Abstract
Machine reading (MR) is essential for unlocking valuable knowledge contained in millions of existing biomedical documents. Over the last two decades1,2, the most dramatic advances in MR have followed in the wake of critical corpus development3. Large, well-annotated corpora have been associated with punctuated advances in MR methodology and automated knowledge extraction systems in the same way that ImageNet4 was fundamental for developing machine vision techniques. This study contributes six components to an advanced, named entity analysis tool for biomedicine: (a) a new, Named Entity Recognition Ontology (NERO) developed specifically for describing textual entities in biomedical texts, which accounts for diverse levels of ambiguity, bridging the scientific sublanguages of molecular biology, genetics, biochemistry, and medicine; (b) detailed guidelines for human experts annotating hundreds of named entity classes; (c) pictographs for all named entities, to simplify the burden of annotation for curators; (d) an original, annotated corpus comprising 35,865 sentences, which encapsulate 190,679 named entities and 43,438 events connecting two or more entities; (e) validated, off-the-shelf, named entity recognition (NER) automated extraction, and; (f) embedding models that demonstrate the promise of biomedical associations embedded within this corpus.
Similar content being viewed by others
Introduction
Even the relatively specialized subfields of present-day biology and medicine are facing a deluge of accumulating research articles, patents, and white papers. It is increasingly difficult to stay up-to-date with contemporary biomedicine without the use of sophisticated machine reading (MR) tools. MR tool development, in turn, has been limited by the availability of biomedical corpora carefully annotated by experts. This is especially true with respect to information extraction, such as named entity recognition (NER) and relation or event extraction. Although several corpora have been developed for specialized biomedical subdomains, the need for a corpus that can bridge biological, general scientific, environmental, and clinical scientific sublanguages is greater than ever before.
Unfortunately, the annotation of natural science texts is more challenging than in other domains. Biomedical language is replete with ambiguity distinct from that observed in news articles or informal text online. When a word or phrase’s semantic meaning is clearly separated (the east bank of the Danube versus Deutsche Bank), we can implement automated sense disambiguation using machine learning tools. In biomedical texts, however, alternative meanings are not always clearly separated. The problem is not that a phrase can refer to several distinct, real-world entities in different contexts, but that scientists writing articles typically do not separate competing, close meanings. For example, in some biomedical contexts, the words for a named entity may refer to a gene or a protein with nearly equal probability; for example, “a mutant hemoglobin α2” can refer to either a gene or a protein. If the author meant gene-or-protein A, and we force an annotator to choose either interpretation gene A or protein A, the resulting annotation is of limited utility because the choice between gene and protein is random if the meanings are equally likely based on context. Ideally, a specialized ontology of text entities would allow an annotator to choose the proper level of annotation granularity (the words represent a gene-or-protein, in this example), minimizing the need for forced, random decisions. To the best of our knowledge, there is no biomedical ontology that meets the requirements for capturing semantic ambiguity. We aimed to fill this gap by developing a specialized, variable-level meaning resolution ontology, a carefully curated corpus, along with corpus annotation tools, and a collection of text embedding analyses to evaluate our annotated corpus.
Results
Our new ontology, called NERO, short for Named Entity Recognition Ontology, attempts to minimize unwarranted, arbitrary annotative semantic label assignments for textual entities, see Fig. 1. NERO represents textual entities, and more specifically named entities recognized by text mining tools. NERO is designed as an extension of the IAO (Information Artifact Ontology: http://www.obofoundry.org/ontology/iao.html), one of the widely used OBO ontologies for representing information. The IAO class InformationContentEntity is employed as the top-level class in NERO. It has two subclasses: TextualEntity defined by IAO and the class AnnotationInText to capture information about annotations. The branch TextualEntity in IAO includes various parts of text (e.g., Document title, Abstract, and Table), but it does not include entities that are of concern to text mining processes, e.g. NamedEntity. NERO enables representations required by text mining and formally defines such classes as NamedEntity, NamedEntityGroup, Relationship, and Pronoun as subclasses of the IAO class TextualEntity.
