Abstract
We performed an independent comparison of neutralizing and cross-neutralizing antibody (ab) levels seven months after initiation of three-dose, six-month vaccination schedules with the bivalent and quadrivalent human papillomavirus (HPV) vaccines in adolescent Finnish and Indian females, respectively. We used a semi-automated Pseudovirion-Based Neutralization Assay and observed significantly higher HPV16/18 peak ab-levels in bivalent as compared to quadrivalent vaccine recipients. Bivalent vaccine induced cross-neutralizing HPV31/33/45/52/58 antibodies significantly more frequently and to higher levels than the quadrivalent vaccine. The correlation of bivalent vaccine-induced HPV45 ab-levels with HPV16/18 ab-levels was stronger than that of corresponding quadrivalent vaccine-induced ab-levels, suggesting a qualitatively different cross-reactive response. Our findings on the comparison of the immunogenicity of two HPV vaccine tested in two different populations indicate that further head-to-head studies are warranted.
Similar content being viewed by others
Introduction
Differences in the breadth of cross-protective vaccine efficacies (VE) of prophylactic bivalent and quadrivalent human papillomavirus (HPV) virus-like particle (VLP) vaccines against infections with high-risk (hr) HPV types and associated neoplasia are well established. These extend to significantly different vaccine-specific overall efficacies against associated high-grade squamous intraepithelial neoplasia irrespective of HPV type.1,2 The considerable overlap, with regard to age, ethnicity and sexual risk-taking behavior of different female target populations in the major clinical phase III trials involving the two vaccines2,3, suggests that the differences in efficacy most likely are vaccine-specific.1,2,3,4
Neutralizing antibodies induced upon VLP vaccination have been suggested to be the primary mechanism in mediating protection from HPV infection.5 The impact of HPV cross-neutralizing antibodies in promoting cross-protection against non-vaccine types is, however, uncertain. Independent studies, considering early adolescent girls or HIV-positive individuals, have demonstrated that cross-neutralizing antibodies induced by the bivalent vaccine confer wider cross-neutralizing antibody response and wider protection against cervical hrHPV infections and associated intraepithelial lesions than the quadrivalent vaccine.6,7,8 Also an independent study on vaccine-induced antibody sustainability in adolescent females found that bivalent vaccine-induced total HPV16 and HPV18 L1-VLP binding antibody levels were 5- and 18-fold (respectively) higher than those for the quadrivalent vaccine up to 12 years post vaccination.9
The launching of a nonavalent HPV6/11/16/18/31/33/45/52/58 VLP vaccine to the market raises two pivotal questions for public health decision makers in respect to national vaccination programs: (1) how broad is the cross-neutralization ability of the bivalent vaccine-induced antibodies? (2) How sustainable are the quadri/nonavalent vs. bivalent vaccine-induced neutralizing and cross-neutralizing antibodies?
In this report, we particularly address the extent and type-specific pattern of cross-neutralization induced by two different HPV vaccines. Cross-neutralizing antibody titers are always substantially lower than titers against vaccine types. Specifically, we investigated peak (seven months post vaccination) neutralizing antibody titers induced by the bivalent and quadrivalent vaccines to HPV types 6/16/18/31/33/45/52/58 in adolescent Finnish and Indian women, respectively, to reveal the breadth of the cross-neutralizing antibody responses of bivalent versus multivalent vaccines.
Results
Neutralizing antibody levels and seroprevalence
All vaccinated study participants at Month 7 showed neutralizing antibodies to HPV types 16 and 18 (Fig. 1a) shared by both vaccines. Bivalent/Finnish and quadrivalent/Indian vaccine recipients differed in median titers (166,681 (5,373 IU/ml) versus 46,400 (1,495 IU/ml) for HPV16 and 57,369 (1,599 IU/ml) versus 8859 (247 IU/ml) for HPV18) and percentage of sera with titers >180,000 (47% versus 6% for HPV16, and 20% versus 0% for HPV18). Without correction for titers >180,000, geometric mean neutralization titers against HPV16 and HPV18 were, respectively, 2.7- and 6.9-fold higher in the bivalent/Finnish vaccine recipients than in the quadrivalent/Indian vaccine recipients (Fig. 1b). Notably, the vaccine-induced median HPV16 antibody titer of 166,681 in the bivalent/Finnish vaccine recipients was above the upper 95% confidence limit of the HPV16 GMT in the quadrivalent/Indian vaccine recipients.
