Abstract
Asthma and COPD are defined as different disease entities, but in practice patients often show features of both diseases making it challenging for primary care clinicians to establish a correct diagnosis. We aimed to establish the added value of spirometry and more advanced lung function measurements to differentiate between asthma and COPD. A cross-sectional study in 10 Dutch general practices was performed. 532 subjects were extensively screened on respiratory symptoms and lung function. Two chest physicians assessed if asthma or COPD was present. Using multivariable logistic regression analysis we assessed the ability of three scenarios (i.e. only patient history; diagnostics available to primary care; diagnostics available only to secondary care) to differentiate between the two conditions. Receiver operator characteristics (ROC) curves and area under the curve (AUC) were calculated for each scenario, with the chest physicians’ assessment as golden standard. Results showed that 84 subjects were diagnosed with asthma, 138 with COPD, and 310 with no chronic respiratory disease. In the scenario including only patient history items, ROC characteristics of the model showed an AUC of 0.84 (95% CI 0.78–0.89) for differentiation between asthma and COPD. When adding diagnostics available to primary care (i.e., pre- and postbronchodilator spirometry) AUC increased to 0.89 (95% CI 0.84–0.93; p = 0.020). When adding more advanced secondary care diagnostic tests AUC remained 0.89 (95% CI 0.85–0.94; p = 0.967). We conclude that primary care clinicians’ ability to differentiate between asthma and COPD is enhanced by spirometry testing. More advanced diagnostic tests used in hospital care settings do not seem to provide a better overall diagnostic differentiation between asthma and COPD in primary care patients.
Similar content being viewed by others
Introduction
Asthma and chronic obstructive pulmonary disease (COPD) are both common chronic respiratory diseases affecting approximately 1 in 12 people worldwide1,2. The two conditions are defined as different disease entities with unique pathophysiological mechanisms and characteristic clinical features1,2. The underlying pathophysiology in COPD is characterized predominantly by neutrophilic inflammation, whereas in asthma the inflammatory pattern is mostly due to eosinophilic inflammation3. Asthma typically presents with intermittent respiratory symptoms caused by airflow obstruction predominantly due to bronchial hyperresponsiveness4. Asthma is often presented at younger age as part of an atopic constitution, but can also be diagnosed in adulthood1. In contrast, COPD is a slowly progressive lung disease with patients having persistent respiratory symptoms and airflow obstruction2. In high-income countries like the Netherlands COPD usually presents in patients older than forty who are generally current or former smokers2. Patients with asthma or COPD are mostly diagnosed and managed by primary care clinicians.
Looking at the classic pathophysiological and clinical presentations, the distinction between asthma and COPD seems clear, but in clinical practice patients often show features of both diseases5,6. These similarities make it difficult for clinicians to distinguish between asthma and COPD7, especially in older and more diverse patient populations encountered in primary care8,9,10. However, differentiating between the two respiratory conditions is important as they have different pharmacotherapeutic regimens. In patients with asthma, inhaled corticosteroids (ICS) are highly effective in reducing symptoms and reducing the risk of asthma-related mortality1. In contrast, patients with COPD respond poorly to ICS and are mainly treated with (long-acting) bronchodilators to relieve symptoms2. In addition to this, misdiagnosing asthma for COPD could lead to serious health risks considering that monotherapy with long-acting bronchodilators is contra-indicated in asthmatics since it increases the risk of severe exacerbation11,12,13. On the other hand, (unnecessary) treatment with ICS may cause pneumonia and increased risk of osteoporosis14,15,16,17.
Thus, establishing a correct diagnosis is essential for optimal treatment of asthma and COPD, but this can be challenging for primary care clinicians. Supporting them in the diagnostic process seems therefore essential, but this also depends on the availability of diagnostic tools. Although quality spirometry has shown to be feasible in primary care settings18 there is substantial room for improvement of its use to accurately diagnose chronic respiratory diseases19,20. Thus, the first aim of our current study was to establish which patient characteristics distinguish between patients diagnosed with asthma or COPD. The second and main aim was to establish the added value of spirometry and more advanced lung function measurements to differentiate between these two chronic airways diseases.
