Abstract
While the efficacy of deep brain stimulation (DBS) is well-established in Parkinson’s Disease (PD), the benefit of DBS varies across patients. Using imaging features for outcome prediction offers potential in improving effectiveness, whereas the value of presurgical brain morphometry, derived from the routinely used imaging modality in surgical planning, remains under-explored. This review provides a comprehensive investigation of links between DBS outcomes and brain morphometry features in PD. We systematically searched PubMed and Embase databases and retrieved 793 articles, of which 25 met inclusion criteria and were reviewed in detail. A majority of studies (24/25), including 1253 of 1316 patients, focused on the outcome of DBS targeting the subthalamic nucleus (STN), while five studies included 57 patients receiving globus pallidus internus (GPi) DBS. Accumulated evidence showed that the atrophy of motor cortex and thalamus were associated with poor motor improvement, other structures such as the lateral-occipital cortex and anterior cingulate were also reported to correlated with motor outcome. Regarding non-motor outcomes, decreased volume of the hippocampus was reported to correlate with poor cognitive outcomes. Structures such as the thalamus, nucleus accumbens, and nucleus of basalis of Meynert were also reported to correlate with cognitive functions. Caudal middle frontal cortex was reported to have an impact on postsurgical psychiatric changes. Collectively, the findings of this review emphasize the utility of brain morphometry in outcome prediction of DBS for PD. Future efforts are needed to validate the findings and demonstrate the feasibility of brain morphometry in larger cohorts.
Similar content being viewed by others
Introduction
Deep brain stimulation (DBS) is a well-established treatment for advanced Parkinson’s Disease (PD)1. Despite the generally promising outcomes, the outcome of DBS varies across patients. Considering the pivotal importance of refining patient selection process and improving surgical benefits, predicting the outcome and complication of the DBS surgery in PD is necessary2. Progress in methodology has provided new insights into outcome prediction. Advanced imaging technologies such as brain tractography and functional MRI have been applied in the outcome prediction of DBS. These techniques share the advantages of testing a wide range of neural properties. However, they also require expertize in acquiring high-quality raw data and in deciphering the results, which may limit their utility in clinical practice3.
As a standard procedure used for presurgical planning4, structural MRI based on T1-weighted or T2-weighted sequences has its advantage in clinical practice owing to the relatively short scanning time and popularization across surgical centers5. Several imaging biomarkers have been proposed to predict the progression of PD including the morphometry of substantia nigra6. Post-processing methods, such as voxel-based morphometry (VBM) are widely applied, which help support disease diagnosis, track clinical progressions, and monitor treatment effects7,8,9.
However, recent findings on the T1-weighted imaging associated with DBS benefits revealed more discrepancies than it solved. Analyses of the motor cortex showed promising results for the prediction of motor outcomes10,11, whereas some study negated the associations12. Volumes of brain ventricles were suggested in some studies to predict postsurgical outcomes13,14,15,16, while other studies did not corroborate these associations17,18. Also, the value of these biomarkers in the prediction of DBS outcomes has not been fully investigated. It, therefore, remains to be clarified how and where presurgical MRI findings on brain morphometry can help in predicting clinical response to DBS in PD patients. This review systematically analyzes existing evidence on the association of DBS outcome and MRI findings on brain morphometry. Interpretations of associations between brain structures and surgery outcomes are discussed. Limitations in current studies are also pointed out to assist future efforts in the search for reliable imaging predictors.
Results
Literature search
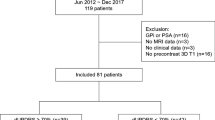
After the removal of duplicates, 793 publications were scanned for title and abstract and a total of 25 studies were included in this review after full-text review (Fig. 1): 13 studies analyzed the motor outcome of DBS including two studies focusing on the axial symptoms (Table 1, Supplementary Table 1); and 13 studies investigated non-motor outcomes, including cognitive impairment (eight studies) and psychiatric changes (six studies) (Fig. 2).
Predictors of motor outcome
The motor symptoms are mainly measured by UPDRS III or MDS-UPDRS III scales. Four studies measured the outcome by percent improvement by comparing UPDRS III total score at presurgical medication-off state (UPDRSIII med off) with postsurgical medication-off and stimulation-on state (UPDRSIII med off, stim on)10,19,20,21. Among them, one study also measured percent improvement in UPDRSIII at medication-on state (UPDRSIII med on/ med on, stim on)19. Two studies used the absolute change of UPDRSIII med off/ med off, stim on13,17. Two studies used the ratio of UPDRSIII med off/ med off, stim on11,14. Two study using the absolute postsurgical UPDRSIII med off, stim on score21,22. One study used percent improvement in postoperative UPDRSIII (UPDRSIII med off, stim off / med off, stim on)23. One study regressed postoperative scores on variables adjusting for presurgical scores12.
