Abstract
It is recognized that hazardous emissions produced from frying oils may be related to oil properties, particularly the fatty acid composition. However, investigations have been limited and partial. In this work, the emissions from deep-frying foods with three oils (palm, olive, and soybean oils) with distinct fatty acid profiles were comprehensively examined in a simulated kitchen, and the interrelationship among emitted substances, oil quality parameters, and fatty acids profiles was explored. Firstly, palm oil emitted the highest number concentration of total particle matters ((3895 ± 1796) × 103 #/cm3), mainly in the Aitken mode (20–100 nm). We observed a positive correlation between particle number concentration and levels of palmitic acid, a major saturated fatty acid (SAFA) (rs = 0.73, p < 0.05), and total polar compounds (TPC) (rs = 0.68, p < 0.05) in the fried oil, a degradation marker which was also positively correlated with that of black carbon (BC) (rs = 0.68, p < 0.05). Secondly, soybean oil emitted the highest level of gaseous aldehydes (3636 ± 607 μg/m3), including acrolein, propinoaldehyde, crotonaldehyde, hexanal, and trans-2-heptenal; the total aldehyde concentration were positively correlated with α-linolenic acid (ALA) percentage (rs = 0.78, p < 0.01), while hexanal and trans-2-heptenal were with linoleic acid (LA) (rs = 0.73 and 0.67, p < 0.05). LA and ALA were two major polyunsaturated fatty acids in non-tropical plant oils. Thirdly, palm oil emitted the most particle-bound polycyclic aromatic hydrocarbons (PAHs), and a positive association was discovered between two PAHs and SAFA percentage. Olive oil seems superior to soybean and palm oils with regards to toxic emissions during deep-frying.
Similar content being viewed by others
Introduction
Cooking with oils, especially deep-frying foods emits a significant amount of particulate and gaseous pollutants1,2,3. Cooking emissions have been found as one of the most important sources of organic particulate matter, contributing to 10–34% of total ambient primary organic aerosol4,5 and have been associated with adverse health effects, such as elevated risks of lung cancer and mutagenicity even in non-smokers6,7,8,9,10,11. A higher incidence of respiratory diseases in cooks has also been attributable to frequent exposure to the degradation products of cooking12. Various gaseous- and particle-phase toxic contaminants in the cooking emissions have been documented in the literature. Polycyclic aromatic hydrocarbons (PAHs) and aldehydes derived from pyrolysis and oxidation of organic substances are two well-recognized toxic chemical species produced during cooking13,14. The study also found that kitchens are an important source of black carbon (BC)15,16 and ultrafine particles17. Although the range hood is commonly used to discharge these pollutants in the kitchen, the performance of the range hood usually is not altogether satisfactory18,19.
Previous animal study has found that smaller particles do more damage than their larger counterparts20. Once these ultrafine particles are inhaled into the lung, they can travel to internal organs from the exchange region (alveolar cell) via the pulmonary vasculature and may directly injure distant organs21. Another potential mechanism is to spread inflammatory metabolites or mediators generated from the lung to distant organs21, leading to the progression of inflammatory diseases22.
It has been reported that deep-frying generates a higher magnitude of air pollutants than other cooking methods1,18. Controlled comparative studies demonstrated that the levels of PM2.5, PAHs, and aldehydes from cooking emissions were significantly varied by oil properties19,23. However, oil emissions from the deep-frying process have not been comprehensively investigated across oils with different fatty acid compositions. Therefore this study intended to compare multiple harmful emissions, either particulate or gaseous-phase contaminants, from deep-frying using three popular cooking oils with distinct fatty acid compositions. In addition, the relationship was also explored between emitted pollutant concentration and oil characteristics such as peroxide value (POV), acid value (AV), and total polar compounds (TPC) to shed lights on mechanisms.
Results and discussions
In this study, all experiments were carried out in a standard kitchen, and the height of the range hood was installed according to the common height of ordinary Chinese families. Based on previous COF-related research, the samplers were set 15 cm beneath the range hood and 40 cm above the electric fryer to collect oil emissions. This sampling distance can minimize the effect of the turbulence from the range hood.
In addition, the flow rate of range hoods in general households is about 15 m3/min. In order to avoid affecting the sampling results due to the turbulence from the range hood, this experiment set a reasonable flow rate as 4 m3/min based on previous research.
