Abstract
Petroleum-hydrocarbons spilt in surface seawater may pose potential threats to the corrosion of steel infrastructures. We show that crude oil accelerated steel corrosion mainly by accelerating microbiologically influenced corrosion (MIC). Crude oil led to the dominance of marine oil-degraders including Alcanivorax and Marinobacter in both seawater and steel rust, rather than sulfate-reducing bacteria (SRB) which dominated the rust microbial community in the no-oil group. Crude oil not only enhanced microbial oxygen respiration and aerobic hydrocarbon degradation but also nitrate reduction and anaerobic hydrocarbon degradation process in steel rust, indicating more heterogeneous microenvironments formed on steel surfaces. Furthermore, the low abundance of SRB and dissimilatory sulfate reduction gene (dsr), and the existence of iron-carbonate and iron-sulfate minerals implied that microbial sulfide, previously regarded as the main cause of MIC, was not the major contributor to steel corrosion in early petroleum-polluted seawater. Marine specialized oil-degraders seem to play more significant roles under such conditions.
Similar content being viewed by others
Introduction
Corrosion of iron-based materials in oil-containing environments such as oil and gas fields is of great concern to the reliability of industrial infrastructures worldwide. In China alone, the total direct corrosion cost in the oil and gas industry has been estimated to be as high as 2.82% of the total production value in 20141. In marine environments, the corrosion of metallic materials exposed to oil-containing and sulfate-rich seawater during the oil production and transportation process has attracted great attention because of the deleterious reservoir souring and material perforation, which are largely accounted for the activities of complex microbial communities2,3. Such deleterious effects on materials influenced by microorganisms directly or indirectly, depending on the specific reactions between microorganisms (sulfate-reducing bacteria (SRB), acid-producing bacteria (APB), etc.)/materials (metal, concrete, etc.)/media (chemical composition and physical parameters such as nutrients, sulfate, and sulfides), are termed as microbiologically influenced corrosion (MIC)4,5.
Recently, attention has focused on the MIC process under oil-containing conditions where anaerobic conditions will eventually develop in the marine environment, such as oil transportation pipelines, oil storage equipment and seawater-compensated fuel ballast systems6,7. Under such conditions, the anaerobic biodegradation of hydrocarbons such as petroleum-derived fuels and alternative biofuels can be accomplished independently by some specialized hydrocarbon-degraders or syntrophically by various functional microbes. SRB are the key players involved in hydrocarbon-degradation and corrosion-causing processes8. For instance, Desulfoglaeba alkanexedensa, a sulfate-reducing marine bacterium9, can completely oxidize alkanes independently by using sulfate as the terminal electron acceptor and produce sulfide and low-molecular organic acids that generally accelerate steel corrosion10,11,12. Thus, microbial sulfate reduction performed by SRB, which can be accelerated by anaerobic hydrocarbon degradation, are frequently regarded as the main cause of MIC in these oil-containing environments6,10,11. However, some researchers think that the role of SRB is overstated13 and other functional microbes such as APB is the major causative factor14.
For steel infrastructures, another corroding condition in marine oil-seawater environments that should not be overlooked is the coastal surface seawater polluted by petroleum-hydrocarbons, where hydrocarbon degradation is mainly performed aerobically15. Petroleum hydrocarbons are ubiquitous in the oceans, where natural seepage and human activities including discharges of tanker ballast water and off-shore petroleum platform leakages16, are estimated to release between 0.4 and 4.0 million tons of crude oil into the ocean ecosystems annually17. A large amount of petroleum-hydrocarbons was observed in the surface seawater and could result in extensive effects on the coastal ecosystems18,19. Typically, the structure of indigenous microbial communities could be shaped by oil which provides additional nutrients for microorganisms and contributes to the enrichment of specific oil-degraders20,21,22. A variety of marine steel infrastructures, especially coastal bridges and ships in oil wharves, platforms and transportation pipelines of offshore oil-gas exploration, are exposed to the oil-contaminated seawaters and provide habitats for biofilm-forming microbes23,24. Once exposed to crude oil, both the attached and planktonic microbial communities would be shifted21,22,24,25 and thus exert influence on the MIC processes. The change of microbial composition and MIC process caused by oil contimanation under such aerobic conditions may be different from that in anaerobic environments26,27. Despite the possibility, MIC of steel in these surface marine waters has been largely neglected, leaving open questions of how the planktonic and attached microorganisms succeeded around/on the steel surface, and how they influence the MIC processes in oil-exposed surface seawaters.
Previous studies were mostly done using microcosms under strictly anaerobic or transient oxygen conditions10,11,23,24. In this study, we performed microcosm experiments with/without crude oil exposure under aerobic conditions open to the air, using natural surface seawater and bellowed sea mud collected from oil wharves as inocula. The key geochemical parameters of seawater, the corrosion parameters including corrosion rate and mineralogy of a commonly used steel in marine applications, and both the planktonic and attached microbial community were analyzed to determine the MIC mechanisms in oil-containing seawaters. We propose that increased acidic metabolic production and the more heterogeneous aerobic/anaerobic microenvironments formed on the steel surface, which are induced by marine oil-degraders Alcanivorax and Marinobacter, are the main contributing factors to the exacerbated steel corrosion. These findings offer insights into the understanding of the effects of hydrocarbon on MIC process in marine oil-polluted environments like oil wharves.