Cardinal motor symptoms
Cortical thickness
Four studies included in our review analyzed the associations of cortical thickness with motor outcomes (Table 1)10,11,12,13. All the studies used freesurfer to conduct the analysis and the analysis methods were further summarized in supplementary table 1. Positive correlations between cortical thickness in the motor cortex and motor outcome were found in two studies (Fig. 3). Muthuraman et al. found the cortical thickness of the frontal lobe (paracentral area and superior frontal region) predicted the UPDRSIII med off, stim on/ UPDRSIII med off and an improved postoperative outcome at low stimulation voltages. In addition, precuneus, superiortemporal, inferiorparietal, and superiorparietal areas in the left hemisphere also predicted better postoperative outcomes at low stimulation voltages11. Chen et al. found a positive association between percent improvement in MDS-UPDRSIII med-off/med-off, stim-on and thickness of the right precentral cortex10. Frizon et al. found that cortical thickness of the left lateral occipital region was strongly related to the improvement of postsurgical MDS-UPDRS III med off/med off, stim on, as confirmed in both region of interest (ROI) analysis and voxel-wised analysis12. Additionally, moderate negative correlations were also observed between the MDS-UPDRS III score and the right lateral occipital area, pars opercularis, posterior cingulate, the superior-temporal area in the right hemisphere as well as the lateral orbitofrontal, transverse temporal, and insula regions in the left hemisphere in ROI analysis, which was not confirmed in voxel-wised analysis12.
Brain volumetry
Ten studies investigating the relationship between volumetric changes and motor improvement were identified13,14,17,19,20,21,22,23,24,25. One study utilized 2D measurements including the bicaudate ratio (ratio between the inter-caudate and skull distance)26, the Evans index (width of the anterior ventricular horn divided by biparietal cranium diameter)27, and the third ventricular width (largest width of the third ventricle at the level of the posterior commissure)14 to assess subcortical changes14. Other studies used automated or semi-automated volumetry to analyze the alterations of the whole brain or ROIs.
Consistent with the findings of cortical thickness, the volume of the frontoparietal cortex assessed by VBM was found associated with DBS response. Jergas et al. found that volumes of the frontoparietal cortical regions were significantly associated with the percent improvement in UPDRSIII med off/med off, stim on20. The gray matter loss was identified in clusters in the bilateral medial prefrontal cortex, paracingulate and cingulate gyrus, and a large cluster in the left parietal lobe and the angular gyrus in patients with less improvement in UPDRSIII. In addition, smaller clusters of atrophy were found in the right superior frontal gyrus, reaching the supplementary motor area, as well as in the left middle frontal gyrus and the left precuneus in patients with the less motor improvement20.
Morphometry of thalamus was also found associated with motor outcomes. Yim et al. showed that patients with larger improvement had increased volumes of the right thalamus, anterior cingulate and the left anterior middle frontal cortex23. The results of the thalamus were partially replicated by Younce et al. who found that smaller thalamic volumes and larger ventricular (the lateral and 3rd ventricles) volumes (which were significantly inter-correlated) predicted less absolute improvement in UPDRS III med off/med off, stim on after STN-DBS13.
While Younce et al. demonstrated the predictive capability of the ventricle volume (containing the lateral and 3rd ventricles) in motor outcome after bilateral STN-DBS13, Price et al. evaluating patients receiving unilateral STN-/GPi-DBS showed that ventricle volume (as measured by the lateral ventricle) and the measurements of contralateral/ipsilateral volumes to the side of symptom onset or DBS lead placement did not predict absolute improvement in UPDRSIII med off/med off, stim on or axial motor changes measured by UPDRS subscores17. Also, patients with higher motor improvements did not have smaller ventricles relative to those with less motor improvement17. Another study used the bicaudate ratio, the Evans index, and the third ventricular width to measure subcortical volumes. The indices were able to predict postsurgical UPDRSIIImed off, stim on /UPDRSIII med off for patients receiving bilateral pallidal DBS14. The Evans index and third ventricular width also correlated with presurgical medication response.
Axial symptoms
Two studies focused on the axial symptoms of PD patients24,25. Karachi et al. found that PD patients with developed or aggravated freezing of gait (FOG) within 1 year (as measured by the subitem of UPDRS II) showed significantly reduced bilateral gray matter density in the sensorimotor and associative putamen, and an increased gray matter density in the left inferior frontal and right inferior temporal gyri24. PD patients with falls after surgery had significantly reduced gray matter density in the left postcentral gyrus and an increase in the culmen of the right cerebellar lobe relative to non-falling PD patients. Statistical maps of stimulation showed that the best effects of STN-DBS on FOG and falls were associated with the location of contacts within the STN, but no specific locations were related to aggravation24. Additionally, the atrophy of basalis of Meynert (NBM) showed a trend for predicting the degree of increase in swing time variability after 3 years of continuous STN-DBS25.
Predictors of non-motor outcome
13 studies included in our review evaluated the relationship between brain morphometry and non-motor symptoms after DBS, with nine studies investigating cognitive abilities and five studies focusing on psychiatric complications (Table 2).
Cognitive decline
Different outcome measures of cognitive decline including clinical scales (Mini Mental Status Examination, MMSE; Mattis Dementia Rating Scale, MDRS), indexes of specific cognitive domains as well as diagnostics criteria (the DSM-IV dementia criteria, the Movement Disorders Society Task Force Criteria level 2)28,29 were adopted in studies evaluating cognitive functions. The follow-up duration varied from 6 months to over 2 years. Different covariates were considered in the studies and were collected in Supplementary Table 2.
Brain volumetry
Eight studies that were included in our review reported associations of brain volumetry with the cognitive outcome15,22,28,29,30,31,32,33. ROI analysis assessing specific structures was adopted in these studies. (Fig. 2).
A case report from Puy et al. highlighted the association of hippocampus volume with dementia after DBS31. Replicated results were found in the prediction of cognitive decline by reduced hippocampal volumes28,30. Compared with the age and gender-matched control group who underwent STN-DBS but did not develop dementia after surgery, patients who developed dementia had significantly smaller presurgical hippocampal volume. Logistic regressions showed that hippocampal volume was an independent predictor of dementia. Hippocampal volumes were significantly associated with postoperative MMSE in the whole population but were not related to memory scores or executive function scores28.