The Ontology is shown here as a multifurcating tree, with taxonomy nodes corresponding to ontology classes. Class name and class mentions count in the corpus are shown in parentheses next to each named entity class. Each taxonomy class is provided with a unique pictogram (black and red shapes on yellow background) intended to simplify expert manual annotation of the corpora. In total, we annotated 35,865 sentences. These sentences encapsulated 190,679 named entities and 43,438 events connecting two or more entities. In addition to the almost two dozen, more sparsely-used branches (such as ExperimentalFactor and GeographicalLocation) under the NamedEntity cluster, there are three heavily-represented branches in our corpus: AnatomicalPart, Chemical, and Process. Slightly more than half (51.6%) of all entities are from these three classes, with 26.6% of all entities originating from Process alone. We designed our ontology and its annotations to capture the named entities associated with research activities and facilities; these types of entities can be important for encoding methods used in scientific experiments or patient treatment. The semantic classes ResearchActivity and MedicalProcedures turn out to be the ninth and the tenth most frequent, respectively. Other top concepts related to the research include Measurement, IntellectualProducts, PublishedSourceofInformation, Facility, and MentalProcess.
NERO defines ambiguous concepts, such as GeneOrProtein, which subsumes both Gene and Protein using the following axiom: EquivalentTo: “Gene”or “Protein.” There are no biological entities that are either a gene or a protein, but there are lexical entities that can correspond to either or both of these entities. NERO uses this pattern to express appropriate ambiguity regarding textual entities, preserving uncertainty from the text. In this way, NERO classes represent textual instances and not the actual biological entities to which these instances refer. Often, it is possible to link recognized in the text lexical entities to corresponding biological entities, e.g., through a relationship “is about”.
For example, in some cases the considered previously textual entity “a mutant hemoglobin α2” can be identified as a protein and linked to the corresponding biological entity defined, e.g., by UniProtKB with URI: U6A3P2. However, as previously discussed sometimes that is not possible. The role of NERO is to provide a logically sound and computationally processable representation of such ambiguous cases.
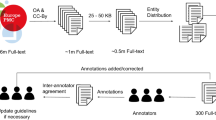
Striving to make the ontology practically useful, we designed guidelines for annotators making decisions in annotating text entities, available in the Supplementary Data. Furthermore, by recruiting a team of postdoctoral-level and industry experts, we annotated a large biomedical corpus to enable a broad range of natural language processing and biomedical machine learning tasks. Our annotations span 35,865 unique sentences, 8650 of which were annotated by multiple annotators with a remarkably high inter-annotator agreement (see Supplementary Table 1). In our annotated corpus, we aimed to encompass all entity types that might occur in biomedical literature. In addition to named entities, our ontology captures Events that represent relationships between biomedical concepts. The frequencies of all diverse entity types in our corpus are shown in Fig. 2A, B shows the frequencies of relations represented in the taxonomy. The most frequent text entity type is GeneOrProtein, which accounts for 14.7% of all named entities in the corpus (see Fig. 2A). The second most populous textual entity category is Process, with nine percent tagged. Process has six sub-concepts and almost half of Process instances (49.7%) are annotated as more specific sub-concepts; the BiologicalProcess and the MolecularProcess are the fifth and seventh most frequent text entity types (see Fig. 2). Entity type frequencies follow a heavy-tail distribution, with the least frequent types being Journal, Unit, and Citation (see Fig. 2). In addition to 190,679 named entities, we annotated 43,438 action terms, events connecting two or more entities. The most annotated action term is bind, accounting for 28.4% of all actions, see Supplementary Fig. 1. When we normalize the action terms and combine actions such as bind, binds, and binding, the normalized action bind accounts for 31.8% of all actions, as shown in Supplementary Fig. 1. We deployed a package called NERO-nlp for researchers interested in diving deeper into our annotated corpus; the installation guides and scripts are available online at https://pypi.org/project/NERO-nlp and https://github.com/Bohdan-Khomtchouk/NERO-nlp respectively.