a Percentage of vaccine recipients with neutralizing antibody titers of >40 to vaccine HPV types ((6)/16/18 and non-vaccine HPV types (6)/31/33/45/52/58). Bars indicate boundaries of 95% confidence interval (CI). Sera of quadrivalent (Gardasil, 6/11/16/18) and bivalent (Cervarix, 16/18) vaccine recipients are shown in blue and orange columns, respectively. b Neutralizing peak (Month 7) antibody levels (Geometric mean titer, GMT) in neutralization-positive samples. The gray dots represent the EC50 values of one serum. Serum concentrations inhibiting 50% of the PsV infection (EC50 values) were calculated from median of triplicates.
Antibodies cross-neutralizing HPV45 and HPV52, respectively, were found more frequently (18% vs 84%, and 25% vs 84%) (Fig. 1a) and at 2- to 3-fold higher titers in the bivalent/Finnish vaccine recipients as compared to the quadrivalent/Indian vaccine recipients (Fig. 1b). Although HPV31 seroprevalence in bivalent/Finnish vaccine recipients (99.0%) was not much higher than that observed in quadrivalent/Indian vaccine recipients (87.6%) (Fig. 1a), the cross-neutralizing HPV31 antibody titers induced by the bivalent vaccine were 4.1-fold higher than those induced by the quadrivalent vaccine (Fig. 1b). Contrarily, the seroprevalence observed to HPV33 and HPV58 in bivalent/Finnish vaccine recipients was 2.4- to 3.5-fold higher than in quadrivalent/Indian vaccine recipients, although cross-neutralizing antibody levels were not much different.
HPV6 is part of the quadrivalent vaccine but absent in the bivalent vaccine. All quadrivalent/Indian vaccine recipient sera neutralized HPV6. In the bivalent/Finnish vaccine recipients we observed cross-neutralization of HPV6 (Fig. 1a) but with a significantly lower prevalence (57.2%). The GMT of quadrivalent vaccine-induced neutralizing HPV6 antibodies was 407-fold higher than that of the cross-neutralizing HPV6 antibodies induced by the bivalent vaccine (Fig. 1b).
For both vaccines, higher vaccine HPV type antibody levels were associated with more frequent and stronger cross-neutralization of non-vaccine HPV types. Ranked correlation of the neutralizing antibody levels to HPV16/45 (rs = 0.53, 95% CI 0.37–0.66), and especially HPV18/45 (rs = 0.61, 95% CI 0.47–0.72) was twice as strong in the bivalent/Finnish (Fig. 2a, b) than in the quadrivalent/Indian vaccine recipients (rs = 0.34, 95% CI 0.16–0.49 and rs = 0.28, 95% CI 0.10–0.44, Fig. 2c, d) even if the exact bivalent vaccine-induced HPV16 or HPV18 antibody levels were not always estimable. On the contrary, comparable correlation coefficients of HPV16/31 and HPV18/31 neutralizing antibodies were observed for both the bivalent vaccine (rs = 0.66, 95% CI 0.53–0.75; and rs = 0.68, 95% CI 0.56–0.77, Fig. 2e, f) and for the quadrivalent vaccine (rs = 0.61, 95% CI 0.49–0.72; and 0.54, 95% CI 0.40–0.66, Fig. 2g, h). All ranked correlations of the neutralizing antibody levels are provided in the Supplementary Table.