In the evaluation of specific verbal memory functions of PD patients, Geevarghese et al. found that hippocampal (left and right) and thalamic volumes (left) were significant predictors of changes in List Learning score after STN-DBS. Additionally, patients with a more severe decline in the Delayed Story Recall test had significantly smaller thalamic and hippocampal volumes compared to those in the stable group30.
Apart from the hippocampus and thalamus, atrophy of the nucleus accumbens was also suggested to correlate with cognitive decline after DBS15. The atrophy of left nucleus accumbens predicted postoperative decline in executive functions of patients after STN-DBS in 1 year15. Presurgical left nucleus accumbens volume was strongly correlated with the variation in the MDRS initiation/perseveration subscore. The volume of the presurgical left nucleus accumbens also correlated with the right and left ventricle volumes. Additionally, correlations were found between the variation of total MDRS and the left lateral ventricle volume, between the variation of the initiation/perseveration subscore and the left lateral ventricle volume, between the variation of the backward digit-span task and the right and left superior frontal gyrus thickness, after adjustments for age, gender, disease severity, decrease in L-DOPA equivalent dose following surgery and “anterior” contact location. No significant association between contact location and cognitive decline was found in the study.
The volume of NBM was correlated with the relative change of MMSE or DemTect scores of PD patients 1 year after STN-DBS33. The results were demonstrated in both the regression model and the machine learning model, where NBM volume was the most predictive variable. The cognitive outcome was significantly influenced by the NBM/total intracranial volume ratio, but not by the number of cognitive domains affected presurgically nor UPDRS III med off. Nevertheless, only a small amount of variability could be predicted by NBM size (R2 of 0.149 in a multiple linear regression model), which needs further validation from larger cohorts.
Psychiatric changes
Of the six studies that evaluated psychiatric changes, five studies focused on immediate psychiatric alterations16,18,34,35,36, and only one study evaluated post-operative clinical scales including neuropsychiatric assessments after 6 months22. Most studies evaluated postoperative confusion, delirium, or impulsivity, whereas long-term mood disturbances such as apathy and depression lacked attention.
Brain atrophy was recognized as an independent risk factor for post-operative psychiatric changes16,35,36. Patients who developed neuropsychiatric complications (7/22) had significantly smaller gray matter thickness and white matter volume when compared to patients without acute postoperative neuropsychiatric complications36. Subsequent analysis showed that, patients with acute neuropsychiatric changes had significantly smaller white matter volumes in the left caudal middle frontal gyrus, left lingual gyrus, left pericalcarine gyrus, and left precuneus, as well as significantly reduced cortical thickness bilaterally in the frontal lobe (caudal middle frontal and precentral gyruses), temporal lobe (inferior and middle temporal gyruses) and parietal lobe (postcentral, superior parietal and supramarginal gyruses). Significant cortical atrophy was found on the left-brain hemisphere in caudal middle frontal and inferior temporal areas36. Wang et al. found that presurgical brain atrophy as visually inspected was an independent factor that influenced the occurrence of postoperative delirium (POD)35. Bourne et al. showed that patients with post-operative confusion after DBS for the treatment of various diseases including PD had a significantly larger minimum width of the lateral ventricles than that of the controls16. Additionally, greater maximum and minimum width of the lateral ventricles were also associated with a higher occurrence of other complications. However, the correlation demonstrated between changes in ventricle morphometry and psychiatric complications was not consistent18. Increased inter-mammillary distance (>8 mm) was found in five PD patients older than 60 years old, among whom the incidence of early postoperative mental changes reached 60%18.
At the basal ganglia level, Lu et al. found that the left STN volume differed significantly between PD patients and healthy control subjects and was negatively correlated with postsurgical Hamilton Anxiety Scale and Hamilton Depression Scale (HAMD) score. The right STN volume was negatively correlated with the post-operative HAMD score. The lateralized difference of STN may be explained by the fact that 46/59 patients in the study had disease onset in the right limb, and most patients exhibited more severe right limb symptoms22.
Discussion
This systematic review investigated the associations between brain morphometric features and clinical outcomes of PD patients following DBS treatment: A total of 25 articles with 1316 patients containing structural MRI data were identified and reviewed. Replicated results showed that reduced brain volume/cortical thickness in the motor cortex and the thalamus were associated with less improvement in cardinal motor symptoms, while other structures including anterior cingulate and brain ventricles were also suggested to hold contributory roles. Regarding non-motor outcomes, reduced volume of the hippocampus was repeatedly reported to associate with poor cognitive performance after DBS, while other structures including the nucleus accumbens, thalamus, and ventricles were also reported. NBM was suggested to predict cognitive performance and axial symptoms after DBS surgery. Different anatomical features are responsible for specific or joint pathophysiological functions related to clinical manifestations.