Methods
Study design and population
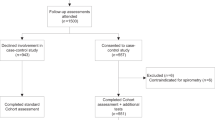
In this observational multi-centre cross-sectional study, we compared patients diagnosed with asthma, patients diagnosed with COPD, and subjects without underlying chronic obstructive lung conditions using data from a previous study, i.e., the Detection, Intervention and Monitoring of COPD’ (DIMCA) program21. This program was originally set up to improve early detection of chronic airways disease in general practices. A random sample of 1,749 adult subjects (20–70 years) from ten general practices in The Netherlands were invited to participate21. At the start of the program, patients with pre-existing asthma, COPD or another airway disease were excluded. In 2007, ten years after the start of the initial DIMCA program, all subjects (now aged 30–80 years) received an invitation for a comprehensive respiratory assessment consisting of extensive lung function measurements and a myriad of medical history questions22. A total of 532 subjects agreed to participate in this follow-up study. The results of the respiratory assessment of these subjects were submitted to two experienced chest physicians who assessed if a chronic airways disease (i.e., COPD or asthma) was present or absent using a standardized protocol22 that was based on the international clinical guideline criteria that applied at the time of the study (see below). The results of the chest physicians’ assessments were used as the golden standard in the current study.
The study was approved by the medical ethics review board CMO Regio Arnhem – Nijmegen (https://www.radboudumc.nl/over-het-radboudumc/kwaliteit-en-veiligheid/commissie-mensgebonden-onderzoek; file number: 2002/028). Participants provided written informed consent to take part in the study.
Measurements
Study participants were instructed to interrupt the use of any bronchodilators they might use for a specified number of hours before their visit to the pulmonary function laboratory. Lung function testing involved pre- and postbronchodilator spirometry (both static and dynamic) and measurement of carbon monoxide diffusion capacity (DLCO) and bronchial hyperresponsiveness (BHR)22. Aerosolized salbutamol 800 µg and/or ipratropium 160 µg were used as bronchodilators and were administered by volume spacer. Postbronchodilator forced expiratory volume in one second (FEV1) was measured 15 min after salbutamol and 45 min after ipratropium. Bronchodilator reversibility was defined as an increase in FEV1 after bronchodilation by at least 12% and 200 mL. BHR was assessed by histamine challenge test and considered positive in case of a >20% drop in FEV1 at a provocative dose histamine of ≤8 mg/mL (PC20)1,23. All lung function tests were conducted by certified lung function technicians in a hospital-based pulmonary function laboratory and were performed in accordance with the 1994 American Thoracic Society standards24. Predicted normal lung function values for FEV1 were calculated using European Community for Coal and Steel reference values25. Following lung function testing, subjects were interviewed by the lung function technician regarding respiratory symptoms, smoking behaviour, presence of allergies and eczema, respiratory problems triggered by environmental exposures, and family history of COPD or asthma22.
Diagnostic assessment
Based on the results of the respiratory assessment the chest physicians assessed if a chronic airways disease (i.e., asthma or COPD) was present or absent using guideline criteria, their expert knowledge, and their clinical expertise22. Study subjects were randomly assigned to the chest physicians in a 1:1 ratio. If a subject was diagnosed with a chronic airways disease by the assigned chest physician the subject’s data was also presented to the other chest physician and a final joint diagnosis was established. To standardize the diagnostic process, a decision tree (Fig. 1) was created based on international clinical guideline criteria for diagnosing asthma (GINA guideline, 2007 update26) and COPD (GOLD guideline, 2006 update27) that applied at the time, in co-operation with the two chest physicians. In case of uncertainty about the respiratory diagnosis the chest physicians could request additional diagnostic tests (i.e., allergy skin testing, peak expiratory flow (PEF) monitoring) in order to maximize their diagnostic certainty22. Because the concept of asthma-COPD overlap (ACO) was introduced after the current study was conducted, the chest physicians did not consider a diagnosis of ACO as a part of their assessment. They were instructed to, based on their systematic assessment of all diagnostic information available, assign one single preferred diagnosis (i.e., either asthma or COPD) that fitted best according to their expert opinion. Figure 2 illustrates the spectrum of chronic obstructive airways disease diagnoses and the parts of the spectrum on which the current study focuses. Strictly for the purpose of describing the study population and its diagnostic subgroups (see Table 1) the Global Lung function Initiative (GLI) reference equations were applied at the time of the data analysis for the current paper28.