Real-time particle concentrations
Levels of particle mass and number concentrations, and BC concentration with real-time measurements obtained from emissions of deep-frying three cooking oils are presented in Table 1. We found the total particle number concentration emitted from palm oil ((3896 ± 1797) × 103#/cm3) was significantly higher than that from soybean oil ((469 ± 476) × 103#/cm3) and olive oil ((400 ± 156) × 103#/cm3). In particular, a higher magnitude of particle number concentrations in Aitken (20–100 nm) mode and in accumulation mode (100–1000 nm) was observed for palm oil.
Although the suitability of palm oil for frying has been widely propagated due to its oxidative stability24, the potential health hazard from the elevated level of ultrafine particles (20–100 nm) during deep-frying should not be overlooked. Consistent with the above phenomenon, the particle number concentration was positively correlated with palmitic acid (rs = 0.78, p < 0.05), a major SAFA in palm oil (Supplementary Table 5). Previous studies also demonstrated that SAFA emitted higher particle number concentration than PUFA and MUFA-rich oils25,26,27 and palmitic acid has been shown to be the predominant component of cooking-derived particulate matter28,29. Compared to PUFA or MUFA-rich oils, palm oil has a large quantity of small SAFA molecules and less polymerization to form large nonvolatile molecules during deep-frying30. We also found a positive correlation between the particle number concentration and total polar components (TPCs) (rs = 0.68, p < 0.05). The TPCs refer to degraded products from the triglycerides during the deep-frying process, including fatty acids, aldehydes, ketones, alcohol, and nonvolatile products31. The result implies more degradation occurred in palm oil than in the other two. We also noticed that the magnitude of variation of the total number concentration of the soybean oil is relatively higher than those of palm oil and olive oil. This may be due to the fact that PUFA contains more carbon-carbon double bonds so their structures are unstable compared to SFA and MUFA32. Based on our experience, we would recommend to include more replicates for future studies.
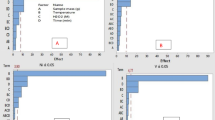
Figure 1 illustrated the particulate size distributions with respect to numbers and mass concentrations. We observed a unimodal distribution for particulate number concentration and a bimodal distribution for particulate mass concentration in all three oils. The deep-frying with palm oil, soybean oil, and olive oil produced the peak concentration of the particulate number at diameters of 57.3, 109.4, and 94.7 nm, respectively. Since similar particulate mass concentration was observed across three oils, it appears the smaller the emitted molecules the greater the particle number concentration and the larger the emitted molecules the lower the number concentration of particles. The size distribution of particles from olive oil is similar to that from other cooking oils with high oleic acid content such as the rapeseed oil33.
Comparing particulate size distributions across three oils, a using mean* particulate number conc. generated as intensity or b using mean* particulate mass conc. as intensity, during the 2-h process of deep-frying French fries at 180 °C. *Mean of three repeats, each repeat provides the mean number conc. and the mean mass conc. of 120 data points (one per minute) for each oil (The blue, red, and green circles represent soybean oil, palm oil, and olive oil, respectively).
Figure 2 shows the particle number concentrations, particle mass concentrations, and BC concentrations changed along with the 12 consecutive batches of frying. The particle number and mass concentrations as well as BC concentrations increased quickly within a few minutes and reached the peak levels for all three oils for deep-frying French fries. The mean concentration of BC generated from soybean oil, palm oil, and olive oil was 0.93 ± 0.80, 1.99 ± 1.01, and 1.74 ± 1.05 μg/m3, respectively (Table 1). Although the mean levels were not statistically different, a positive correlation between the average BC concentration and TPC was observed (rs = 0.68, p < 0.05) and soybean oil peaked much higher than the other two oils (Fig. 2c). Previous studies had found that a huge amount of BC concentration may be emitted with traditional cookstoves34,35. Cooking with biomass solid fuels is one of the major sources of BC and the average concentration of BC ranges from 5.4 to 34.9 μg/m3 in household kitchens with biomass solid fuels15,36,37. Compared with previous studies using biomass solid fuels, BC levels (<2 μg/m3) in our study was much lower. It is likely due to the lower heating temperature (180 °C) and the use of an electric fryer. The positive correlations between TPC and the average particle number concentration (rs = 0.68, p = 0.042) and between TPC and BC concentration (rs = 0.68, p = 0.042) were observed (Supplementary Table 5). This is the first to correlate cooking emission of pollutant concentrations and oil quality indices. The correlation results may provide some clues and suggestions for further studies on reducing cooking oil emissions with respect to oil properties.
a Particle number, b particle mass, and c black carbon. Y axis (RT/BK): The fold change was calculated by dividing real-time measurement levels (RT) (one per minute) by the mean background (BK) value. X axis: batch number for frying French fries. BK: 10 min-background measurements before heating oil. PreH: 10 min measurements during the preheating period before frying French fries. (The red, blue, and green lines represent soybean oil, palm oil, and olive oil, respectively).