The findings of brain morphometry related to DBS response of cardinal motor symptoms mainly resided in sensorimotor cortex, thalamus and brain ventricles. The sensorimotor cortex constitutes a critical component of the hyperdirect pathway. Antidromic modulations from targets of DBS of the pathway were evidenced by both human and animal studies37. Connectivity analysis showed that structural and functional connectivity to this region was associated with the clinical improvement of DBS38,39,40,41,42. The reduced cortical thickness and diminished volume of this region were also reported to correlate with motor outcomes in our review10,11. However, the finding was not replicated except for two analyses which found a non-significant correlation between the volume of sensorimotor regions and motor response13,23. The sensitivity of the method may be a reason. Regional ROI analysis can fail to detect changes in small clusters as determined by voxel-wised analysis. The study conducted by Chen et al.10 used the same clinical scale (MDS-UPDRS) and had a similar disease duration with that of Frizon’s study12, while the number of subjects enrolled was twice as many12 (73 vs 36), of which the analysis should therefore be more sensitive.
Ventroanterior and ventrolateral motor thalamic areas are critically positioned in the pallidal and cerebellar thalamocortical pathways43,44. The reduced thalamic volume was associated with poor DBS outcome, which was alerted in three of our included studies13,23,30. Thalamus comprises multiple subnuclei with different connections and functions45. Morphological changes in thalamic subnuclei were associated with clinical symptoms of PD individuals and have been proposed to predict clinical improvement46. Nevertheless, none of the existing studies have investigated the associations of thalamic subregions with DBS effects. Structural assessments of thalamus subnuclei can be achieved by conventional structural MRI, the value of which is yet to be explored.
Ventricle volume is regarded as a good indicator of brain atrophy13,14. It allows the estimation of disease progression and is correlated with atrophy of other critical brain areas47,48. Additionally, the enlargement of ventricles was associated with operative problems such as cerebrospinal fluid leakage49,50,51 and was considered the most reliable predictor for brain shift52. Four studies included in our review supported the role of ventricle volume in the prediction of DBS outcomes13,14,15,16 whereas two studies did not support its value17,18. Several differences should be noted in the studies. First, the calculation of ventricle volumes was handled by different methods (linear measurements or automated segmentation algorithms). Second, the definition of brain ventricles differed. Younce et al. evaluated ventricle volumes containing the lateral and third ventricles13 whereas Price et al. investigated only the lateral ventricles17, which yielded contradictory results. Third, different study designs may hamper cross-comparisons. Differences in Younce et al.’s and Price et al.’s studies were also observed in surgery selections (bilateral STN DBS vs unilateral GPi/STN DBS), patient cohorts (n = 86, age mean = 62.9, UPDRSIII 36 ± 10 vs n = 37, age mean = 58.8, UPDRSIII 44 ± 11) and follow-up durations (15 vs 4 months), which should all be noted while interpreting the results.
Aside from the above regions, other indicators that are suggested to predict cardinal motor improvement of DBS include (1) Lateral-occipital cortex, which contains the extrastriate body area responsible for the visual identification of body parts and movements12. Functional imaging analysis showed that the area may play a compensatory role in PD for impaired motor functions performed by the premotor cortex53, while reduced cortical thickness may disrupt the compensatory activity. (2) Anterior cingulate gyrus, an important cortical component of the motor circuit responsible for various movement disorders23,54. Probabilistic tractography studies showed that the cingulate gyrus was composed of the anatomical network with the frontal and prefrontal cortex, whose dysfunction may alter the connectivity between the STN and frontal lobe55.
Axial symptoms are largely resistant to dopaminergic therapy and respond variably to DBS treatment. Some symptoms such as gait impairment and FOG seem to be partially improved by DBS, whereas others such as postural instability and related falls might worsen after surgery56. Growing evidence suggests the degeneration of cholinergic neurons, a pathological hallmark of progressive cognitive deterioration, is crucial in the pathophysiology of gait and balance disorders57. Studies involved in our systematic review suggested the role of NBM25 and several other cortical and subcortical regions24 in associations with gait impairment and falls after DBS surgery. Patients with NBM degeneration showed a significant cognitive decline, which was also associated with higher axial scores, highlighting the relationship between axial symptoms and cognitive functions58. The findings shed light on the understanding of gait and balance control in PD patients.
Although DBS does not seem to affect the overall cognitive profile in PD patients, it is associated with focal cognitive deficits59. Apart from the limbic and cortical spread of Lewy body pathology, several factors contribute to the cognitive deterioration of PD patients including neuronal loss, neurotransmitter deficits, and cerebral network disruptions60. These can be partially reflected by imaging parameters, which provides potential biomarkers for the prediction of non-motor symptoms in supplement to neuropsychological evaluations. Hippocampus is a major structure responsible for complex episodic memory processes, the atrophy of which is an established biomarker of degenerative processes such as AD61. The involvement of HA in the progression of Lewy bodies contributes significantly to the development of dementia in PD, which is demonstrated in imaging, proteomics and biochemical analysis62,63,64. Four studies identified in our review noted the associations of reduced presurgical hippocampal volume with verbal memory decline or overall cognitive impairment after DBS, suggesting hippocampal atrophy as a preexisting risk factor for cognitive decline for patients receiving DBS surgery28,30,31,32. Connections between hippocampal subfields with cognitive functions and motor subtypes of PD patients have been proposed, the study of which might exert increased potential in patient selection with candidate stratified to different motor subtypes65,66. Other brain structures were also reported to correlate with the cognitive outcome of DBS, including thalamus30, nucleus accumbens15 and NBM33. These structures provide important crossroads for cognition-relevant information processing. As the progress of cognitive deterioration is increasingly impacted by cholinergic pathology, shrinkage of the central node in the cortical cholinergic system may disturb cognitive functions, which may be due to the imbalance of neurotransmitter systems67,68.
