Abstract
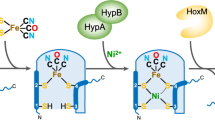
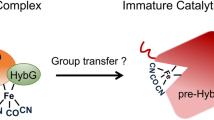
Biosynthesis of the [FeFe] hydrogenase active site (the 'H-cluster') requires the interplay of multiple proteins and small molecules. Among them, the radical S-adenosylmethionine enzyme HydG, a tyrosine lyase, has been proposed to generate a complex that contains an Fe(CO)2(CN) moiety that is eventually incorporated into the H-cluster. Here we describe the characterization of an intermediate in the HydG reaction: a [4Fe–4S][(Cys)Fe(CO)(CN)] species, 'Complex A', in which a CO, a CN− and a cysteine (Cys) molecule bind to the unique 'dangler' Fe site of the auxiliary [5Fe–4S] cluster of HydG. The identification of this intermediate—the first organometallic precursor to the H-cluster—validates the previously hypothesized HydG reaction cycle and provides a basis for elucidating the biosynthetic origin of other moieties of the H-cluster.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Vignais, P. M. & Billoud, B. Occurrence, classification, and biological function of hydrogenases: an overview. Chem. Rev. 107, 4206–4272 (2007).
Lubitz, W., Ogata, H., Rudiger, O. & Reijerse, E. Hydrogenases. Chem. Rev. 114, 4081–4148 (2014).
Barber, J. & Tran, P. D. From natural to artificial photosynthesis. J. R. Soc. Interface 10, 20120984 (2013).
Peters, J. W., Lanzilotta, W. N., Lemon, B. J. & Seefeldt, L. C. X-ray crystal structure of the Fe-only hydrogenase (CpI) from Clostridium pasteurianum to 1.8 ångström resolution. Science 282, 1853–1858 (1998).
Nicolet, Y., Piras, C., Legrand, P., Hatchikian, C. E. & Fontecilla-Camps, J. C. Desulfovibrio desulfuricans iron hydrogenase: the structure shows unusual coordination to an active site Fe binuclear center. Structure 7, 13–23 (1999).
Broderick, J. B. et al. H-cluster assembly during maturation of the [FeFe]-hydrogenase. J. Biol. Inorg. Chem. 19, 747–757 (2014).
Vincent, K. A., Parkin, A. & Armstrong, F. A. Investigating and exploiting the electrocatalytic properties of hydrogenases. Chem. Rev. 107, 4366–4413 (2007).
Suess, D. L. M., Kuchenreuther, J. M., de la Paz, L., Swartz, J. R. & Britt, R. D. Biosynthesis of the [FeFe] hydrogenase H cluster: a central role for the radical SAM enzyme HydG. Inorg. Chem. 55, 478–487 (2016).
Birrell, J. A., Rüdiger, O., Reijerse, E. J. & Lubitz, W. Semisynthetic hydrogenases propel biological energy research into a new era. Joule 1, 61–76 (2017).
Shepard, E. M. et al. [FeFe]-hydrogenase maturation. Biochemistry 53, 4090–4104 (2014).
Peters, J. W. et al. [FeFe]- and [NiFe]-hydrogenase diversity, mechanism, and maturation. Biochim. Biophys. Acta 1853, 1350–1369 (2015).
Mulder, D. W. et al. Insights into [FeFe]-hydrogenase structure, mechanism, and maturation. Structure 19, 1038–1052 (2011).
Boyer, M. E., Stapleton, J. A., Kuchenreuther, J. M., Wang, C. W. & Swartz, J. R. Cell-free synthesis and maturation of [FeFe] hydrogenases. Biotechnol. Bioeng. 99, 59–67 (2008).
Posewitz, M. C. et al. Discovery of two novel radical S-adenosylmethionine proteins required for the assembly of an active [Fe] hydrogenase. J. Biol. Chem. 279, 25711–25720 (2004).
McGlynn, S. E. et al. HydF as a scaffold protein in [FeFe] hydrogenase H-cluster biosynthesis. FEBS Lett. 582, 2183–2187 (2008).
Pilet, E. et al. The role of the maturase HydG in [FeFe]-hydrogenase active site synthesis and assembly. FEBS Lett. 583, 506–511 (2009).