Gaseous- and particle-phase PAHs
The gaseous- and particle-phase concentrations of 21 PAH compounds during deep-frying French fries of three oils were shown in Table 2. The total PAHs concentrations (gaseous + particle) were reported as 22.43 ± 18.62 ng/m3, 16.44 ± 7.09 ng/m3, and 7.32 ± 10.5 ng/m3 for soybean, palm, and olive oils, respectively. Naphthalene was the predominant gaseous-phase PAHs, accounting for 97, 62, and 87% of the gaseous-phase PAHs for soybean oil, palm oil, and olive oil, respectively. Chen et al. also found that naphthalene (67–89%) was the most abundant gaseous-phase PAHs among all exhaust samples gathered from commercial restaurants38. The particle-phase PAHs with high molecular weights containing 5–7 aromatic rings are toxic and carcinogenic and are considered to be hazardous substances to health39. Palm oil emitted significantly higher particle-phase PAHs than soybean oil and olive oil. Cyclopenta(c,d)pyrene was the predominant particle-phase PAHs, accounting for 62, 56, and 37% of the particle-phase PAH for soybean oil, palm oil, and olive oil, respectively. Benzo(a)pyrene listed as a Group 1 carcinogen by the International Agency Research on Cancer (IARC) was found in particle-bound PAHs in all three oils with concentrations of 0.039 ± 0.067, 0.11 ± 0.097, and 0.14 ± 0.023 ng/m3 in soybean oil, palm oil, and olive oil, respectively.
The mechanisms of PAH formation has been primarily studied in combustion. Fewer studies were based on heating lipids. SAFA and palmitic acid may contribute to the formation of PAHs (Supplementary Table 7), since positive correlations were found between palmitic acid and acenaphthene (rs = 0.74, p < 0.05) and benzo(e)pyrene (rs = 0.79, p < 0.05) and a positive relationship was also observed between chrysene and total SAFA (rs = 0.86, p < 0.01). A positive correlation (rs = 0.72, p < 0.05) between benzo(k)fluoranthrene and oleic acid was found, although most PAHs were not correlated with oleic acid percentage and the total amount of PAHs emitted is very low for olive oil. These results are consistent with previous model lipids study which found methyl oleate produced more benzo(k)fluoranthrene than methyl linolenate, methyl linoleate, and methyl stearate40. The TPC concentrations were significantly correlated with the concentrations of acenaphthene (rs = 0.83, p < 0.05) and benzo(e)pyrene (rs = 0.79, p < 0.05), but there was no significant correlations between the total PAH concentration and TPC in our study, although a previous study found that TPC was significantly correlated with the concentrations of the sum of the 16 PAHs in fried oil41.
Gaseous- and particle-phase aldehydes
Table 3 shows the mean concentrations of 19 gaseous- and particle-phase aldehydes during deep-frying French fries with three cooking oils. The mean concentrations of total aldehydes were significantly higher in soybean oil (3655 ± 598 μg/m3) than those in olive oil (2453 ± 1304 μg/m3) and palm oil (2197 ± 841 μg/m3), indicating oil rich in PUFA emitted more aldehydes than that in MUFA and SAFA. Previous studies found that oil containing more PUFA emitted more aldehydes than other oils during deep-frying1,42.