As is typically the case with human languages, semantic classes are represented unevenly in free texts, following a heavy-tail (Zipf’s) distribution. a In biomedical corpora, unsurprisingly, named entities associated with genes and proteins are the most prevalent (15%), followed by processes (9%), medical findings (8.8%), and chemicals (6.7%). At the low-frequency end of the named entity spectrum, we find journal names, units, citations, and languages. b Events connecting two or more entities are also approximately Zipf-law distributed. Event frequencies are closely tracking corresponding named entity classes. For example, the most frequent event, bind, is associated with the most frequently named entity, GeneOrProtein. We tried fitting the rank-ordered frequency distribution of annotated named entities with a Discrete Generalized Beta Distribution (DGBD). The result showed a significant deviation from Zipf’s law33: The observed distribution’s tail was not heavy enough to match Zipf’s distribution, most likely due to the relatively small number of classes in our ontology34. In other words, we expect that frequencies of semantic classes in a very large corpus, annotated with classes from a hypothetical perfect named entity ontology, would follow a Zipfian (discrete Pareto) distribution of named entity classes. Our action annotations have moved beyond interactions between proteins and genes (e.g., bind, inhibit, phosphorylate, encode), into interactions involving genetic variants and environmental factors (e.g., associated with, occur in presence of, trigger, lack). Ambiguity levels varied broadly across the named entities captured in our corpus. For example, in the class AnatomicalPart, almost all (99.3%) are annotated at the most specific levels, with the majority of entities belonging to BodyPart, CellularComponent, and Cell. In contrast, the general (most vague) concept, Chemical, turns out to be the most annotated within its cluster, although more specific subclasses, such as Protein, NucleicAcid, and Drug are also well represented in the corpus. In the Process concept cluster, about a third of all concept instances are annotated at a more general Process level, and the rest of them are specific concepts, such as MedicalProcedure, MolecularProcess, ResearchActivity, and BiologicalProcess. In addition to these major clusters of concepts, several individual concepts are well represented in the corpus. For example, MedicalFinding represents 7.3% of all entities. Other well-represented concepts include Duration, IntellectualProduct, Measurement, Organism, PersonGroup, PublishedSourceOfInformation, and Quantity. In total, about 70.4% of all entities are annotated at the most specific ontology level. There are five concepts in the NERO ontology that allow the semantic flexibility needed to avoid arbitrary concept assignment. Entities annotated as AminaoAcidOrPeptide, QuantityOrMeasurement, PublicationOrCitation, MedicalProcedureOrDevice, and GeneOrProtein account for 17.8% of all entities, while less than a quarter (23%) of entities representing either genes or proteins are cleanly annotated with class Gene or class Protein. The remainder are annotated with class GeneOrProtein. In addition to the action bind, actions indicating entities’ attributes are the next most frequent. Other biological relationships are also well-represented in this annotation, such as inhibit, activate, mediate, interact, contain, and regulate. The top 30 action categories account for 64.4% of all actions annotated with the top ten action categories accounting for 52.2%. Interestingly, negations of actions were also quite abundant in our annotated corpus. For example, do not bind was the sixth most frequent normalized action. Other well-represented negations of actions include do not affect and do not inhibit (see Supplementary Figs. 1–3).
Below, we present two practical applications of our ontology and text annotations: (1) Machine learning experiments, which automatically identify named entities, and; (2) Word embedding experiments, which leverage the automated discovery of semantic relationships among real-world concepts referenced by a text’s named entities.
Machine learning experiments
Using NERsuite5, we conducted a tenfold cross-validation, dividing the corpus into training and test subsets. The classification results are presented in Supplemental Tables 1 and 2. The overall automated NER performance is moderate, with 54.9% precision, 37.3% recall, and a 43.4% F1 score. The best performance class, GeneOrProtein, had baseline results of 67.0% precision, 65.3% recall, and a 66.2% F1 score. In addition to the default baseline implementation of NERsuite, we added additional features in the training process to improve its performance6. These are dictionary features derived from lookups in technical term dictionaries. The classifier with dictionary features manifests 54.7% precision, 37.9% recall, and a 43.8% F1 score. We observed a scant 0.35% increase in F1 score from adding dictionary features. We then implemented an ensemble method called stacking, where we trained a higher-level model to learn how to best combine contributions from each base model. The base model, in this case, is the baseline model from NERsuite. Stacking yielded a 0.27% increase in F1 score compared to baseline results. While ensemble methods are commonly used to boost model accuracy by combining the predictions of multiple machine learning models, choices of second-level and base models can influence the amount of improvement in model accuracy. The overall performance statistics are shown in Supplementary Table 2. As our corpus is made public with this study’s publication, we hope that other researchers will use this training data to achieve core MR task performance that surpasses our initial experiments.
To examine how NERsuite performs in comparison to other popular open-source NER tools, we trained a custom NER model on our annotated corpus using spaCy7. We evaluated the trained model on the test subset, which consists of a random 10% sample from the corpus. Overall automated NER performance is low, with 30.9% precision, 8.6% recall, and a 13.4% F1 score. The best performance class, GeneOrProtein, had results of 45.1% precision, 36.4% recall, and a 40.3% F1 score. These statistics indicate a much poorer performance of spaCy compared to that of NERsuite.