Treatment effects of DBS are curtailed by psychiatric side effects, which include transient confusion in the brief postoperative period, and depression, apathy, and hypomania that may occur in a longer follow up69. Psychiatric disorders are frequently observed in post-DBS treatment where post-operative delirium (POD) in the immediate duration after STN-DBS surgery is seen in 22% of patients70 and symptoms such as depression and anxiety are observed in up to 50% of patients71. Most of the studies included in our review focused on brief mental alterations. These studies found that brain atrophy, as visually inspected35 or measured by the width of lateral ventricles16, white matter volume34 and frontal cortical regions36 were associated with immediate psychiatric symptoms. Caudal middle frontal area was suggested to correlate with POD after DBS36. The area has been linked with impulse control disorders in PD and brief psychotic disorder72,73. Atrophy of the region may impair both short intralobar connections and top-down inputs to other brain structures, which is considered important pathophysiology of major psychiatric disorders74,75. STN was found to associate with depression after 6 months of DBS22. The anterior limbic part of STN has numerous connections with limbic circuits according to the classic tripartite STN organization model, although recent ultrahigh-resolution MRI studies suggest cortical inputs of STN are overlapping76,77. Mechanical STN lesioning can cause limbic circuitry imbalance exhibited by depression-like behavior in rats78,79. The atrophy of STN may indicate the impairment of the limbic circuitry, while its predictive function remains to be determined.
Several limitations apply to this study. First, 7/25 studies included in this review did not consider the influence of confounding variables (age, disease duration, cognitive status, presurgical levodopa usage, contact locations, DBS procedure and so on) apart from intracranial volume. Especially, only five studies targeting STN considered post-operative lead position in their analysis. The results suggested that the contact locations and the consequent overlap between the motor STN and the individual patient’s VTA were associated with the outcome. It is still worthy of disentangling the variances, either with a multivariate model or building a matched control group, in outcome explained by presurgical morphometry from the ones explained by postsurgical lead locations in future cohorts. Second, only one study with 34 patients in our review analyzed GPi-DBS related brain morphometry separately from STN-DBS14. So far, the morphometric findings are largely inconsistent between studies on different targets or even between those on a single target, which is more likely to be attributed to the limited sample size and varied analytic methodologies than the target itself. In the future, the findings are yet to be validated with a larger sample size of patients with both GPi- and STN- DBS. Third, as described previously, the definition of motor improvement varied among studies. Comparisons of motor scales before and after surgery may reflect the overall benefit of DBS surgery while comparing postsurgical motor scales between stimulation-on and stimulation-off states can minimize the microlesion effect and better represent the short-term stimulation effect.
This study summarizes the state-of-the-art evidences in the association between structural MRI based on T1-weighted sequence and clinical outcome of DBS in PD. Reduced cortical thickness/brain volume of motor cortex and thalamus were associated with less motor improvement. Reduced volume of the hippocampus was associated with impaired cognitive performance after DBS. Other indicators such as the cortical thickness of the lateral-occipital cortex, volumes of the anterior cingulate gyrus, brain ventricles, nucleus accumbens, STN and NBM have also shown predictive utility for postsurgical outcomes. The findings of this systematic review facilitate the construction of applicable prognostic models to assist in the major issues of patient selection and promote the understanding of the DBS mechanism. Due to the relatively small number of subjects and generally inconsistent results, the findings are yet to be validated with a larger sample size and appropriate analytic procedure.
Methods
Search strategy
This review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. We searched the literature in the online databases of PubMed and Embase from inception to April 5, 2022. The MESH terms (PubMed), EMTREE terms (Embase), and keywords we used were as follows: Parkinson’s disease AND stimulation AND (gray matter OR thickness OR volume OR atrophy OR morphometry OR morphology OR structural covariance). Additionally, we scanned the reference lists of included studies and relevant reviews for additional relevant cited articles.
Eligibility criteria
The inclusion criteria were: (1) written in English; (2) full-text articles; (3) included participants with PD; (4) examined the outcome of DBS; (5) contained presurgical morphometric features; (6) examined the relationship between the outcome of DBS and presurgical morphometric features. Studies were excluded if they (1) were conference abstracts, correspondence, editorials, meta-analysis, systematic reviews, reviews, or chapters; (2) were not in English; (3) were animal or other in vitro studies; (4) were simulation studies; (5) did not assess the outcome of deep brain stimulation; (6) did not assess presurgical brain morphometric features; (7) were not relevant as considered by the authors. DBS procedures were summarized in supplementary table 3.
Selection and data extraction process
The following information was extracted: year of publication, sample size, study type, participant demographics including age and sex, disease duration, pre- and post-surgical Unified Parkinson’s Disease Rating Scale (UPDRS) III or Movement Disorder Society (MDS)-UPDRS III scores, morphology indices (brain volume or cortical thickness) and covariates used in the analysis. Two authors (Wang and Lai) screened the abstracts and two authors (Wang and Li) collected the data independently.
Data availability
No datasets were generated or analyzed during the current study.
Code availability
No codes were generated or analyzed during the current study.
References
Hartmann, C. J., Fliegen, S., Groiss, S. J., Wojtecki, L. & Schnitzler, A. An update on best practice of deep brain stimulation in Parkinson’s disease. Ther. Adv. Neurol. Disord. 12, 1756286419838096 (2019).
Limousin, P. & Foltynie, T. Long-term outcomes of deep brain stimulation in Parkinson disease. Nat. Rev. Neurol. 15, 234–242 (2019).