For individual aldehydes, the top three were hexanal (1030 ± 207 μg/m3), trans-2-heptenal (826 ± 196 μg/m3), and acrolein (673 ± 110 μg/m3) for soybean oil; hexanal (634 ± 277 μg/m3), nonanal (438 ± 159 μg/m3), and trans-2-heptenal (399 ± 162 ug/m3) for palm oil; and nonanal (637 ± 269 μg/m3g/m3), hexanal (549 ± 477 μg/m3), and trans-2-heptenal (394 ± 332 μg/m3) for olive oil. Soybean oil emitted higher concentrations of aldehydes than palm oil and olive oil, in particular acrolein, propionaldehyde, crotonaldehyde, hexanal, 2-heptenal, and trans-2 nonanal in gaseous-phase and acetone in particulate-phase. Acrolein may be generated from amino acids, lipids, or carbohydrates43 and has been considered as a major cigarette-related lung cancer-causing agent44,45. While previous study results were inconsistent with respect to the potential formation of acrolein from PUFA46,47,48, the current study showed a strong relationship between acrolein and the α-linolenic acid (rs = 0.72, p < 0.05) (Supplementary Table 6). Perilla oil which is rich in linolenic acid emitted the highest acrolein concentration during heating compared to rice bran oil, rapeseed oil, soybean oil, and sunflower oil46. Crotonaldehyde is also a possible human carcinogen, although there are no human carcinogenicity data49.
Other aldehydes also showing a strong correlation with α-linolenic acid (Supplementary Table 6) include propinoaldehyde (rs = 0. 86, p < 0.05), crotonaldehyde (rs = 0.85, p < 0.05), hexanal (rs = 0.83, p < 0.05), trans-2-heptenal (rs = 0.83, p < 0.05) and trans-2-nonenal (rs = 0.78, p < 0.05). These aldehydes were detected higher in oils rich in linolenic acid also in previous studies50,51. On the other hand, hexanal (rs = 0.73, p < 0.05) and trans-2-heptenal (rs = 0.67, p < 0.05) showed a high correlation with linoleic acid. Nonanal was the only aldehyde which was detected in higher concentration in olive oil emission than soybean oil and palm oil and the correlation analysis indicated that the nonanal emission level was related with MUFA content in oils (rs = 0.67, p < 0.05).
The previous study has shown that the fatty acid with more double bonds are more prone to oxidation50. The relative oxidation rates of methyl oleate, linoleate, and linolenate were 1: 10.3: 21.6. Hydroperoxides are the primary oxidation products first formed during the autoxidation of lipid. The hydroperoxides are quickly decomposed into various volatile compounds, including aldehydes. Our findings are consistent with the above phenomenon such that the correlation between the levels of aldehydes and PUFAs were positive, which indicated that PUFAs contribute to the generation of aldehydes during frying French fries at 180 °C.
In conclusion, fried oil fatty acids composition and oil quality are significantly correlated with particle and gas contaminants of emissions during deep-frying French fries. The main pollutants during deep-frying French fries were particulate matter and aldehydes. Soybean oil emitted the highest level of gaseous aldehydes. Palm oil emitted the most particle-bound PAHs. Olive oil seems superior to soybean and palm oils with regards to toxic emissions during deep-frying.
Methods
Cooking materials and procedure
The experiments were conducted in a simulated kitchen (L × W × H = 3.25 m × 3.10 m × 2.75 m) at the National Health Research Institutes in Taiwan. The deep-frying process was performed in an electric fryer (5.0 L, 2000 W, L × W = 30 cm × 15.3 cm; WFT-4L, WISE Inc., Taipei, Taiwan). As shown in Fig. 3, the electric fryer was placed on the table (1.1 m above the floor) under the kitchen range hood. The hood (L × W = 89 cm × 52 cm; DR-7790ASXL, Sakura Corp., Taichung, Taiwan) was placed at 70 cm above the electric fryer (i.e., 1.8 m above the floor). We set up a backboard from the range hood to the table on the backside of the electric fryer, while front, left, and right sides of the electric fryer are opened. The default flow rate of the range hood was at 4 m3/min. Three popular used cooking oils, soybean oil (53.0% linoleic acid), palm oil (39.7% palmitic acid), and olive oil (72.5% oleic acid) were purchased from the local supermarket. The details on fatty acid composition, oil quality indices, and the analytical methods were provided in Supplementary Method 1 and Supplementary Table 1.
A: XAD-2 for gas-phase PAH samples; B: PEM for PM 2.5-PAH samples; C: PEM with DNPH-coated glass fiber filters for PM2.5-aldehyde collection; D: DNPH-coated silica cartridge for gas-phase aldehyde collection; E: Electric fryer; F: Ventilation range hood; G: SMPS; H: AE33 for real-time BC analysis; I: APS.