To help explain the huge difference in performance between NERsuite and spaCy, we considered the set of input features used by each tool for insight. NERsuite’s baseline implementation uses an extra set of input features including the lemma, POS-feature, and chunk-feature, whereas our custom spaCy NER model only relies on character offsets and entity labels. There is potential for further customizing spaCy’s processing pipelines by adding more components such as tagger and parser7, but no established approaches in this regard have been made available partly because spaCy’s model architecture is different from those of other popular NER tools. We also observed that some entity classes, such as Gene and Protein, have zero values for precisions, recalls, and F1 scores, which likely translate to no correct classifications made for those entities. The zero values occur partly due to the relatively smaller number of tokens for those entity classes in the training set, and as a result, the trained NER model generalized poorly on the minority class entities in the test subset.
Due to spaCy’s computational demands, we did not conduct tenfold cross-validation. NERsuite provides a well-integrated pipelined system where training a new model consists of a few lines of code. In addition, NERsuite has a demonstrated record5 on two biomedical tasks, the BioCreative2 gene mention recognition task and the NLPBA 2004 NER task. Therefore, one could argue that it offers an advantage over spaCy for NLP tasks in specialized domains such as biomedicine.
We further applied another package, called scispaCy8, that contains spaCy models for processing biomedical, scientific, or clinical text. SciSpaCy acts as an extension to spaCy and provides a set of practical tools for text processing in the biomedical domain8. In particular, scispaCy includes a set of spaCy NER models trained on popular biomedical corpora, which covers entity types such as chemicals, diseases, cell types, proteins, and genes. As an extension to spaCy, it also has the flexibility for users to train a custom NER model from scratch or update the existing NER models with users’ own training data. Since our NER ontology adopts a more diverse and detailed annotation methodology for named entity types, it will be challenging to update scispaCy’s pretrained named entity recognizer with our annotated corpora.
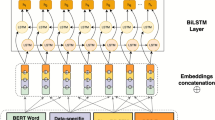
Note that NERsuite was implemented in C++ and consists of three modularized programs: a tokenizer, a tagger, and a named entity recognizer9,10. The algorithm used behind the named entity recognizer is conditional random fields (CRF), which is often applied in tasks such as NER, part-of-speech tagging, and gene prediction. The NER model in spaCy features a sophisticated word embedding strategy using subword features and “Bloom” embeddings, a deep convolutional neural network with residual connections, and a novel transition-based approach to named entity parsing11,12,13. The Spark NLP library is inspired by a former state-of-the-art model for NER, which adopts a novel neural network architecture that automatically detects word- and character-level features using a hybrid bidirectional LSTM and CNN architecture, eliminating the need for most feature engineering14.
Next, we built a NER model with BERT in the Spark NLP library, which is inspired by a former state-of-the-art model for NER: Chiu & Nicols, NER with Bidirectional LSTM-CNN. The paper presents a novel neural network architecture that automatically detects word- and character-level features using a hybrid bidirectional LSTM and CNN architecture, eliminating the need for most feature engineering. The overall automated NER performance is low, with 28.2% precision, 8.4% recall, and a 12.9% F1 score. The best performance class, GeneOrProtein, had results of 32.3% precision, 27.2% recall, and a 29.5% F1 score. We observed 8 out of 13 entity classes have zero values for precision, recalls, and F1 scores. The zero values occur partly due to the relatively smaller number of tokens for those entity classes in the training set, and as a result, the trained NER model generalized poorly on the minority class entities in the test subset. Another reason for the large proportion of zeros in our results might be that Spark NLP requires a stricter input data format, that is, conll 2003. Therefore, further data normalization and cleaning can potentially improve upon our current results.
Finally, we compared our work against a nested NER neural learning model as proposed by Ju et al. (2018)15. Similar to our previous experiments, we divide the corpus into training and test data, where a full description of the results can be found in Supplementary Tables 1–3. We chose to use the nested NER for comparison, because of its ability to capture fine-grained semantic information in text by stacking NER layers. This enables the learning model to extract entities in an inside-out way using all the encoded information available15. Overall, the learning model achieves a 53.527% precision, 54.29% recall, and 53.906% F1 score, improving upon previous results using the NERsuite by 10%. The best performing class is Gene or Protein achieving 65.64% precision, 75.992% recall, and 70.439% F1 score.