EC50 against HPV16/45 (a and c), HPV18/45 (b and d), HPV16/31 (i and g) and HPV18/31 (f and h) induced in the bivalent/Finnish and the quadrivalent/Indian vaccine recipients. Dotted lines indicate the lowest detectable neutralization titer (EC50 value of 41), and non-neutralizing titers (EC50 from 10–30) were arbitrarily set to 12 or 25 and boxed in the gray rectangle. Serum concentrations inhibiting 50% of the PsV infection (EC50 values) were calculated from median of triplicates.
Discussion
We found significantly higher peak (Month 7) neutralizing HPV16 and HPV18 antibody levels in Finnish adolescent females, who received three doses of the bivalent vaccine, than in corresponding Indian quadrivalent vaccine recipients. We also found significantly increased seroprevalence to HPV33, HPV45, HPV52 and HPV58, and higher cross-neutralizing HPV31, HPV45 and HPV52 antibody titers in the bivalent/Finnish vaccine recipients as compared to the quadrivalent/Indian vaccine recipients.
Our findings on distinguishable cross-neutralizing antibody responses to HPV31/33/45/52/58 in adolescent women receiving the bivalent vaccine are in line with the earlier observations on the induction of wide cross-neutralizing peak antibody responses by the bivalent vaccine in adolescents [6]. The majority of the quadrivalent/Indian vaccine recipients had cross-neutralizing antibodies to HPV31 only. Moreover, strong correlation values were observed only between HPV16 and HPV31, and HPV18 and HPV31 in the quadrivalent/Indian vaccine recipients. Similar strong correlation between HPV16 and HPV31, HPV16 and HPV45, HPV18 and HPV31, and HPV18 and HPV45 neutralizing antibody levels was induced by the bivalent vaccine.
The cross-neutralizing antibody response induced by the two vaccines is largely the result of structurally comparable conformational epitopes shared among phylogenetically related HPV types.10 The fact that the bivalent/Finnish vaccine recipients presented stronger cross-neutralizing antibody responses, however, is intriguing. Probably a certain amount of antibodies differently induced by the presented adjuvants, a threshold level apparently between the bivalent and quadrivalent vaccine-induced ones, needs to be reached in order for the cross-neutralizing antibodies to be detectable. On the other hand, the low cross-reactive HPV45 antibody levels and the low correlation both between HPV16 and HPV45, and HPV18 and HPV45 antibodies in the quadrivalent vaccine recipients also suggest a qualitative difference. Presumably this might arise from better exposed cross-neutralizing epitopes on the bivalent vaccine VLPs vs. the quadrivalent vaccine VLPs, respectively produced in insect cells vs. yeast cells.5 Nevertheless, while the correlation data (Fig. 2 and Supplementary Table) implies that the same antibodies probably are able to mediate neutralization of two phylogenetically more or less related HPV types, this is particularly difficult to firmly state in polyclonal sera. It might be that boosting of low-level responses to alternate types occurs, but the type-specific antibodies are still narrowly reactive. The analysis of panels of neutralizing monoclonal antibodies would be required to conclusively explore this scenario.
Three main serological assays have been explored to validate immunogenicity of the current HPV vaccines in clinical trials and further monitoring the L1 VLP antibody responses in observational studies: antibody competition assays, L1 and L1-VLP-IgG binding assays and pseudovirion-based neutralization assays (PBNA), the latter of which should be considered the reference standard for assessing protective antibody responses induced by the vaccines, as it measures a pertinent biological antibody activity.11 The High-Throughput-Pseudovirion-Based Neutralization Assay (HT-PBNA) deployed here represents the most sophisticated and sensitive assay, which allows the simultaneous testing of larger numbers of samples by minimizing the complexity inherent to the manual neutralization assay, previously employed for HPV vaccine studies.12 Although in line with reports on differences in the HPV type-specific overall VE, the different GMT ratios presented here do not necessarily reflect differences between the two vaccines in protection against HPV infection. This strongly indicates that a head-to-head comparison of the sustainability of the neutralizing and cross-neutralizing antibody levels and protection induced by both vaccines is warranted.9
There are two relevant limitations to the evidence presented in our study. Firstly, two different cohorts were available for this independent study to investigate the peak neutralizing and cross-neutralizing antibody titers induced by the two HPV vaccines. It is arguable whether unknown environmental factors or even the different ethnicities related to each cohort here studied might have differentially influenced the antibody response. Despite of that, differences in bivalent and quadrivalent vaccine immunogenicity among adolescent women from geographically different regions, including Finland and India, were not reported by previous clinical trials.9,13,14,15,16 Secondly, given that the endpoint titers for many of bivalent/Finnish recipients were not accessed, as they reached values >180,000, the reported higher HPV16/18 antibody levels induced in the bivalent/Finnish recipients are under-estimations of the true difference in antibody response. Nevertheless, our conservative estimations oversee the need to investigate the cross-neutralizing properties of the two vaccines in further details.