Categorization of variables
In the present study, we categorized all items of the respiratory assessment in three subsections based on their availability in different healthcare settings, i.e., public health, primary care, and secondary care (Table 2). Subsection 1 consists of items that are available in any public health or healthcare setting since they require no measurements or testing equipment but only medical history questions (i.e., respiratory symptoms, smoking behaviour, body mass index (BMI)). Subsection 2 contains lung function test results that are available to primary care clinicians (i.e., spirometry and reversibility testing) in countries with well-developed healthcare systems29,30,31. Finally, Subsection 3 contains results from more advanced diagnostic tests as performed mainly in lung function laboratories in hospital care settings. These tests include measurement of static lung volumes, diffusion capacity, and histamine challenge testing.
Statistical analysis
Demographic characteristics, clinical features and lung function values were univariately compared between the subgroups of patients diagnosed with asthma and COPD using independent t-tests and Chi-square tests. The further analysis focussed on assessing the ability to differentiate between these chronic obstructive lung diseases in different healthcare settings. Since physicians are not limited to asking a single medical history question or to conducting a single diagnostic test, we used multivariable logistic regression analysis to construct predictive models based on the data of the subjects who were diagnosed with asthma or COPD by the chest physicians (i.e., the binary outcome measure for this analysis was to have a diagnosis of asthma or a diagnosis of COPD). As described above, the items from the patient assessment were categorized in three subsections based on diagnostic availability and multivariable logistic regression models were run for three ‘scenarios’ (Table 2). In the first scenario, we only used the medical history items from Subsection 1 in the model. In the second scenario, we added diagnostic items available to primary care clinicians (i.e., Subsections 1 plus 2) to the model. In the third scenario, we added diagnostic items available to secondary care clinicians to the model (i.e., Subsections 1 plus 2 plus 3). Only items with a p-value ≤0.20 in the univariate analysis were considered relevant as predictors and were included in the respective models. In each scenario, the item with the highest p-value was manually removed from the model after which the logistic model was re-run (‘backward selection’). This step was repeated until only variables with p-values < 0.10 remained in the model for each scenario. Odds ratios for diagnosing asthma were calculated with COPD as reference group and vice versa. For each scenario a receiver operator characteristics (ROC) curve was created and the percentage explained variance (Nagelkerke R square) determined. Area under the curve (AUC) values from the ROC curves of the three scenarios were statistically compared using a non-parametric approach for correlated ROC curves32. SPSS statistics version 25.0 and SAS version 9.4 were used for the analyses. Missing data were not imputed. Two-sided p-values < 0.05 were considered statistically significant, except for the testing of the AUC values between Scenarios 1 and 2 and Scenarios 2 and 3, respectively, in which multiple testing was taken into account by using p < 0.025 to define statistical significance (i.e., Bonferroni correction: p = 0.05/2 = 0.025).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Results
Study population
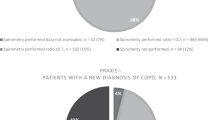
In the total sample of 532 study subjects (all Caucasians), 84 (16%) were diagnosed with asthma, 138 (26%) were diagnosed with COPD, and in 310 subjects (58%) no chronic airways disease was diagnosed (Table 1). Compared to patients with COPD the patients diagnosed with asthma were significantly younger (mean age 50.2 (SD 11.4) versus 57.8 (SD 10.0); p < 0.001) and more likely to be female (59.5% versus 44.2%; p = 0.027). There was no statistically significant difference in BMI between the two diagnostic subgroups (p = 0.22).
Differences and similarities in clinical features and lung function
Table 1 gives an overview of the differences and similarities in demographic characteristics, clinical features and lung function values between patients with asthma and patients with COPD. Patients diagnosed with COPD were significantly more likely to be former or current smokers and had more packyears compared to patients with asthma (21.3 (SD 19.5) versus 9.1 (14.4); p < 0.001). Patients with asthma were significantly more likely to have allergies compared to patients with COPD (p < 0.001) but there was no difference in the prevalence of eczema between the subgroups (p = 0.99). Patients with asthma had significantly more often symptoms of wheezing (p = 0.006) compared to patients with COPD. The prevalence of having chronic cough, phlegm or breathlessness was not significantly different between the groups. Patients with COPD had significantly lower % predicted postbronchodilator FEV1 values (88.2% versus 98.9%; p < 0.001) compared to patients with asthma. There were no differences in the presence of reversibility (p = 0.75) or bronchial hyperresponsiveness (p = 0.68) between the two subgroups. No additional diagnostic tests were requested by the two chest physicians.