Word embedding experiments
Semantic associations, automatically extracted from text using neural network embedding operations, can function as a kind of “digital double” of real-world phenomena embedded in the text, facilitating inferences that were previously imagined only possible from the original experimental data. For example, word embeddings built from chemical and materials science texts predict much of the subsequent decades’ material discoveries8, just as the corpus of molecules can recover the periodic table9, and texts are able to recover the subtle, psychological, and sociological biases of cultures that produced them16,17. We used word embedding models to evaluate the biomedical veracity of NERO and its text annotation. Embedding models like Google’s word2vec18,19 initially received substantial attention based on their capacity to solve analogy problems and automatically capture deep semantic relationships among concepts. Building on these capacities16,20,21, we proposed a general method for constructing meaningful dimensions by taking the arithmetic mean of word vectors representing antonyms along a dimension and using them to diagnose their meanings. This approach has been widely validated11,21,22,23,24, and we employed it here to construct and compare the meanings embedded in NERO and our annotated corpus with ground truth data about drugs and diseases. In order to evaluate word embeddings based on NERO, we identified two disease properties —(1) severity and (2) gender specificity—and likewise two therapeutic drug properties —(1) toxicity and (2) expense—not directly present in the text, but highly relevant to diagnosis and treatment, and on which text-independent ground truth data exists.
We embedded named entities associated with diseases and drugs into a high-dimensional space in which every NERO term was assigned a 300-dimensional vector, (see Fig. 3 for a three-dimensional projection of this embedding), along with a selection of diseases and medications used to treat them. For all embedding experiments, we used a word2vec18,19 implementation provided by the package gensim25. The corpus that we used included (1) English Wikipedia download; (2) A collection of articles from 15 Elsevier journals licensed to the University of Chicago; and (3) A collection of Reuters newswire articles purchased from the provider. The corpus was represented as a shuffled set of sentences stored in an Oracle Berkeley DB, allowing dynamic reordering of sentences, followed by feeding one sentence at a time into a word2vec bag of words model. For incorporating disease-specific vocabulary we used the dis2vec biomedical wrapper for gensim12. The parameters used for analysis included an embedding dimension (300, but we also tried 100 and 500 with results essentially unchanged), a sentence window size of ten words, and training run off 20 epochs.
Properties of diseases and drugs are visible in the first three principal components of our multi-dimensional text embedding. The figure shows a projection of text embedding into three-dimensional space, with named entities corresponding to diseases and drugs shown with prisms and spheres, respectively. The figure represents several projections of the same embedding, preserving spatial layout and projection, with distinct elements of the embedding indicated by shape color. The central image shows all disease systems and their corresponding medications together. More specifically, the additional projections show: a Zollinger–Ellison syndrome and associated medications; b cancers and associated therapies; c central nervous system diseases and corresponding medications; d, e Viral and bacterial infectious diseases, respectively, together with corresponding antiviral and antibiotic agents, and f 3-dimensional projection of embedding drug- and disease-related named entities corresponding to CNS/Psychiatric- (red), digestive- (yellow), infectious/immune- (green), neoplastic- (cyan), and other diseases (grey). Another view of the same dataset is presented in Fig. 4.
We then compared drug and disease projections into the embedding dimensions for severity, gender, toxicity, and expense with ground truth about these qualities. We constructed the severe-mild axis with the following contrasting term pairs: (harmful, beneficial), (serious, benign), (life-altering, common), (disruptive, undisruptive), (dying, recovering), (dangerous, safe), (threatening, low-priority), (high mortality, harmless), (costly, cheap), (hospitalized, self-administered), (hospital, work), (debt, savings), (low quality of life, undisruptive), and (hazard, routine). Then we compared disease projection in this dimension with World Health Organization data on the burden of living with each of those diseases (DALYs13) and found a correlation of 0.329 (p = 0.0614, n = 33). We then constructed a gender dimension with similarly contrasting pairs: (male, female), (prostate, ovary), (penile, uterine), (penis, uterus), (man, woman), (men, women), (masculine, feminine), (he, she), (him, her), (his, hers), (boy, girl), and (boys, girls). We compared the disease projection in this gender dimension with the prevalence of those diseases for men and women from a substantial sample of doctor–patient insurance records capturing ~47% of all of US doctor–patient visits between 2003 and 2011 and found a correlation of 0.436 (p = 1.46 × 10−13, n = 261).
We tested the robustness of our 300-dimensional embedding by comparing it with 100-dimensional and 500-dimensional embeddings obtained using the same corpus and comparing distances between the same pair of named entities (disease or drug) in embeddings of different dimensionality. The results appear to be very stable with respect to dimensionality of the embedding: distances between named entities were highly correlated at ρ = 0.89 for 100- and 300-dimensional embeddings comparison, and at ρ = 0.95 for 500- to 300-dimensional embedding comparison (see Fig. 4 in the Supplementary Data).