Wong, J. K. et al. A comprehensive review of brain connectomics and imaging to improve deep brain stimulation outcomes. Mov. Disord. 35, 741–751 (2020).
Silberstein, P. et al. Deep brain stimulation for Parkinson’s disease: Australian referral guidelines. J. Clin. Neurosci. 16, 1001–1008 (2009).
Artusi, C. A., Lopiano, L. & Morgante, F. Deep brain stimulation selection criteria for Parkinson’s disease: time to go beyond CAPSIT-PD. J. Clin. Med. 9, 3931 (2020).
Pyatigorskaya, N., Gallea, C., Garcia-Lorenzo, D., Vidailhet, M. & Lehericy, S. A review of the use of magnetic resonance imaging in Parkinson’s disease. Ther. Adv. Neurol. Disord. 7, 206–220 (2014).
Giorgio, A. & De Stefano, N. Clinical use of brain volumetry. J. Magn. Reson Imaging 37, 1–14 (2013).
Chaudhary, S. et al. Cortical thickness and gyrification index measuring cognition in Parkinson’s disease. Int J. Neurosci. 131, 984–993 (2021).
Boutzoukas, E. M. et al. Cortical thickness in childhood left focal epilepsy: thinning beyond the seizure focus. Epilepsy Behav. 102, 106825 (2020).
Chen, Y. et al. Predict initial subthalamic nucleus stimulation outcome in Parkinson’s disease with brain morphology. CNS Neurosci. Ther. (2022).
Muthuraman, M. et al. Effects of DBS in parkinsonian patients depend on the structural integrity of frontal cortex. Sci. Rep. 7, 43571 (2017).
Frizon, L. A. et al. Cortical thickness in visuo-motor areas is related to motor outcomes after STN DBS for Parkinson’s disease. Parkinsonism Relat. Disord. 71, 17–22 (2020).
Younce, J. R., Campbell, M. C., Perlmutter, J. S. & Norris, S. A. Thalamic and ventricular volumes predict motor response to deep brain stimulation for Parkinson’s disease. Parkinsonism Relat. Disord. 61, 64–69 (2019).
Hamed, M. A., Ponce, F. A., Lambert, M. & Moguel-Cobos, G. Subcortical atrophy and motor outcomes in pallidal deep brain stimulation for parkinson disease. World Neurosurg. 142, e89–e94 (2020).
Planche, V. et al. Anatomical predictors of cognitive decline after subthalamic stimulation in Parkinson’s disease. Brain Struct. Funct. 223, 3063–3072 (2018).
Bourne, S. K., Conrad, A., Konrad, P. E., Neimat, J. S. & Davis, T. L. Ventricular width and complicated recovery following deep brain stimulation surgery. Stereotact. Funct. Neurosurg. 90, 167–172 (2012).
Price, C. C. et al. Lateral ventricle volume is poor predictor of post unilateral DBS motor change for Parkinson’s disease. Parkinsonism Relat. Disord. 17, 343–347 (2011).
Hrabovsky, D. et al. Factors responsible for early postoperative mental alterations after bilateral implantation of subthalamic electrodes. Br. J. Neurosurg. 31, 212–216 (2017).
Hamasaki, T., Yamada, K., Hirai, T. & Kuratsu, J. A positive correlation between fractional white matter volume and the response of Parkinson disease patients to subthalamic stimulation. Acta Neurochir. (Wien.) 152, 997–1006 (2010).
Jergas, H. et al. Brain morphometry associated with response to levodopa and deep brain stimulation in Parkinson disease. Neuromodulation https://doi.org/10.1016/j.neurom.2022.01 (2022).
Bonneville, F. et al. Parkinson disease, brain volumes, and subthalamic nucleus stimulation. Neurology 64, 1598–1604 (2005).
Lu, L. et al. Measuring subthalamic nucleus volume of Parkinson’s patients and evaluating its relationship with clinical scales at pre- and postdeep brain stimulation treatment: a magnetic resonance imaging study. Biomed. Res. Int. 2021, 6646416 (2021).
Yim, Y. et al. Pretreatment brain volumes can affect the effectiveness of deep brain stimulation in Parkinson’s disease patients. Sci. Rep. 10, 22065 (2020).
Karachi, C. et al. Clinical and anatomical predictors for freezing of gait and falls after subthalamic deep brain stimulation in Parkinson’s disease patients. Parkinsonism Relat. Disord. 62, 91–97 (2019).
Wilkins, K. B., Parker, J. E. & Bronte-Stewart, H. M. Gait variability is linked to the atrophy of the Nucleus Basalis of Meynert and is resistant to STN DBS in Parkinson’s disease. Neurobiol. Dis. 146, 105134 (2020).
Aylward, E. H., Schwartz, J., Machlin, S. & Pearlson, G. Bicaudate ratio as a measure of caudate volume on MR images. AJNR Am. J. Neuroradiol. 12, 1217–1222 (1991).
Ambarki, K. et al. Brain ventricular size in healthy elderly: comparison between Evans index and volume measurement. Neurosurgery 67, 94–99 (2010).
Aybek, S. et al. Hippocampal atrophy predicts conversion to dementia after STN-DBS in Parkinson’s disease. Parkinsonism Relat. Disord. 15, 521–524 (2009).
Blume, J. et al. The impact of white matter lesions on the cognitive outcome of subthalamic nucleus deep brain stimulation in Parkinson’s disease. Clin. Neurol. Neurosurg. 159, 87–92 (2017).