The significantly different levels of quadrivalent and bivalent vaccine induced cross-neutralizing HPV31 antibodies are associated with reportedly comparable, high VEs against HPV31.1 On the other hand, the low levels of cross-neutralizing HPV33 and HPV45 antibodies confer moderate to high protection (>75% VE) only in the bivalent vaccine recipients. Our data also fit recent reports on VE from the Scotland, where significantly protective VEs against HPV31, HPV33, and HPV45 persistent infections, were observed among participants in a cervical cancer-screening program seven years after bivalent vaccination.17,18 Comparable VEs against HPV31, HPV45, HPV52, and HPV58 have also been reported in adolescent Japanese and Dutch girls vaccinated with the bivalent vaccine.17,19,20
The differences between the high neutralizing HPV6 antibody levels and high VE induced by the quadrivalent vaccine as compared to those induced by the bivalent vaccine (cross-neutralizing HPV6 antibody levels and low VE) are notable.1,6 It is surprising that many of the bivalent/Finnish recipients show cross-neutralizing antibodies to HPV6—a phylogenetically distant genotype to the vaccine incorporated ones. We think that, occasionally, the very low GMT to HPV6 (Fig. 2b) could have been triggered by natural HPV6/11 infection.
Correlates of protection provided by the vaccines, e.g., the threshold of antibody level that confers immunity against HPV infection, have been difficult to identify due to the high vaccine-specific VEs. Understanding the differences in the cross-neutralizing antibodies and cross-protection induced by the vaccines may ultimately help to identify protective vaccine-induced antibody levels. In view of recent studies on HPV VE upon a single-dose schedule, it may be critically important to follow-up the antibody titers over time by vaccine (bivalent vs multivalent) and by vaccine dose (1–3). The different adjuvants and number of doses are fundamentally linked to vaccine dissemination and uptake. This, however, was not the scope of our current study.
In conclusion, peak neutralizing antibody levels induced by the bivalent vaccine are 2- to 7-times higher in adolescent women for high-risk HPV types 16/18/31/33/45/52/58 than those induced by the quadrivalent vaccine. Notably, our data represent an initial preliminary study that requires confirmation in a head-to-head study throughout. Considering that the recently licensed nonavalent vaccine targets additional types associated with virtually all cervical cancer cases, independent post-licensure studies on protection and cross-protection are important to assure best vaccine effectiveness and health economic impact from the vaccine to be chosen for a national vaccination program.
Methods
Participants
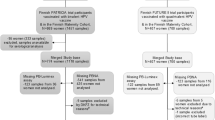
The Finnish post vaccination (Month 7) serum samples were collected following an informed consent in conjunction with the randomized controlled PATRICIA trial.2,21 A total of 4,808 adolescent, female, PATRICIA trial participants received either the AS04-adjuvanted bivalent HPV16/18 vaccine (GlaxoSmithKline Biologicals, Rixensart, Belgium) or a control hepatitis A vaccine (GSK Biologicals). A random sample of 103 originally 16–17 year-old recipients of three doses (Months 0, 1, and 6) of the HPV16/18 vaccine was selected out of 2,409 HPV16/18 vaccinated participants.2,21 In this work, subjects from this cohort are referred as bivalent vaccine/Finnish recipients.