Left We projected diseases into two dimensions: female-male (X-axis) and severe-mild (Y-axis). We defined the “male–female” axis using the following pairs of terms: (“male,” “female”), (“prostate,” “ovary”), (“penile,” “uterine”), (“penis,’’ “uterus”), (“man,” “woman”), (“men,” “women”), (“masculine,” “feminine”), (“he,“ “she”), (“him,” “her”), (“his,” “hers”), (“boy,” “girl”), and (“boys,” “girls”). We defined the severe-mild axis with the following term pairs: (“harmful,” “beneficial”), (“serious,” “benign”), (“life-altering,” “common“), (“disruptive,” “undisruptive”), (“dying,’’ “recovering”), (“dangerous,” “safe”), (“threatening,” “low-priority”), (“high mortality,” “harmless”), (“costly,” “cheap”), (“hospitalized,” “self-administered”), (“hospital,” “work”), (“debt,” “savings”), (“low quality of life,” “undisruptive”), and (“hazard,” “routine'). Right We projected medications into “benign-toxic” (X-axis) and “cheap-costly” (Y-axis). For the “benign-toxic” axis, we used the following pairs of antonym words: (“harmful,” “beneficial”), (“toxic,” “nontoxic”), and (“noxious,” “benign”). We defined the “expensive–inexpensive” dimension using the following pairs of terms: (“expensive,” “inexpensive“), (“costly,” “cheap”), (“brand,” “generic”), and (“patented,” “off-patent”).
Next, we projected medication-related NERs onto a toxicity axis composed from the following antonym pairs: (harmful, beneficial), (toxic, nontoxic), and (noxious, benign) and an expense dimension anchored by: (expensive, inexpensive), (costly, cheap), (brand, generic), and (patented, off-patent). The embedding-derived drug projections onto the toxic-harmless dimension correlates at 0.32 (p = 1.1 × 10−4) with the corresponding drug-specific median lethal dose (dose required to kill 50% of model animals as documented in the LD50 database26). Finally, the correlation of drug projections into an expense dimension and the actual price of each drug as listed in the IBM MarketScan database27 was 0.42 (p = 1.5 × 10−15) (see Fig. 4). When a disease projects low in the male–female dimension, it is much more likely to afflict women than men, such as ornithosis and related infectious diseases. When a disease projects high in the serious–benign dimension like leprosy, it is likely to incur substantial suffering. Medications projecting with negative (toxic) values in the toxic–nontoxic dimension, as the name suggests, tend to be associated with more severe side effects. For example, the drug Riluzole, a treatment for amyotrophic lateral sclerosis, has side effects ranging from unusual bleeding to nausea and vomiting. Drug projections high in the expensive–inexpensive dimension forecast a stiff medical bill, as in the case of Simvastatin, which is used to reduce the risk of heart attack and stroke, and which, before it went off-patent, used to cost hundreds of dollars per bottle. The robustness of these results suggests that scientific corpora can be used for the automated generation of robust hypotheses meriting follow-up empirical study.
Discussion
This study’s main limitation is that, even though our NERO ontology aimed to cover all entities contained in the biomedical research literature, we did not cover all levels of granularity in classifying entities. Moreover, while the major concepts are well-annotated, several concept types were not well-represented because of the heavy-tail distribution of ontological class frequencies. In addition, we note that satisfactory results of NER rely heavily on a large quantity of hand-annotated data, which is often costly in terms of time and resources spent. Therefore, the adoption of semi-supervised learning methods, which incorporates unlabeled data to improve learning accuracy, could reduce the need for manual annotation28.
While there is a popular belief that pretraining on general-domain text can be helpful for developing domain-specific language models, a recent study has shown that for specialized domains, such as biomedicine, pretraining on in-domain text from scratch offers noticeable improvements in model accuracy compared to continual pretraining of general-domain language models29. Therefore, we trained our annotated corpus from scratch in our machine learning experiments30.
The resources offered in our study can be applied to a wide range of scientific problems. First, the proposed NERO ontology can facilitate more robust and accurate large-scale text mining of biomedical literature. As discussed above, NERO is the first knowledge graph in this field, accounting for context-relevant levels of ambiguity. Graph neural networks31 can leverage such prior knowledge from human experts for learning embeddings of biomedical entities, which is likely to preserve both semantic meaning in the original literature and domain knowledge from human experts. Second, researchers can combine the curated corpus from this study with self-supervised learning32. Such a learning scenario can utilize the unlabeled data in a supervised way by predicting part of the sentence using the rest of the sentence. The annotated corpus from this study can be used to fine-tune language models, orienting them for critical biomedical tasks.