Geevarghese, R. et al. Verbal memory decline following DBS for Parkinson’s disease: structural volumetric MRI relationships. PLoS One 11, e0160583 (2016).
Puy, L. et al. Acute dementia after deep brain stimulation in parkinson disease. World Neurosurg. 119, 63–65 (2018).
Weinkle, L. J. et al. Association of MRI measurements with cognitive outcomes after STN-DBS in Parkinson’s disease. Mov. Disord. Clin. Pr. 5, 417–426 (2018).
Kübler, D. et al. Nucleus basalis of Meynert predicts cognition after deep brain stimulation in Parkinson’s disease. Parkinsonism Relat. Disord. 94, 89–95 (2022).
Tanaka, M. et al. Risk factors for postoperative delirium after deep brain stimulation surgery for Parkinson disease. World Neurosurg. 114, e518–e523 (2018).
Wang, X. Q. et al. Nomogram for predicting postoperative delirium after deep brain stimulation surgery for Parkinson’s disease. World Neurosurg. 130, e551–e557 (2019).
Radziunas, A. et al. Neuropsychiatric complications and neuroimaging characteristics after deep brain stimulation surgery for Parkinson’s disease. Brain Imaging Behav. 14, 62–71 (2020).
Li, Q., Qian, Z. M., Arbuthnott, G. W., Ke, Y. & Yung, W. H. Cortical effects of deep brain stimulation: implications for pathogenesis and treatment of Parkinson disease. JAMA Neurol. 71, 100–103 (2014).
Lai, Y. et al. Pallidal stimulation as treatment for camptocormia in Parkinson’s disease. NPJ Parkinsons Dis. 7, 8 (2021).
Lai, Y. et al. Subthalamic atimulation for camptocormia in Parkinson’s disease: association of volume of tissue activated and structural connectivity with clinical effectiveness. J. Parkinsons Dis. 11, 199–210 (2021).
Lai, Y. et al. Value of functional connectivity in outcome prediction for pallidal stimulation in Parkinson disease. J. Neurosurg. https://doi.org/10.3171/2022.3.JNS212732 (2022).
Horn, A. et al. Connectivity predicts deep brain stimulation outcome in Parkinson disease. Ann. Neurol. 82, 67–78 (2017).
Chen, Y. et al. Seed-based connectivity prediction of initial outcome of subthalamic nuclei deep brain stimulation. Neurotherapeutics 19, 608–615 (2022).
Tucker, H. R. et al. Deep brain stimulation of the ventroanterior and ventrolateral thalamus improves motor function in a rat model of Parkinson’s disease. Exp. Neurol. 317, 155–167 (2019).
Tucker, H. R. et al. Motor thalamic deep brain stimulation alters cortical activity and shows therapeutic utility for treatment of Parkinson’s disease symptoms in a rat model. Neuroscience 460, 88–106 (2021).
Boelens Keun, J. T. et al. Structural assessment of thalamus morphology in brain disorders: a review and recommendation of thalamic nucleus segmentation and shape analysis. Neurosci. Biobehav Rev. 131, 466–478 (2021).
Chen, Y. et al. The morphology of thalamic subnuclei in Parkinson’s disease and the effects of machine learning on disease diagnosis and clinical evaluation. J. Neurol. Sci. 411, 116721 (2020).
Mak, E. et al. Longitudinal whole-brain atrophy and ventricular enlargement in nondemented Parkinson’s disease. Neurobiol. Aging 55, 78–90 (2017).
Camicioli, R. et al. Ventricular dilatation and brain atrophy in patients with Parkinson’s disease with incipient dementia. Mov. Disord. 26, 1443–1450 (2011).
Martinez-Ramirez, D. et al. Atrophy and other potential factors affecting long term deep brain stimulation response: a case series. PLoS One 9, e111561 (2014).
Machado, A. et al. Deep brain stimulation for Parkinson’s disease: surgical technique and perioperative management. Mov. Disord. 21, S247–258 (2006). Suppl 14.
Piacentino, M. et al. Pneumocephalus in subthalamic deep brain stimulation for Parkinson’s disease: a comparison of two different surgical techniques considering factors conditioning brain shift and target precision. Acta Neurochir. (Wien.) 163, 169–175 (2021).
Obuchi, T. et al. Direction and predictive factors for the shift of brain structure during deep brain stimulation electrode implantation for advanced Parkinson’s disease. Neuromodulation 11, 302–310 (2008).
van Nuenen, B. F. et al. Compensatory activity in the extrastriate body area of Parkinson’s disease patients. J. Neurosci. 32, 9546–9553 (2012).
DeLong, M. R. & Wichmann, T. Basal ganglia circuits as targets for neuromodulation in Parkinson disease. JAMA Neurol. 72, 1354–1360 (2015).
Koirala, N. et al. Frontal lobe connectivity and network community characteristics are associated with the outcome of subthalamic nucleus deep brain stimulation in patients with Parkinson’s disease. Brain Topogr. 31, 311–321 (2018).
Fasano, A., Aquino, C. C., Krauss, J. K., Honey, C. R. & Bloem, B. R. Axial disability and deep brain stimulation in patients with Parkinson disease. Nat. Rev. Neurol. 11, 98–110 (2015).
Karachi, C. et al. Cholinergic mesencephalic neurons are involved in gait and postural disorders in Parkinson disease. J. Clin. Invest 120, 2745–2754 (2010).