The Indian post vaccination (Month 7) serum samples were obtained from a multicentric trial, to investigate the effectiveness of two and three doses of quadrivalent HPV vaccine inducing non-inferior immune response in girls aged 10–18 years.22 The originally planned cluster-randomized trial initiated vaccination on 1 September 2009 and was converted to an observational study due to suspension of vaccination midway by a Governmental order on 8 April 2010 due to reasons unrelated to the study. We selected a random sample of 120 originally 15–18-year-old recipients of three doses of the HPV6/11/16/18 vaccine (Months 0, 2, and 6) out of 4,348 vaccinated female trial participants and representing all study sites. In this work, subjects from this cohort are referred as quadrivalent vaccine/Indian recipients.
Ethics statement
The NCT01393470 trial was reviewed and approved by the Finnish National Ethical Review Board, ETENE/Tukija. The NCT00923702 trial was reviewed and approved by ethics review committee in India (Health Ministry Screening Committee of Government of India) and the IARC Institutional Review Board. Written informed consent from all participants were obtained at the time of the Finnish clinical trial. For the Indian clinical trial, written informed consent was obtained from one of the parents, or legal guardian, along with the assent of the participating girl.
Pseudovirion-based neutralization assay
We determined neutralizing antibodies by a semi-automated and High-Throughput-Pseudovirion-Based Neutralization Assay (HT-PBNA) for HPV6/16/18/31/33/45/52/58.23 Briefly, the assay uses pseudovirions (PsV) made of HPV L1 and L2 proteins, which in turn encapsidate a Gaussia-luciferase reporter plasmid. After production in HEK293TT cells and purification by ultracentrifugation in an Optiprep gradient, these particles are able to bind to and enter in HeLaT cells, allowing transduction of the reporter gene. PsV infection and consequent Gaussia-luciferase activity are measured by luminescent reaction with coelentarazine as substrate. In the presence of neutralizing antibodies, however, PsV infection is blocked and transduction of the reporter gene is reduced or absent. Sera were analysed in serial dilutions ranging from 1:40 to 1:180,000 and serum concentrations inhibiting 50% of the PsV infection (EC50 values) were calculated from median of triplicates. EC50 values >40 (corresponding to 1.3 and 1.1 IU/ml for HPV16 and HPV18, respectively) were defined as neutralization seropositive and EC50 values >180,000 (corresponding to >5,818 and >5,019 IU/ml for HPV16 and HPV18, respectively) could not be differentiated further.
Statistical analysis
HPV type-specific seroprevalence (percentage of neutralization-positive among all sera), geometric mean titers (GMT) (neutralization-positive sera only) with 95% confidence intervals and the two-tailed Spearman nonparametric correlation coefficients (rs) (all sera) were calculated using GraphPad Prism.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The authors confirm that the raw data of this HT-PBNA study were generated at the EMBL-DKFZ Chemical Biology Core Facility. Ranked correlation of the neutralizing antibody levels are available in the Supplementary Table. Additionally derived data supporting the findings of this study are available upon reasonable request to FCM (f.mariz@dkfz.de) and Noemi Bender (n.bender@dkfz.de) according to DKFZ data safety protection regulations.
References
Lehtinen, M. & Dillner, J. Clinical trials of human papillomavirus vaccines and beyond. Nat. Rev. Clin. Oncol. 10, 400–410 (2013).
Lehtinen, M. et al. Overall efficacy of HPV-16/18 AS04-adjuvanted vaccine against grade 3 or greater cervical intraepithelial neoplasia: 4-year end-of-study analysis of the randomised, double-blind PATRICIA trial. Lancet Oncol. 13, 89–99 (2012).
The FUTURE II Study Group. Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N. Engl. J. Med. 356, 1915–1927 (2007).
Arbyn, M. & Xu, L. Efficacy and safety of prophylactic HPV vaccines. A Cochrane review of randomized trials. Expert Rev. Vaccines 17, 1085–1091 (2018).