Methods
Snippets of methods are integrated with results.
Data availability
The datasets generated during and/or analyzed during the current study are available in the Github repository at https://github.com/arzhetsky/Chicago_corpus. NERO in OWL format is available at: https://bioportal.bioontology.org/ontologies/NERO.
Code availability
We also deployed a package called NERO-nlp for researchers interested in diving deeper into our annotated corpus; the installation guides and scripts are available online at https://pypi.org/project/NERO-nlp and https://github.com/Bohdan-Khomtchouk/NERO-nlp, respectively.
References
Banko, M. & Brill, E. Scaling to very very large corpora for natural language disambiguation. In Proc. 39th Annual Meeting on Association for Computational Linguistics 26–33 (Association for Computational Linguistics, 2001).
Halevy, A., Norvig, P. & Pereira, F. The unreasonable effectiveness of data. IEEE Intell. Syst. 24, 8–12 (2009).
Dogan, R. I., Leaman, R. & Lu, Z. NCBI disease corpus: a resource for disease name recognition and concept normalization. J. Biomed. Inf. 47, 1–10 (2014).
Deng, J. et al. ImageNet: a large-scale hierarchical image database. 2009 IEEE Conference on Computer Vision and Pattern Recognition 248–255 (IEEE, 2009).
Wijffels, J. & Okazaki, N. crfsuite: Conditional Random Fields for Labelling Sequential Data in Natural Language Processing based on CRFsuite: a fast implementation of Conditional Random Fields (CRFs) https://github.com/bnosac/crfsuite (2007–2018).
Friedrich, C., Revillion, T., Hofmann-Apitius, M. & Fluck, J. Biomedical and chemical named entity recognition with conditional random fields: the advantage of dictionary features. SMBM 85–89 (2006).
Honnibal, M. & Montani, I. spaCy 2: Natural language understanding with Bloom embeddings, convolutional neural networks and incremental parsing https://spacy.io (2017).
Neumann, M., King, D., Beltagy, I. & Ammar, W. ScispaCy: fast and robust models for biomedical natural language processing. In Proc. 18th BioNLP Workshop and Shared Task (Association for Computational Linguistics, 2019).
Tsujii, J. NERsuite: a named entity recognition toolkit https://nersuite.nlplab.org/index.html (2020).
Ananiadou, S. NERsuite package: a named entity recognition toolkit http://nersuite.nlplab.org (2014).
Kang, D. & Evans, J. Against method: exploding the boundary between qualitative and quantitative studies of science. Quant. Sci. Studies 1, 930–944 (2020).
Ghosh, S., Chakraborty, P., Cohn, E., Brownstein, J. S. & Ramakrishnan, N. Characterizing diseases from unstructured text: a vocabulary driven word2vec approach. In Proc. 25th ACM International on Conference on Information and Knowledge Management, CIKM ’16, 1129–1138. (Association for Computing Machinery, New York, NY, United States, 2016).
Mathers, C. D. History of global burden of disease assessment at the World Health Organization. Arch. Public Health 78, 77 (2020).
Chiu, J. P. C. & Nichols, E. Named entity recognition with bidirectional LSTM-CNNs. Trans. Assoc. Comput. Linguist. 4, 357–370 (2016).
Ju, M., Miwa, M. & Ananiadou, S. A neural layered model for nested named entity recognition. 2018 Conference of the North American Chapter of the Association for Computational Linguistics: Human Language Technologies. 1446–1459 (Association for Computational Linguistics, 2018).
Caliskan, A., Bryson, J. J. & Narayanan, A. Semantics derived automatically from language corpora contain human-like biases. Science 356, 183–186 (2017).
Garg, N., Schiebinger, L., Jurafsky, D. & Zou, J. Word embeddings quantify 100 years of gender and ethnic stereotypes. Proc. Natl Acad. Sci. USA 115, E3635–E3644 (2018).
Mikolov, T., Sutskever, I., Chen, K., Corrado, G. S. & Dean, J. Distributed representations of words and phrases and their compositionality. Adv. Neural Inf. Process. Syst. 3111–3119 (2013).
Mikolov, T., Chen, K., Corrado, G. & Dean, J. Efficient estimation of word representations in vector space. ICLR (2013).
Austin, C. K., Taddy, M. & Evans, J. A. The geometry of culture: analyzing the meanings of class through word embeddings. Am. Sociol. Rev. 85, 197–197 (2020).