Schulz, J., Pagano, G., Fernández Bonfante, J. A., Wilson, H. & Politis, M. Nucleus basalis of Meynert degeneration precedes and predicts cognitive impairment in Parkinson’s disease. Brain 141, 1501–1516 (2018).
Maheshwary, A., Mohite, D., Omole, J. A., Bhatti, K. S. & Khan, S. Is deep brain stimulation associated with detrimental effects on cognitive functions in patients of Parkinson’s disease? A systematic review. Cureus 12, e9688 (2020).
Aarsland, D. et al. Cognitive decline in Parkinson disease. Nat. Rev. Neurol. 13, 217–231 (2017).
Barnes, J., Ourselin, S. & Fox, N. C. Clinical application of measurement of hippocampal atrophy in degenerative dementias. Hippocampus 19, 510–516 (2009).
Delgado-Alvarado, M., Gago, B., Navalpotro-Gomez, I., Jiménez-Urbieta, H. & Rodriguez-Oroz, M. C. Biomarkers for dementia and mild cognitive impairment in Parkinson’s disease. Mov. Disord. 31, 861–881 (2016).
Villar-Conde, S. et al. The human hippocampus in Parkinson’s disease: an integrative stereological and proteomic study. J. Parkinsons Dis. 11, 1345–1365 (2021).
Das, T., Hwang, J. J. & Poston, K. L. Episodic recognition memory and the hippocampus in Parkinson’s disease: a review. Cortex 113, 191–209 (2019).
Luo, C. et al. Distinct hippocampal subfield atrophy in Parkinson’s disease regarding motor subtypes. Parkinsonism Relat. Disord. 93, 66–70 (2021).
Becker, S. et al. Association of hippocampal subfields, CSF biomarkers, and cognition in patients with Parkinson disease without dementia. Neurology 96, e904–e915 (2021).
Halliday, G. M., Leverenz, J. B., Schneider, J. S. & Adler, C. H. The neurobiological basis of cognitive impairment in Parkinson’s disease. Mov. Disord. 29, 634–650 (2014).
Wilson, H., de Natale, E. R. & Politis, M. Nucleus basalis of Meynert degeneration predicts cognitive impairment in Parkinson’s disease. Handb. Clin. Neurol. 179, 189–205 (2021).
Mosley, P. E. & Akram, H. Neuropsychiatric effects of subthalamic deep brain stimulation. Handb. Clin. Neurol. 180, 417–431 (2021).
Carlson, J. D. et al. Postoperative delirium in Parkinson’s disease patients following deep brain stimulation surgery. J. Clin. Neurosci. 21, 1192–1195 (2014).
Thobois, S. et al. Non-motor dopamine withdrawal syndrome after surgery for Parkinson’s disease: predictors and underlying mesolimbic denervation. Brain 133, 1111–1127 (2010).
Li, H. & Kéri, S. Regional brain volumes in brief psychotic disorder. J. Neural Transm. (Vienna) 127, 371–378 (2020).
Biundo, R. et al. Patterns of cortical thickness associated with impulse control disorders in Parkinson’s disease. Mov. Disord. 30, 688–695 (2015).
Catani, M. et al. Short frontal lobe connections of the human brain. Cortex 48, 273–291 (2012).
Rajasethupathy, P. et al. Projections from neocortex mediate top-down control of memory retrieval. Nature 526, 653–659 (2015).
Temel, Y., Blokland, A., Steinbusch, H. W. & Visser-Vandewalle, V. The functional role of the subthalamic nucleus in cognitive and limbic circuits. Prog. Neurobiol. 76, 393–413 (2005).
Emmi, A., Antonini, A., Macchi, V., Porzionato, A. & De Caro, R. Anatomy and connectivity of the subthalamic nucleus in humans and non-human primates. Front Neuroanat. 14, 13 (2020).
Klein, J. et al. Lesion of the medial prefrontal cortex and the subthalamic nucleus selectively affect depression-like behavior in rats. Behav. Brain Res. 213, 73–81 (2010).
Widge, A. S. et al. Psychosis from subthalamic nucleus deep brain stimulator lesion effect. Surg. Neurol. Int. 4, 7 (2013).
Acknowledgements
This study was supported by Shanghai Clinical Research Center for Mental Health (grant nos. 19MC1911100) and SJTU Trans-med Awards Research (grant nos. 2019015) to Dr. Sun, the National Natural Science Foundation of China (grant nos. 81771482 to B.S. and 82101546 to Y.L.) and Shanghai Sailing Program (grant no. 21YF1426700 to Y.L.). The authors have no personal, financial, or institutional interest in any of the drugs, materials, or devices described in this article.
Author information
Authors and Affiliations
Contributions
Conception and design: B.S., Y.L. Acquisition of data: F.W., Y.L., Y.P., H.L. Analysis and interpretation of data: F.W., Y.L., H.L., Q.L. Drafting the article: F.W., Y.L., Y.P. Critically revising the article: all authors. Reviewed submitted version of manuscript: all authors. Approved the final version of the manuscript on behalf of all authors: B.S. Administrative/technical/material support: B.S., Q.L., Y.L., F.W. Study supervision: B.S., Y.P., Y.L. F.W., Y.L. and Y.P. contributed equally as first authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, F., Lai, Y., Pan, Y. et al. A systematic review of brain morphometry related to deep brain stimulation outcome in Parkinson’s disease. npj Parkinsons Dis. 8, 130 (2022). https://doi.org/10.1038/s41531-022-00403-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41531-022-00403-x