Schiller, J. T. & Lowy, D. R. Immunogenicity testing in human papillomavirus virus-like-particle vaccine trials. J. Infect. Dis. 200, 166–171 (2009).
Draper, E. et al. A randomized, observer-blinded immunogenicity trial of Cervarix((R)) and Gardasil((R)) Human Papillomavirus vaccines in 12-15 year old girls. PloS ONE 8, e61825 (2013).
Harper, D. M. & DeMars, L. R. HPV vaccines - A review of the first decade. Gynecologic Oncol. 146, 196–204 (2017).
Faust, H. et al. Human Papillomavirus neutralizing and cross-reactive antibodies induced in HIV-positive subjects after vaccination with quadrivalent and bivalent HPV vaccines. Vaccine 34, 1559–1565 (2016).
Artemchuk, H. et al. Long-term antibody response to human papillomavirus vaccines: up to 12 years of follow-up in the finnish maternity cohort. J. Infect. Dis. 219, 582–589 (2019).
Lehtinen, M. & Paavonen, J. Sound efficacy of prophylactic HPV vaccination: basics and implications. Oncoimmunology 1, 995–996 (2012).
Pinto, L. A., Dillner, J., Beddows, S. & Unger, E. R. Immunogenicity of HPV prophylactic vaccines: serology assays and their use in HPV vaccine evaluation and development. Vaccine 36, 4792–4799 (2018).
Huh, W. K. et al. Final efficacy, immunogenicity, and safety analyses of a nine-valent human papillomavirus vaccine in women aged 16-26 years: a randomised, double-blind trial. Lancet 390, 2143–2159 (2017).
Setiawan, D., Luttjeboer, J., Pouwels, K. B., Wilschut, J. C. & Postma, M. J. Immunogenicity and safety of human papillomavirus (HPV) vaccination in Asian populations from six countries: a meta-analysis. Jpn. J. Clin. Oncol. 47, 265–276 (2017).
Apter, D. et al. Efficacy of human papillomavirus 16 and 18 (HPV-16/18) AS04-adjuvanted vaccine against cervical infection and precancer in young women: final event-driven analysis of the randomized, double-blind PATRICIA trial. Clin. Vaccin. Immunol. 22, 361–373 (2015).
Villa, L. L. et al. Immunologic responses following administration of a vaccine targeting human papillomavirus Types 6, 11, 16, and 18. Vaccine 24, 5571–5583 (2006).
Nygard, M. et al. Evaluation of the long-term anti-human papillomavirus 6 (HPV6), 11, 16, and 18 immune responses generated by the quadrivalent HPV vaccine. Clin. Vaccin. Immunol. : CVI 22, 943–948 (2015).
Donken, R. et al. High effectiveness of the bivalent human papillomavirus (HPV) vaccine against incident and persistent HPV infections up to 6 years after vaccination in young Dutch women. J. Infect. Dis. 217, 1579–1589 (2018).
Kavanagh, K. et al. Changes in the prevalence of human papillomavirus following a national bivalent human papillomavirus vaccination programme in Scotland: a 7-year cross-sectional study. Lancet Infect. Dis. 17, 1293–1302 (2017).
Kudo, R. et al. Bivalent human papillomavirus vaccine effectiveness in a Japanese population: high vaccine-type-specific effectiveness and evidence of cross-protection. J. Infect. Dis. 219, 382–390 (2019).
Bogaards, J. A., van der Weele, P., Woestenberg, P. J., van Benthem, B. H. B. & King, A. J. Bivalent HPV vaccine effectiveness correlates with phylogenetic distance from Hpv vaccine types 16 and 18. J. Infec. Dis. 220, 1141–1146 (2019).
Namujju, P. B. et al. Impact of smoking on the quantity and quality of antibodies induced by human papillomavirus type 16 and 18 AS04-adjuvanted virus-like-particle vaccine - a pilot study. BMC Res. Notes 7, 445 (2014).