Kozlowski, A. C., Taddy, M. & Evans, J. A. The geometry of culture: analyzing the meanings of class through word embeddings. Am. Sociol. Rev. 84, 905–949 (2019).
Kwak, H., An, J., Jing, E. & Ahn, Y.-Y. FrameAxis: characterizing framing bias and intensity with word embedding. PeerJ Comput. Sci. https://doi.org/10.7717/peerj-cs.644 (2020).
Gurevych, I. & Miyao, Y (eds) Proc. 56th Annual Meeting of the Association for Computational Linguistics (Volume 1: Long Papers) (Association for Computational Linguistics, 2018).
Bodell, M. H., Arvidsson, M. & Magnusson, M. Interpretable word embeddings via informative priors. The 2019 Conference on Empirical Methods in Natural Language Processing and the 9th International Joint Conference on Natural Language Processing 6324–6330 (Proceedings of System Demonstrations, 2019).
Rehurek, R. & Sojka, P. Gensim–python framework for vector space modelling (NLP Centre, Faculty of Informatics, Masaryk University, Brno, Czech Republic, 2011).
US National Institutes of Health. ChemIDplus https://chem.nlm.nih.gov/chemidplus/jsp/chemidheavy/help.jsp (2020).
Hansen, L. The Truven Health MarketScan Databases for Life Sciences Researchers (Truven Health Ananlytics IBM Watson Health, 2017).
Liao, W. & Veeramachaneni, S. A simple semi-supervised algorithm for named entity recognition. Proc. NAACL HLT Workshop on Semi-supervised Learning for Natural Language Processing 58–65 (Association for Computational Linguistics, 2009).
Gu, Y. et al. Domain-specific language model pretraining for biomedical natural language processing. Preprint at https://arxiv.org/abs/2007.15779 (2020).
Ju, M., Nguyen, N. T. H., Miwa, M. & Ananiadou, S. An ensemble of neural models for nested adverse drug events and medication extraction with subwords. J. Am. Med. Inform. Assn. 27, 22–30 (2020).
Wu, Z. et al. A comprehensive survey on graph neural networks. IEEE Trans. Neural Netw. Learn. Syst. 32, 4–24 (2019).
Lan, Z. et al. ALBERT: a lite BERT for self-supervised learning of language representations. ICLR (2020).
Zipf, G. K. The meaning-frequency relationship of words. J. Gen. Psychol. 33, 251–256 (1945).
Laherrere, J. & Sornette, D. Stretched exponential distributions in nature and economy: “fat tails” with characteristic scales. Eur. Phys. J. B 2, 525–539 (1998).
Acknowledgements
We are grateful to E. Gannon and M. Rzhetsky, for comments on earlier versions of this manuscript. W.P. thanks J. Li for some technical assistance and discussions. This work was funded by the DARPA Big Mechanism program under ARO contract W911NF1410333, by National Institutes of Health grants R01HL122712, 1P50MH094267, K12HL143959 (BBK), and U01HL108634-01, and by a gift from Liz and Kent Dauten. Additional support came from King Abdullah University of Science and Technology (KAUST), awards number FCS/1/4102-02-01, FCC/1/1976-26-01, REI/1/0018-01-01, and REI/1/4473-01-01.
Author information
Authors and Affiliations
Contributions
A.R., R.S., R.K., L.S., X.G., and J.A.E. designed the study; K.W. analyzed data, performed supervision of corpus annotation, and analyzed embedding data; B.C., J.A.E., H.A., T.B., Y.L., E.W., X.G., and A.R. estimated various versions of test embedding from a large corpus; S.A., M.L., F.C., J.L.A., S.G., U.H., D.M., E.S., B.B.K., J.M., W.P., A.M.S., and A.G. performed a battery of machine learning experiments for provisional NER; B.B.K. produced an interface for easy programmatic access to the ontology and corpus; B.B.K. and W.P. performed additional data analyses; K.W., A.R., R.S., B.B.K., and J.A.E. wrote the manuscript; and all authors reviewed and approved this manuscript.
Corresponding authors
Ethics declarations
Competing interests
B.B.K. is a founder of Dock Therapeutics, Inc. The remaining authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, K., Stevens, R., Alachram, H. et al. NERO: a biomedical named-entity (recognition) ontology with a large, annotated corpus reveals meaningful associations through text embedding. npj Syst Biol Appl 7, 38 (2021). https://doi.org/10.1038/s41540-021-00200-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41540-021-00200-x