Differentiating ability of diagnostic items
Demographic characteristics, clinical features and lung function tests yielded a total of 21 diagnostic variables (Table 1). Excluding items with p-values of >0.20 in the univariate analysis resulted in twelve items that were considered as relevant discriminants to be entered in the multivariable logistic models: age, gender, packyears, wheeze, phlegm, breathlessness, allergy, respiratory symptoms triggered by environmental exposures, postbronchodilator FEV1 % predicted, postbronchodilator FEV1/FVC < 0.70, RV/TLC and diffusion capacity.
Table 3 shows an overview of the differentiating ability of all relevant items. In Scenario 1 (only medical history questions), eight items were included in the model, four of which showed a statistically significant relationship when differentiating between asthma and COPD: packyears, wheeze, phlegm and allergy. In Scenario 2, ten items were included in the model, six of which showed a significant relation in differentiating between asthma and COPD: age, wheeze, breathlessness, allergy, FEV1 % predicted and FEV1/FVC. In Scenario 3, twelve items were included in the model, six showing statistical significance when differentiating between asthma and COPD: age, wheeze, breathlessness, allergy, FEV1 predicted and FEV1/FVC. Independent of the scenario, postbronchodilator FEV1/FVC was an important discriminant.
In Scenario 1 the logistic model showed a percentage explained variance of 41% and ROC characteristics showed an area under the curve (AUC) of 0.84 (95% confidence interval (CI): 0.78–0.89)) (Fig. 3). By adding diagnostic variables available to primary care (i.e., spirometry) in Scenario 2, the explained variance increased to 54% and AUC increased to 0.89 (95% CI 0.84–0.93). Finally, by adding more advanced diagnostic tests available to secondary care in Scenario 3, the explained variance increased to 56% but AUC remained 0.89 (95% CI 0.85–0.94). Statistical testing showed a statistically significant difference between the AUCs of Scenarios 2 and 1 (p = 0.020) but no such difference between the AUCs of Scenarios 3 and 2 (p = 0.967, see Table 3).
*Area under the curve (AUC) values and p-values of comparison between Scenario 1 and 2 and Scenario 2 and 3: see Table 3.
Discussion
In this study, we looked at which patient characteristics distinguish between patients diagnosed with asthma or COPD, and established the added value of spirometry and of more advanced lung function measurements when differentiating between the two chronic airways diseases. Although asthma and COPD are both heterogenous conditions with multiple overlapping features, there are important clinical differences as well. We observed that in the scenario using only medical history questions, it is already possible to reliably distinguish between asthma and COPD. The most important factors to aid differentiation are smoking behaviour, certain respiratory symptoms and the presence of allergies. The use of postbronchodilator spirometry provided important additional discriminative power in correctly labelling a patient as having asthma or COPD. More advanced diagnostic tests that are mainly used in secondary care, such as measuring bronchial hyperresponsiveness and diffusion capacity, did not provide a better differentiation in this primary care study population.
In the present study, both bronchodilator reversibility and bronchial hyperresponsiveness had a similar prevalence in patients diagnosed with asthma and COPD. This finding is noteworthy, as the current GINA guideline refers to reversibility testing and bronchial hyperresponsiveness as criteria supporting the diagnosis of asthma1. However, our finding is not unique as previous studies have concluded that solely the presence of reversibility or bronchial hyperresponsiveness does not distinguish between the two obstructive airways diseases33,34,35,36. Besides these similarities, there were several clinical features that were statistically different between the two diagnostic subgroups and for that reason, these features can aid primary care clinicians when differentiating between asthma and COPD. Using only medical history questions in the logistic model (Scenario 1) already showed rather good differentiating ability (AUC = 0.84). These findings are in line with other studies that assessed the ability of solely using medical history questionnaires to distinguish between asthma and COPD. Beeh et al. concluded that with only medical history questions, it is possible to distinguish between asthma and COPD for the majority of patients with suspected or established obstructive lung disease37. Likewise, in their study Tinkelman et al. reported that a simple self-administered questionnaire can facilitate differentiation between obstructive lung diseases38. However, these studies did not look at the additional use of spirometry or more advanced diagnostic tests to discriminate between asthma and COPD nor did they quantify this in, for instance, an area under the curve analysis like we did. In the present study we found that postbronchodilator spirometry was important when differentiating the two conditions and together with medical history questions, the discriminating ability of the model improved (from AUC = 0.84 in Scenario 1 to AUC = 0.89 in Scenario 2). In contrast, more advanced diagnostic tests did not provide a better diagnostic differentiation (AUC remained 0.89 in Scenario 3). This does not mean that these tests are useless, as they have an important role in evaluating the presence and severity of structural lung damage (like, for instance, in emphysema and bronchiectasis) and in differentiating obstructive lung disease from other aetiologies in selected patients39,40.