Mulder, D. W. et al. Activation of HydAΔEFG requires a preformed [4Fe-4S] cluster. Biochemistry 48, 6240–6248 (2009).
Mulder, D. W. et al. Stepwise [FeFe]-hydrogenase H-cluster assembly revealed in the structure of HydAΔEFG. Nature 465, 248–251 (2010).
Shepard, E. M. et al. Synthesis of the 2Fe subcluster of the [FeFe]-hydrogenase H cluster on the HydF scaffold. Proc. Natl Acad. Sci. USA 107, 10448–10453 (2010).
Kuchenreuther, J. M., Britt, R. D. & Swartz, J. R. New insights into [FeFe] hydrogenase activation and maturase function. PLoS ONE 7, e45850 (2012).
Berto, P. et al. The [4Fe-4S]-cluster coordination of [FeFe]-hydrogenase maturation protein HydF as revealed by EPR and HYSCORE spectroscopies. Biochim. Biophys. Acta 1817, 2149–2157 (2012).
Kuchenreuther, J. M., George, S. J., Grady-Smith, C. S., Cramer, S. P. & Swartz, J. R. Cell-free H-cluster synthesis and [FeFe] hydrogenase activation: all five CO and CN– ligands derive from tyrosine. PLoS ONE 6, e20346 (2011).
Driesener, R. C. et al. [FeFe]-hydrogenase cyanide ligands derived from S-adenosylmethionine-dependent cleavage of tyrosine. Angew. Chem. Int. Ed. 49, 1687–1690 (2010).
Shepard, E. M. et al. [FeFe]-hydrogenase maturation: HydG-catalyzed synthesis of carbon monoxide. J. Am. Chem. Soc. 132, 9247–9249 (2010).
Kuchenreuther, J. M. et al. The HydG enzyme generates an Fe(CO)2(CN) synthon in assembly of the FeFe hydrogenase H-cluster. Science 343, 424–427 (2014).
Driesener, R. C. et al. Biochemical and kinetic characterization of radical S-adenosyl-l-methionine enzyme HydG. Biochemistry 52, 8696–8707 (2013).
Kuchenreuther, J. M. et al. A radical intermediate in tyrosine scission to the CO and CN− ligands of FeFe hydrogenase. Science 342, 472–475 (2013).
Rubach, J. K., Brazzolotto, X., Gaillard, J. & Fontecave, M. Biochemical characterization of the HydE and HydG iron-only hydrogenase maturation enzymes from Thermatoga maritima. FEBS Lett. 579, 5055–5060 (2005).
Nicolet, Y. et al. Crystal structure of HydG from Carboxydothermus hydrogenoformans: a trifunctional [FeFe]-hydrogenase maturase. ChemBioChem 16, 397–402 (2015).
Pagnier, A., Martin, L., Zeppieri, L., Nicolet, Y. & Fontecilla-Camps, J. C. CO and CN− syntheses by [FeFe]-hydrogenase maturase HydG are catalytically differentiated events. Proc. Natl Acad. Sci. USA 113, 104–109 (2016).
Nicolet, Y., Zeppieri, L., Amara, P. & Fontecilla-Camps, J. C. Crystal structure of tryptophan lyase (NosL): evidence for radical formation at the amino group of tryptophan. Angew. Chem. Int. Ed. 53, 11840–11844 (2014).
Dinis, P. et al. X-ray crystallographic and EPR spectroscopic analysis of HydG, a maturase in [FeFe]-hydrogenase H-cluster assembly. Proc. Natl Acad. Sci. USA 112, 1362–1367 (2015).
Suess, D. L. M. et al. Cysteine as a ligand platform in the biosynthesis of the FeFe hydrogenase H cluster. Proc. Natl. Acad Sci. USA 112, 11455–11460 (2015).
Suess, D. L. M. et al. The radical SAM enzyme HydG requires cysteine and a dangler iron for generating an organometallic precursor to the [FeFe]-hydrogenase H-cluster. J. Am. Chem. Soc. 138, 1146–1149 (2016).
Morton, J. R. & Preston, K. F. Atomic parameters for paramagnetic resonance data. J. Magn. Reson. 30, 577–582 (1978).