For each deep-frying process, 3.5 liters of cooking oil was used for frying consecutively 12 batches of French fries. Each batch contains 175 g of French fries (Ya Fang Inc., Taiwan). All experiments were performed following the same procedure: preheating oil for 10 min until the temperature reached 180 °C, adding French fries and frying for 8 min, and then turning off the heat for a 2-min break before the next batch. During the deep-frying process, the windows of the simulated kitchen on the side nearby the cookstove are shut to minimize the environmental draft from the outdoor. To avoid returned contaminants, we also installed the electrostatic precipitator after the range hood duct to remove the exhausted oil fume. The supply air came from the room space and outdoor.
Sampling procedure
The concentrations of the particle number, particle mass, gaseous- and particle-phase PAHs, gaseous- and particle-phase aldehydes, and black carbon (BC) were collected from the cooking emissions of the deep-frying process. During the frying experiments, all the doors and windows were shut to avoid the inflow and environmental draft from ambient air. The sampling inlets were placed 15 cm beneath the range hood and 40 cm above the electric fryer to collect oil emissions. This sampling distance was designed to minimize the effect of the turbulence created by sucking. The kitchen air was purged by the fan for 30 min before and after the frying procedures with the door/window open. Here, we measured the particle number concentration to confirm if the concentration reach the background condition in the testing environment. For each test, the sampling process consists of 10 min-background conditions, 10 min preheating, and 120 min deep-frying (repeated 12 times) using real-time monitors and integrated samplers. The experiment was repeated three times for each cooking oil. The sampling tubes were cleaned and purged before we switched to a new testing oil.
To obtain a wide size range of particles, cooking emissions were monitored by a scanning mobility particle sizer (SMPS, TSI Inc., classifier model 3080, CPC model 3775) and an aerodynamic particle sizer spectrometer 3321(APS, TSI Inc.) for the particle number and mass concentrations. The SMPS is able to classify ultrafine particles (0.02–0.54μm) into 93 size categories and the APS can classify particles from 0.55 to 19.81 μm into 51 channels. The dominant particle size fraction and typical peak concentrations generated by different oils during deep-frying can be determined.
The particle size we measured ranged from 20.2 to 552.3 nm using SMPS and 0.523 to 19.81 μm using APS. The particle size distributions were then classified into Aitken mode (particle diameter between 20 to 100 nm), accumulation mode (particle diameter between100 to 1000 nm), and the coarse mode (>1000 nm) according to diameter52. The particle mass concentration was also calculated from the number concentration of the SMPS and APS using the effective density of 0.9 g/cm3. BC was monitored by an aethalometer (AE33, Magee Scientific, CA, USA) at the wavelength of 880 nm with a time resolution of 60 s (Supplementary Method 2).
For the PAHs, particle-phase PAHs were sampled at the flow of 10 L/min with quartz filters (37 mm, 1 μm, Whatman, UK) installed in Personal Environmental Monitor (PEM) (SKC, PA, USA) with a PM2.5 size and gaseous-phase was collected by a XAD-2 cartridge (SKC, Blandford, Forum, UK) at the flow of 1.0 L/min using the linear air pump (Hiblow HP150, USA). PAHs were extracted by dichloromethane and hexane mixture (2:1) and then analyzed by GC-MS/MS with multiple reaction monitoring (MRM) mode. The method detection limit (MDL) ranged from 0.63 to 2.57 ng/ml for the selected compounds (Supplementary Table 2). A known amount of PAH mixture standards was added to blank mediums through the same procedure of the analyzing samples to evaluate the recovery rate. The recovery efficiency of all compounds ranged from 73.6 to 128% (Supplementary Method 3 and Supplementary Table 2).
As for the aldehydes, particle-phase aldehydes (sampling flow rate = 10 L/min) were collected by 2, 4-DNPH-coated glass fiber filters (37 mm, 1 μm, Supelco, PA, USA) installed in PEM with a PM2.5 size. The gaseous-phase aldehydes were collected by a silica cartridge coated with 2, 4-DNPH (Supelco, PA, USA) after the filter sampler with a linear air pump (Hiblow HP150, USA) at a flow rate of 1.0 L/min. The flow rates of all samplers for each particle and gas sampling were adjusted using a calibration rotameter (MesaLabs Defender 520). The aldehydes in the filter and cartridge were extracted by 5 ml acetonitrile and analyzed by high-performance liquid chromatography (HPLC, PU-2089, Jasco, Japan) (Supplementary Method 4). The gradient program for HPLC aldehydes analysis is detailed in Supplementary Table 3. The MDL ranged from 0.008 to 0.058 μg/ml for the selected compounds (Supplementary Table 4). About 20 μl mixture of the aldehyde standards (10 μg/ml) was added to blank mediums (filter and silica cartridge) to evaluate the recovery efficiency. The recovery efficiency of all compounds ranged from 48.0 to 99.1% (Supplementary Table 4). When the value of PAHs and aldehydes for a given sample fell below the limit of detection (LOD) value, we assigned the corresponding sample as a value of LOD/2 for that compound.