A particular strength of our study is that we used standardized methods to conduct the lung function testing and to obtain the respiratory diagnoses. All questionnaires and lung function tests were standardized and prospectively collected, were supervised by certified lung function technicians, and the lung function tests met established quality standards.
Given the central role of general practice in the Dutch healthcare system, nearly all inhabitants are registered in a general practice of their own choice. Therefore, the subjects who participated in the initial DIMCA program and provided for the sample in the current analysis can be seen as representative for the adult Dutch population. On top of this, our study is original in categorizing diagnostic variables based on their availability in different healthcare settings.
However, there were limitations as well. We only looked at the diagnosis itself and did not consider the severity of the diagnosed chronic airways diseases. Because each subject was initially assessed by only one of the chest physicians we were not able to look at the interobserver agreement. Subjects who were considered to have no asthma or COPD were not mutually discussed by the chest physicians to reach a maximum substantiated outcome. However, given that the aim of our study was to differentiate between asthma and COPD and not to distinguish between being ‘respiratory healthy’ or not, we do not consider this to be a relevant limitation of the study.
In some cases the chest physicians’ assessment may have led to false positive diagnoses of COPD, as some subjects who had a post-BD FEV1/FVC value >0.70 (n = 3; see Table 2) or reported to never have smoked (n = 27) were assigned a COPD diagnosis nonetheless. Unfortunately, we cannot in retrospect ascertain the chest physicians’ specific considerations for assigning this diagnosis in these cases.
Whereas the data collection and diagnostic approach in the DIMCA study by Albers et al.22 were conducted in a prospective manner, our study was retrospective in design and we were limited to using a pre-existing list of diagnostic items. The data collection dates from more than a decade ago and therefore several more recent diagnostic tests were not included. For instance, several recent studies have shown that the underlying type of inflammation in patients with asthma and COPD is markedly different3,41. Tests like sputum cell count, peripheral eosinophil count, serum IgE and fractional exhaled nitric oxide (FeNO) provide relevant information about the underlying inflammatory process and could support differentiation between asthma and COPD, but were not assessed in our study. Besides their differentiating potential, these inflammatory markers could have taught us more about the pathogenesis of ‘Asthma-COPD Overlap’ (ACO), which is the subject of ongoing debate42,43. We call for researchers to perform a similar study as ours in a heterogenous sample of appropriate study subjects, with the addition of the aforementioned contemporary inflammatory markers to the study protocol.
By using the two distinct diagnoses (i.e., asthma and COPD) our study does not increase knowledge on how to identify patients with ACO. However, as the majority (i.e., two-thirds or more)6 of patients with chronic obstructive airways disease do not concern ACO, our observations do add insight into how to discriminate between these two diagnoses in a substantial part of the overall group of patients with chronic obstructive airways disease. In other words, the study does not ‘solve’ the wider problem of how to distinguish patients with ACO from those with an ‘unambiguous’ diagnosis of asthma or COPD, but it does contribute to the issue of how to diagnose and distinguish the patients in which there is no overlap.
Lastly, it is important to note that the subgroups of patients labelled with asthma or COPD are defined by the diagnostic criteria used by Albers et al.22. These criteria were based on GOLD and GINA guidelines from 2006 and 2007, respectively26,27. But despite new pathophysiological insights, the definition, description and diagnostic criteria of asthma and COPD have not substantially changed ever since1,2. The fact that we did not apply the current Global Lung Function Initiative (GLI) references values nor the lower limit of normal definition of airway obstruction44 will not have had a significant impact on our findings, as this mainly influences the interpretation of presence or absence of obstruction in elderly subjects45 who were hardly present in our middle-aged study sample. Thus, in our view using the older guideline-based classification does not render the results of the present study obsolete or invalid.
