protein facilitates the rational design of a miniature photocatalytic CO2-reducing enzyme. Nature Chem 10, 1201–1206 (2018). https://doi.org/10.1038/s41557-018-0150-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-018-0150-4
This article is cited by
-
A light-driven enzymatic enantioselective radical acylation
Nature (2024)
-
Remote stereocontrol with azaarenes via enzymatic hydrogen atom transfer
Nature Chemistry (2024)
-
Photocatalytic CO2 reduction
Nature Reviews Methods Primers (2023)
-
Direct visible-light-excited flavoproteins for redox-neutral asymmetric radical hydroarylation
Nature Catalysis (2023)
-
A designed photoenzyme for enantioselective [2+2] cycloadditions
Nature (2022)