The fatty acid composition of the fresh oils was analyzed by the Official AOAC 996.06 method53. Acid value (AV) and peroxide value (POV) were analyzed by official CNS 3647 N6082 and CNS 3650 N6085. Total polar compounds (TPC) were analyzed by the column chromatography method (AOAC 982.27). The detailed information on sampling, analysis methods, and QA/QC data for BC, PAHs, and aldehydes is provided in supplementary Methods.
Statistical analysis
Non-parametric Kruskal–Wallis test was used to test whether there were significant differences among cooking emissions of three oils in concentrations of particulate matter, BC, PAHs, and aldehydes. Spearman rank correlation was used to evaluate the relationship between major fatty acid (PUFA, MUFA, or SAFA) percentage, oil quality indices (AV, POV, and TPC), and emission concentrations of the particle mass, number, and selected compounds. The time sequence curves for emission quantities of the particle mass, particle number, and BC concentrations and the correlation scatter plot were made with SigmaPlot 12.0 software (Systat Software Inc.).
Data availability
All data are available from the authors upon reasonable request.
References
Peng, C. Y., Lang, C. H., Lin, P. C. & Kuo, Y. C. Effects of cooking method, cooking oil, and food type on aldehyde emissions in cooking oil fumes. J. Hazard Mater. 324, 160–167 (2017).
Abdullahi, K. L., Delgado-Saborit, J. M. & Harrison, R. M. Emissions and indoor concentrations of particulate matter and its specific chemical components from cooking: A review. Atmos. Environ. 71, 260–294 (2013).
Wang, L. et al. Role of Chinese cooking emissions on ambient air quality and human health. Sci. Total Environ. 589, 173–181 (2017).
Allan, J. D. et al. Contributions from transport, solid fuel burning and cooking to primary organic aerosols in two UK cities. Atmos. Chem. Phys. 10, 647–668 (2010).
Ots, R. et al. Model simulations of cooking organic aerosol (COA) over the UK using estimates of emissions based on measurements at two sites in London. Atmos. Chem. Phys. 16, 13773–13789 (2016).
Yu, I. T. S., Chiu, Y. L., Au, J. S. K., Wong, T. W. & Tang, J. L. Dose-response relationship between cooking fumes exposures and lung cancer among Chinese nonsmoking women. Cancer Res. 66, 4961–4967 (2006).
Zhong, L., Goldberg, M. S., Gao, Y. T. & Jin, F. Lung cancer and indoor air pollution arising from Chinese-style cooking among nonsmoking women living in Shanghai, China.Epidemiology 10, 488–494 (1999).
Gao, Y. T. et al. Lung cancer among Chinese women. Int.J. Cancer 40, 604–609 (1987).
Xue, Y. B., Jiang, Y., Jin, S. & Li, Y. Association between cooking oil fume exposure and lung cancer among Chinese nonsmoking women: a meta-analysis. Oncotargets Ther. 9, 2987–2992 (2016).
Shields, P. G. et al. Mutagens from heated Chinese and Us cooking oils. J. Natl Cancer Inst. 87, 836–841 (1995).
Chen, T. Y. et al. Impact of cooking oil fume exposure and fume extractor use on lung cancer risk in non-smoking Han Chinese women. Sci. Rep. 10, 6774 (2020).
Katragadda, H. R., Fullana, A., Sidhu, S. & Carbonell-Barrachina, A. A. Emissions of volatile aldehydes from heated cooking oils. Food Chem. 120, 59–65 (2010).
Tiwari, J., Tarale, P., Sivanesan, S. & Bafana, A. Environmental persistence, hazard, and mitigation challenges of nitroaromatic compounds. Environ. Sci. Pollut. Res. Int. 26, 28650–28667 (2019).
Wu, M. T., Lin, P. C., Pan, C. H. & Peng, C. Y. Risk assessment of personal exposure to polycyclic aromatic hydrocarbons and aldehydes in three commercial cooking workplaces. Sci. Rep. 9, 1661 (2019).