A final limitation of the study that should be mentioned is that younger adults (i.e., those aged 18–30) were not included in the study. However, as the aim of the study was to differentiate between asthma and COPD and a diagnosis of COPD below the age of 30 is highly unlikely, we do not think this has had a relevant impact on the findings as reported.
Besides the good discriminating ability of solely using anamnestic questions, our results emphasize the importance of postbronchodilator spirometry in distinguishing asthma from COPD and vice versa. However, it is important to realise that the lung function tests in our study were conducted by well-trained staff in a pulmonary function laboratory and interpreted by experienced chest physicians. To translate these results to the real-life setting, it requires standardized procedures, quality assurance and trained clinicians to interpret the spirometry data accurately and this may be difficult to achieve in primary care46,47,48. However, previous studies have shown that it is feasible to conduct reproducible and clinically meaningful spirometry tests in primary care and that primary care clinicians can interpret spirometry test results correctly29,49,50. Even while in our study bronchial hyperresponsiveness testing did not improve diagnostic differentiation, it has been shown that bronchial challenge testing is safe and feasible in a suitably equipped primary care diagnostic centre51. Referral to secondary care is indicated in the few cases in which it is not possible to establish a diagnosis on the basis of thorough medical history taking and well-conducted spirometry alone, or to exclude other possible underlying conditions.
In conclusion, primary care clinicians should be able to reliably differentiate between asthma and COPD with the combination of relevant patient history questions and postbronchodilator spirometry tests for the majority of patients with suspected chronic airways disease. More advanced diagnostic tests used in hospital care settings do not seem to provide a better overall diagnostic differentiation between asthma and COPD in primary care patients. Given the important additional role of postbronchodilator spirometry in this process of differentiating, the implementation of quality-assured spirometry testing and sufficient training should be mandatory in primary care practices. Furthermore, the availability of inflammatory markers in primary care could potentially provide better discriminating diagnostic ability but we did not investigate this in the current study.
Data availability
The data from the DIMCA study are not made publicly accessible because the variable names, labels and codebook are all in the Dutch language. The dataset can be requested from the corresponding author without restrictions, in which case relevant variables and labels will be translated to English.
Code availability
Readers can access the code (i.e., SPSS syntax) of the statistical analyses by sending a request to the corresponding author.
References
Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention. www.ginasthmaorg (2021).
Global Initiative for Chronic Obstructive Lung Disease (GOLD). The Global Strategy for the Diagnosis, Management and Prevention of COPD. www.goldcopdorg (2021).
Barnes, P. J. Immunology of asthma and chronic obstructive pulmonary disease. Nat. Rev. Immunol. 8, 183–192 (2008).
Doeing, D. C. & Solway, J. Airway smooth muscle in the pathophysiology and treatment of asthma. J. Appl Physiol. 114, 834–843 (2013).
Gibson, P. G. & Simpson, J. L. The overlap syndrome of asthma and COPD: what are its features and how important is it? Thorax 64, 728–735 (2009).
Leung, J. M. & Sin, D. D. Asthma-COPD overlap syndrome: pathogenesis, clinical features, and therapeutic targets. BMJ 358, j3772 (2017).
Carolan, B. J. & Sutherland, E. R. Clinical phenotypes of chronic obstructive pulmonary disease and asthma: recent advances. J. Allergy Clin. Immunol. 131, 627–634.quiz 635 (2013).
Tinkelman, D. G., Price, D. B., Nordyke, R. J. & Halbert, R. J. Misdiagnosis of COPD and asthma in primary care patients 40 years of age and over. J. Asthma 43, 75–80 (2006).
Miravitlles, M. et al. Difficulties in differential diagnosis of COPD and asthma in primary care. Br. J. Gen. Pr. 62, e68–e75 (2012).
Jones, R. C., Dickson-Spillmann, M., Mather, M. J., Marks, D. & Shackell, B. S. Accuracy of diagnostic registers and management of chronic obstructive pulmonary disease: the Devon primary care audit. Respir. Res. 9, 62 (2008).