Sankaranarayanan, R. et al. Immunogenicity and HPV infection after one, two, and three doses of quadrivalent HPV vaccine in girls in India: a multicentre prospective cohort study. Lancet Oncol. 17, 67–77 (2016).
Sehr, P. et al. High-throughput pseudovirion-based neutralization assay for analysis of natural and vaccine-induced antibodies against human papillomaviruses. PloS ONE 8, e75677 (2013).
Acknowledgements
The authors thank Kerstin Putzker (EMBL) for valuable technical support and Tapio Luostarinen (Finnish Cancer Registry) for discussion of the correlation analysis. This work was funded by the Finnish Cancer Organization, Academy of Finland and The Bill & Melinda Gates Foundation. The funders had no role in the data collection and analysis, or preparation of the manuscript. This manuscript contains information presented during the 1st German Cancer Research Conference (DKFK) held in Heidelberg, February 2019 (abstract ID-203).
Author information
Authors and Affiliations
Contributions
F.C.M., M.P. and M.L. were the principal contributors to the manuscript preparation. F.C.M. was also responsible for the production and characterization of PsV preparations and contributed to the statistical analysis and data interpretation. M.P. was also involved in the conception and design of the study, in the supervision of laboratory analysis of sera, data analysis, and interpretation. M.L. was responsible for the conception, design, monitoring, and supervision of this study, data analysis and interpretation. N. Bender and K. P. were responsible for the study planning. D.A., P.B., N. Bhatla, M.R.P. and P.R.P. were involved in conduct, monitoring, supervision, and data reporting of the multicentric trial with the quadrivalent vaccine in India. R.S. was responsible for the conception, design, and supervision of the multicentric trial with the quadrivalent vaccine in India. T.E. was involved with laboratory analysis. P.S. was responsible for the laboratory analysis of sera for this study in Heidelberg, data acquisition and analysis. T.W. was involved in the supervision of laboratory analysis on sera, study design, and manuscript review. M.M. was responsible for the supervision of laboratory analysis on PsV production and characterization, data analysis, and interpretation. All authors reviewed and commented on a draft of the manuscript and gave final approval for submission.
Corresponding author
Ethics declarations
Competing interests
M.L. has received grants for HPV vaccination studies from Merck Sharp & Dohme and GlaxoSmithKline Biologicals through University of Tampere. M.M., M.P. and P.S. are inventors on a patent application by DKFZ on the high-throughput-pseudovirion-based neutralization assay (HT-PBNA). M.M. is an inventor of patents related to virus-like particle vaccines owned by Loyola University Chicago (Chicago, USA) and licensed to GlaxoSmithKline Biologicals. M.M. is an inventor of Trx-L2 vaccines and received personal fees from Merck Sharp & Dohme for analyses of clinical trial sera. N. Bhatla has received research funding from GlaxoSmithKline and Merck Sharp & Dohme through All India Institute of Medical Sciences. T.W. serves on advisory boards for MSD Sharpe & Dohme. The other authors declare no competing interests. P.B. and R.S. are staff members of the International Agency for Research on Cancer/World Health Organization. The authors alone are responsible for the views expressed in this article and they do not necessarily represent the decisions, policy or views of the International Agency for Research on Cancer/World Health Organization.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mariz, F.C., Bender, N., Anantharaman, D. et al. Peak neutralizing and cross-neutralizing antibody levels to human papillomavirus types 6/16/18/31/33/45/52/58 induced by bivalent and quadrivalent HPV vaccines. npj Vaccines 5, 14 (2020). https://doi.org/10.1038/s41541-020-0165-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41541-020-0165-x
This article is cited by
-
Lack of detectable HPV18 antibodies in 14% of quadrivalent vaccinees in a longitudinal cohort study
npj Vaccines (2024)
-
Can prophylactic HPV vaccination reduce the recurrence of cervical lesions after surgery? Review and prospect
Infectious Agents and Cancer (2023)