Ravindra, K. Emission of black carbon from rural households kitchens and assessment of lifetime excess cancer risk in villages of North India. Environ. Int. 122, 201–212 (2019).
Van Vliet, E. D. et al. Personal exposures to fine particulate matter and black carbon in households cooking with biomass fuels in rural Ghana. Environ. Res. 127, 40–48 (2013).
Wan, M.-P. et al. Ultrafine particles, and PM2.5 generated from cooking in homes. Atmos. Environ. 45, 6141–6148 (2011).
Zhang, Q. F., Gangupomu, R. H., Ramirez, D. & Zhu, Y. F. Measurement of ultrafine particles and other air pollutants emitted by cooking activities. Int J. Env. Res. Pub. He. 7, 1744–1759 (2010).
Li, S. et al. Determination of time- and size-dependent fine particle emission with varied oil heating in an experimental kitchen. J. Environ. Sci. 51, 157–164 (2017).
Cassee, F. R. et al. Particle size-dependent total mass deposition in lungs determines inhalation toxicity of cadmium chloride aerosols in rats. Application of a multiple path dosimetry model. Arch. Toxicol. 76, 277–286 (2002).
Schraufnagel, D. E. The health effects of ultrafine particles. Exp. Mol. Med. 52, 311–317 (2020).
Woo, K. S. et al. The impact of particulate matter air pollution (PM2.5) on atherosclerosis in modernizing China: a report from the CATHAY study. Int. J. Epidemiol. 50, 578–588 (2021).
Kozliak, E. et al. Pathways toward PAH formation during fatty acid and triglyceride pyrolysis. J. Phys. Chem. A 124, 7559–7574 (2020).
Matthaus, B. Use of palm oil for frying in comparison with other high-stability oils. Eur. J. Lipid Sci. Tech. 109, 400–409 (2007).
Liu, T. Y., Wang, Z. Y., Huang, D. D., Wang, X. M. & Chan, C. K. Significant production of secondary organic aerosol from emissions of heated cooking oils. Environ. Sci. Tech. Let. 5, 32–37 (2018).
Gao, J. et al. Determination of size-dependent source emission rate of cooking-generated aerosol particles at the oil-heating stage in an experimental kitchen. Aerosol Air Qual. Res. 13, 488–496 (2013).
Torkmahalleh, M. A., Kaibaldiyeva, U. & Kadyrbayeva, A. A new computer model for the simulation of particulate matter formation from heated cooking oils using Aspen Plus. Build Simul. China 10, 535–550 (2017).
Zhao, Y. L., Hu, M., Slanina, S. & Zhang, Y. H. The molecular distribution of fine particulate organic matter emitted from Western-style fast food cooking. Atmos. Environ. 41, 8163–8171 (2007).
Wang, G. et al. Chemical characteristics of fine particles emitted from different Chinese cooking styles. Aerosol Air Qual. Res. 15, 2357–2366 (2015).
Bastida, S. & Sanchez-Muniz, F. J. Thermal oxidation of olive oil, sunflower oil and a mix of both oils during forty discontinuous domestic fryings of different foods. Food Sci. Technol. Int. 7, 15–21 (2001).
Xu, X. Q., Tran, V. H., Palmer, M., White, K. & Salisbury, P. Chemical and physical analyses and sensory evaluation of six deep-frying oils. J. Am. Oil Chem. Soc. 76, 1091–1099 (1999).
Takashima, S., Toyoshi, K., Yamamoto, T. & Shimozawa, N. Positional determination of the carbon-carbon double bonds in unsaturated fatty acids mediated by solvent plasmatization using LC-MS. Sci. Rep. 10, 12988 (2020).
Yeung, L. L. & To, W. M. Size distributions of the aerosols emitted from commercial cooking processes. Indoor Built. Environ. 17, 220–229 (2008).
Patange, O. S. et al. Reductions in indoor black carbon concentrations from improved biomass stoves in rural India. Environ. Sci. Technol. 49, 4749–4756 (2015).
Soneja, S. I. et al. Determining particulate matter and black carbon exfiltration estimates for traditional cookstove use in rural Nepalese Village households. Environ. Sci. Technol. 49, 5555–5562 (2015).
Rupakheti, D. et al. Indoor levels of black carbon and particulate matters in relation to cooking activities using different cook stove-fuels in rural Nepal. Energy Sustain. Dev. 48, 25–33 (2019).