Nelson, H. S., Weiss, S. T., Bleecker, E. R., Yancey, S. W. & Dorinsky, P. M. The Salmeterol Multicenter Asthma Research Trial: a comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterol. Chest 129, 15–26 (2006).
McMahon, A. W., Levenson, M. S., McEvoy, B. W., Mosholder, A. D. & Murphy, D. Age and risks of FDA-approved long-acting β2-adrenergic receptor agonists. Pediatrics 128, e1147–e1154 (2011).
Rodrigo, G. J. & Castro-Rodríguez, J. A. Safety of long-acting β agonists for the treatment of asthma: clearing the air. Thorax 67, 342–349 (2012).
Ernst, P., Gonzalez, A. V., Brassard, P. & Suissa, S. Inhaled corticosteroid use in chronic obstructive pulmonary disease and the risk of hospitalization for pneumonia. Am. J. Respir. Crit. Care Med. 176, 162–166 (2007).
Crim, C. et al. Pneumonia risk in COPD patients receiving inhaled corticosteroids alone or in combination: TORCH study results. Eur. Respir. J. 34, 641–647 (2009).
Chalitsios, C. V., Shaw, D. E. & McKeever, T. M. Corticosteroids and bone health in people with asthma: a systematic review and meta-analysis. Respir. Med. 181, 106374 (2021).
Yang, I. A., Clarke, M. S., Sim, E. H. & Fong, K. M. Inhaled corticosteroids for stable chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. 7, CD002991 (2012).
Derom, E. et al. Primary care spirometry. Eur. Respir. J. 31, 197–203 (2008).
Casas Herrera, A. et al. COPD Underdiagnosis and misdiagnosis in a high-risk primary care population in four Latin American countries. A key to enhance disease diagnosis: The PUMA Study. PLoS ONE 11, e0152266 (2016).
Yu, W. C. et al. Spirometry is underused in the diagnosis and monitoring of patients with chronic obstructive pulmonary disease (COPD). Int. J. Chron. Obstruct. Pulmon Dis. 8, 389–395 (2013).
van den Boom, G. et al. Active detection of chronic obstructive pulmonary disease and asthma in the general population. Results and economic consequences of the DIMCA program. Am. J. Respir. Crit. Care Med. 158, 1730–1738 (1998).
Albers, M. et al. Do family physicians’ records fit guideline diagnosed COPD? Fam. Pr. 26, 81–87 (2009).
Sterk, P. J. et al. Standardized challenge testing with pharmacological, physical and sensitizing stimuli in adults. Eur. Respir. J. 6(Suppl 16), 53–83 (1993).
American Thoracic Society. Standardization of Spirometry, 1994 Update. Am. J. Respir. Crit. Care Med. 152, 1107–1136 (1995).
Quanjer, P. H. et al. Lung volumes and forced ventilatory flows. Report Working Party Standardization of Lung Function Tests, European Community for Steel and Coal. Official Statement of the European Respiratory Society. Eur. Respir. J. Suppl. 16, 5–40 (1993).
Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention. www.ginasthmaorg. (2007).
Global Initiative for Chronic Obstructive Lung Disease (GOLD). The Global Strategy for the Diagnosis, Management and Prevention of COPD. www.goldcopdorg. (2006).
Quanjer, P. H. et al. Multi-ethnic reference values for spirometry for the 3-95 year age range: the global lung function 2012 equations. Eur. Respir. J. 40, 1324–1343 (2012).
Yawn, B. P. et al. Spirometry can be done in family physicians’ offices and alters clinical decisions in management of asthma and COPD. Chest 132, 1162–1168 (2007).
The Dutch College of General Practitioners (NHG) guideline Adult asthma, fifth version [NHG-Standaard Astma bij volwassenen, versie 5.0]. https://richtlijnen.nhg.org/ (2020).
The Dutch College of General Practitioners (NHG) guideline COPD, fifth version [NHG Standaard COPD, versie 5.0]. https://richtlijnen.nhg.org/ (2021).
DeLong, E. R., DeLong, D. M. & Clarke-Pearson, D. L. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44, 837–845 (1988).
Tashkin, D. P. et al. Bronchodilator responsiveness in patients with COPD. Eur. Respir. J. 31, 742–750 (2008).
Tashkin, D. P. et al. A 4-year trial of tiotropium in chronic obstructive pulmonary disease. N. Engl. J. Med. 359, 1543–1554 (2008).