Lanz, N. D. et al. Characterization of a radical intermediate in lipoyl cofactor biosynthesis. J. Am. Chem. Soc. 137, 13216–13219 (2015).
Silakov, A., Reijerse, E. J., Albracht, S. P., Hatchikian, E. C. & Lubitz, W. The electronic structure of the H-cluster in the [FeFe]-hydrogenase from Desulfovibrio desulfuricans: a Q-band 57Fe-ENDOR and HYSCORE study. J. Am. Chem. Soc. 129, 11447–11458 (2007).
Gilbert-Wilson, R. et al. Spectroscopic investigations of [FeFe] hydrogenase maturated with [57Fe2(adt)(CN)2(CO)4]2–. J. Am. Chem. Soc. 137, 8998–9005 (2015).
Carepo, M. et al. 17O ENDOR detection of a solvent-derived Ni-(OHx)-Fe bridge that is lost upon activation of the hydrogenase from Desulfovibrio gigas. J. Am. Chem. Soc. 124, 281–286 (2002).
Pandelia, M. E., Ogata, H. & Lubitz, W. Intermediates in the catalytic cycle of [NiFe] hydrogenase: functional spectroscopy of the active site. ChemPhysChem 11, 1127–1140 (2010).
Christner, J. A., Janick, P. A., Siegel, L. M. & Munck, E. Mössbauer studies of Escherichia coli sulfite reductase complexes with carbon monoxide and cyanide. Exchange coupling and intrinsic properties of the [4Fe-4S] cluster. J. Biol. Chem. 258, 11157–11164 (1983).
Krueger, R. J. & Siegel, L. M. Spinach siroheme enzymes: isolation and characterization of ferredoxin-sulfite reductase and comparison of properties with ferredoxin-nitrite reductase. Biochemistry 21, 2892–2904 (1982).
Krueger, R. J. & Siegel, L. M. Evidence for siroheme–Fe4S4 interaction in spinach ferredoxin-sulfite reductase. Biochemistry 21, 2905–2909 (1982).
Winkler, M. et al. Accumulating the hydride state in the catalytic cycle of [FeFe]-hydrogenases. Nat. Commun. 8, 16115 (2017).
Mulder, D. W., Guo, Y., Ratzloff, M. W. & King, P. W. Identification of a catalytic iron-hydride at the H-cluster of [FeFe]-hydrogenase. J. Am. Chem. Soc. 139, 83–86 (2017).
Reijerse, E. J. et al. Direct observation of an iron-bound terminal hydride in [FeFe]-hydrogenase by nuclear resonance vibrational spectroscopy. J. Am. Chem. Soc. 139, 4306–4309 (2017).
Acknowledgements
We thank J. Swartz from Stanford University for providing the E. coli strain overexpressing SoHydG. This work is supported by National Institute of Health (GM104543). Correspondence and requests for materials should be addressed to R.D.B. The authors declare no competing financial interests.
Author information
Authors and Affiliations
Contributions
G.R., L.T., D.L.M.S. and R.D.B. designed the experiments. G.R., L.T. and D.L.M.S. performed the experiments and analysed the data. All the authors contributed to writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Table 1, Spectra, Figures and Notes
Rights and permissions
About this article
Cite this article
Rao, G., Tao, L., Suess, D.L.M. et al. A [4Fe–4S]-Fe(CO)(CN)-l-cysteine intermediate is the first organometallic precursor in [FeFe] hydrogenase H-cluster bioassembly. Nature Chem 10, 555–560 (2018). https://doi.org/10.1038/s41557-018-0026-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-018-0026-7
This article is cited by
-
A personal account on 25 years of scientific literature on [FeFe]-hydrogenase
JBIC Journal of Biological Inorganic Chemistry (2023)
-
Bioassembly of complex iron–sulfur enzymes: hydrogenases and nitrogenases
Nature Reviews Chemistry (2020)
-
[FeFe]-hydrogenase maturation: H-cluster assembly intermediates tracked by electron paramagnetic resonance, infrared, and X-ray absorption spectroscopy
JBIC Journal of Biological Inorganic Chemistry (2020)