Saliba, G. et al. Aerosol optical properties and climate implications of emissions from traditional and improved cookstoves. Environ. Sci. Technol. 52, 13647–13656 (2018).
Chen, Y. et al. Gaseous and particulate polycyclic aromatic hydrocarbons (PAHs) emissions from commercial restaurants in Hong Kong. J. Environ. Monit. 9, 1402–1409 (2007).
Chang, K. F., Fang, G. C., Chen, J. C. & Wu, Y. S. Atmospheric polycyclic aromatic hydrocarbons (PAHs) in Asia: a review from 1999 to 2004. Environ. Pollut. 142, 388–396 (2006).
Chen, B. H. & Chen, Y. C. Formation of polycyclic aromatic hydrocarbons in the smoke from heated model lipids and food lipids. J. Agric. Food Chem. 49, 5238–5243 (2001).
An, K. J., Liu, Y. L. & Liu, H. L. Relationship between total polar components and polycyclic aromatic hydrocarbons in fried edible oil. Food Addit. Contam. A 34, 1596–1605 (2017).
Klein, F. et al. Characterization of gas-phase organics using proton transfer reaction time-of-flight mass spectrometry: cooking emissions. Environ. Sci. Technol. 50, 1243–1250 (2016).
Abraham, K. et al. Toxicology and risk assessment of acrolein in food. Mol. Nutr. Food Res. 55, 1277–1290 (2011).
Feng, Z. H., Hu, W. W., Hu, Y. & Tang, M. S. Acrolein is a major cigarette-related lung cancer agent: preferential binding at p53 mutational hotspots and inhibition of DNA repair. Proc. Natl Acad. Sci. USA 103, 15404–15409 (2006).
Alwis, K. U., deCastro, B. R., Morrow, J. C. & Blount, B. C. Acrolein exposure in US tobacco smokers and non-tobacco users: NHANES 2005-2006. Environ. Health Persp. 123, 1302–1308 (2015).
Endo, Y. et al. Linolenic acid as the main source of acrolein formed during heating of vegetable oils. J. Am. Oil Chem. Soc. 90, 959–964 (2013).
Ewert, A., Granvogl, M. & Schieberle, P. Development of two stable isotope dilution assays for the quantitation of acrolein in heat-processed fats. J. Agr. Food Chem. 59, 3582–3589 (2011).
Ewert, A., Granvogl, M. & Schieberle, P. Isotope-labeling studies on the formation pathway of acrolein during heat processing of oils. J. Agr. Food Chem. 62, 8524–8529 (2014).
Bagchi, P. et al. Crotonaldehyde exposure in US tobacco smokers and nonsmokers: NHANES 2005-2006 and 2011-2012. Environ. Res. 163, 1–9 (2018).
Cao, J. et al. Novel approach to evaluate the oxidation state of vegetable oils using characteristic oxidation indicators. J. Agr. Food Chem. 62, 12545–12552 (2014).
Granvogl, M. Development of three stable isotope dilution assays for the quantitation of (E)-2-butenal (Crotonaldehyde) in heat-processed edible fats and oils as well as in food. J. Agr. Food Chem. 62, 1272–1282 (2014).
Raes, F. et al. Formation and cycling of aerosols in the global troposphere. Atmos. Environ. 34, 4215–4240 (2000).
Shin, J. M. et al. Comparison of different methods to quantify fat classes in bakery products. Food Chem. 136, 703–709 (2013).
Acknowledgements
This study was supported by the Center for Sustainability Science in Academia Sinica (AS-SS-107-03). The authors would like to express their appreciation to the Institute of Biomedical Science at Academia Sinica for their administrative support. The authors acknowledge National Health Research Institutes for providing the experimental kitchen and instrumental support.
Author information
Authors and Affiliations
Contributions
W.-H.P. conceived the idea. L.X., C.-Y.P., and Y.-C.C. carried out the experiments. W.-H.P., Y.-C.C., and S.-C.C.L. supervised the project. L.X. drafted the manuscript. K.-M.C. and W.-H.P. finalized the manuscript. K.-M.C. and L.X. contributed equally to this article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chiang, KM., Xiu, L., Peng, CY. et al. Particulate matters, aldehydes, and polycyclic aromatic hydrocarbons produced from deep-frying emissions: comparisons of three cooking oils with distinct fatty acid profiles. npj Sci Food 6, 28 (2022). https://doi.org/10.1038/s41538-022-00143-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41538-022-00143-5