Grootendorst, D. C. & Rabe, K. F. Mechanisms of bronchial hyperreactivity in asthma and chronic obstructive pulmonary disease. Proc. Am. Thorac. Soc. 1, 77–87 (2004).
van den Berge, M. et al. Clinical and inflammatory determinants of bronchial hyperresponsiveness in COPD. Eur. Respir. J. 40, 1098–1105 (2012).
Beeh, K. M., Kornmann, O., Beier, J., Ksoll, M. & Buhl, R. Clinical application of a simple questionnaire for the differentiation of asthma and chronic obstructive pulmonary disease. Respir. Med. 98, 591–597 (2004).
Tinkelman, D. G. et al. Symptom-based questionnaire for differentiating COPD and asthma. Respiration 73, 296–305 (2006).
Hegewald, M. J. Diffusing capacity. Clin. Rev. Allergy Immunol. 37, 159–166 (2009).
Lutfi, M. F. The physiological basis and clinical significance of lung volume measurements. Multidiscip. Respir. Med. 12, 1–12 (2017).
Mauad, T. & Dolhnikoff, M. Pathologic similarities and differences between asthma and chronic obstructive pulmonary disease. Curr. Opin. Pulm. Med. 14, 31–38 (2008).
Orie, N. G. M. & Sluiter, H. J. Bronchitis: an international symposium. (Royal van Gorcum, Assen, Netherlands, 1962).
Postma, D. S., Weiss, S. T., van den Berge, M., Kerstjens, H. A. & Koppelman, G. H. Revisiting the Dutch hypothesis. J. Allergy Clin. Immunol. 136, 521–529 (2015).
Hall, G. L. & Stanojevic, S. Executive GLIN, Members of the GLINE. The Global Lung Function Initiative (GLI) Network ERS Clinical Research Collaboration: how international collaboration can shape clinical practice. Eur. Respir. J. 53, 1–4 (2019).
Schermer, T. R. et al. Current clinical guideline definitions of airflow obstruction and COPD overdiagnosis in primary care. Eur. Respir. J. 32, 945–952 (2008).
Enright, P. L. Should we keep pushing for a spirometer in every doctor’s office? Respir. Care 57, 146–51. (2012).
Johns, D. P., Burton, D., Walters, J. A. & Wood-Baker, R. National survey of spirometer ownership and usage in general practice in Australia. Respirology 11, 292–298 (2006).
Eaton, T. et al. Spirometry in primary care practice: the importance of quality assurance and the impact of spirometry workshops. Chest 116, 416–423 (1999).
Schermer, T. R. et al. Validity of spirometric testing in a general practice population of patients with chronic obstructive pulmonary disease (COPD). Thorax 58, 861–866 (2003).
Ruppel, G. L., Carlin, B. W., Hart, M. & Doherty, D. E. Office spirometry in primary care for the diagnosis and management of COPD: national lung health education program update. Respir. Care 63, 242–252. (2018).
Bins, J. E., Metting, E. I., Muilwijk-Kroes, J. B., Kocks, J. W. H. & In ‘t Veen, J. The use of a direct bronchial challenge test in primary care to diagnose asthma. NPJ Prim. Care Respir. Med. 30, 45 (2020).
Acknowledgements
The authors appreciate the statistical advice provided and analysis performed by Reinier Akkermans of the Department of Primary and Community of the Radboud University Medical Center regarding the comparison of the ROC curves for the three Scenarios. The study was funded by the Radboud University Medical Center, Nijmegen, The Netherlands.
Author information
Authors and Affiliations
Contributions
J.D.M.B. and T.R.S. initiated the study, performed the data analysis, and wrote the initial draft version of the paper. E.W.M.A.B. and J.C.C.M.V. critically reviewed the paper. All authors approved the final version of the paper that was submitted.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bouwens, J.D.M., Bischoff, E.W.M.A., in ’t Veen, J.C.C.M. et al. Diagnostic differentiation between asthma and COPD in primary care using lung function testing. npj Prim. Care Respir. Med. 32, 32 (2022). https://doi.org/10.1038/s41533-022-00298-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41533-022-00298-4
This article is cited by
-
Chronic comorbid conditions and asthma exacerbation occurrence in a general population sample
npj Primary Care Respiratory Medicine (2023)