Results and discussion
In situ characterization of coastal seawater in the oil wharf
To confirm the existence of oil-polluted seawaters in natural marine environments, the hydrocarbon component of the sampled surface water in an oil wharf of Qingdao Port (China) was determined. The GC-MS spectrogram showed that some of the components of crude oil including n-alkanes and butyl-phenol were identified in field seawater (Supplementary Fig. 1A). Moreover, in situ analysis of bacterioplankton in the coastal seawater (Fig. 5b) showed that the relative abundance of marine oil-degraders especially Alcanivorax (3.2%) and Marinobacter (1.3%) in this oil wharf was higher than that (< 0.01%) in other sea areas we sampled28. Since oil-degrading bacteria usually grow and multiply rapidly in oil-impacted marine environments, they could be regarded as biomarkers of oil contamination22. These results revealed the existence of the oil-seawater corroding condition in natural marine environments which should not be overlooked.
Crude oil changed the geochemical factors of surface seawater
To determine whether crude oil changed geochemical factors of seawater, sulfate, pH, and low-molecular organic substances were tested as the major geochemical indicators (Fig. 1). Sulfate concentration decreased in both assays. But the consumption of sulfate was significantly higher (P < 0.05) at all the three time points in crude oil-exposed group (With Oil group) than that in no crude oil group (No Oil group) (Fig. 1A). It indicates that crude oil stimulated sulfate consumption. The strengthened sulfate consumption by crude oil was also observed in a previous study on carbon steel corrosion exposed to biofuels under anaerobic seawater conditions11.
Changes of A sulfate concentration in seawater and B pH value along with incubation time. C The concentration of low-molecular organic acids and alcohols after 85 days of incubation. D Cell number of acid-producing bacteria (APB) and sulfate-reducing bacteria (SRB) calculated using MPN enumeration methods. “*” indicated the significant differences between groups by t-tests; “▾” indicated the undetected substances. “With Oil”: With crude oil amendment; “No Oil”: Without crude oil. T1: 25 days; T2: 55 days; T3: 85 days. Error bars represent standard deviations of three independent samples from triplicate systems.
Although the pH value increased in both treatment groups, it was lower (P < 0.05) in With Oil groups than that in No Oil groups at day 25 (Fig. 1B). The lower pH may be due to the organic acids produced by planktonic hydrocarbon-degradation bacteria (Fig. 5). Then the pH value of With Oil groups increased sustainably and exceeded that of No Oil groups at the last time point of incubation (P < 0.05). At T3 time point, anaerobic environments were formed on the surface of steel. Under such anaerobic conditions, sulfate was used by increased SRB (compared with that at T2 time point, Fig. 5) as an electron acceptor for oxidation of organic carbon (petroleum-hydrocarbon, lactate, acetate, etc.) to produce H2S and HCO3− 29. The acidic H2S may precipitate as iron-sulfides in the steel surface, whereas the higher concentration of organic carbon in With Oil group than that in No Oil group at T3 time point stimulated more HCO3− produced and released into the seawater that contributed to the higher pH value in With Oil group. The same phenomenon of increased pH value after the addition of organic carbon was also observed in other systems30,31.
As products of microbial hydrocarbon degradation, low-molecular organic acids and alcohols were also examined after 85 days incubation (Fig. 1). Compared with that in With Oil group, most of the determined organic acids including lactate, butyrate, and alcohols including methanol, propanol and butanol, were lower or undetectable in No Oil group (P < 0.05). This suggests that the hydrocarbon degradation process was stimulated by crude oil. The alcohols and organic acids identified in the current study may be the intermediates of aerobic hydrocarbon degradation or the end products of anaerobic hydrocarbon degradation by marine oil-degrading bacteria. Aerobic degradation of oil usually occurs in the surface seawater and produces intermediate products including alcohols and organic acids32. For example, alkanes, as major components of crude oil (Supplementary Fig. 1B), are degraded firstly to primary alcohols and further oxidized to the corresponding aldehydes. These aldehydes are converted to fatty acids, which are processed by beta-oxidation and finally oxidized to CO233. Further, along with the formation of anoxic areas on the steel surface, anaerobic fermentation of crude oil would happen within these areas and produced various low-molecular-weight organic substances as the end-products, such as lactate and butyrate, which was observed in higher concentrations in our study (Fig. 1C). The formation of these acidic intermediates helped explain the accelerated corrosion rate of steel (Figs. 2 and 3).
The number of APB and SRB in seawater were detected by MPN enumeration methods (Fig. 1D). The number of APB increased from 10 cells mL−1 at the initial stage to 103 cells mL−1 at the last stage of incubation when exposed to crude oil. In contrast, it was undetectable in No Oil group. It indicates that crude oil enhanced the growth of APB. This result is consistent with that of the high concentration of organic acids (Fig. 1C). In contrast, the number of planktonic SRB was below detectable levels in both treatments. This is consistent with the results that very few sequenced 16S rRNA gene sequences were affiliated with SRB. The same finding was also shown in a previous study23. Such finding is not surprising, since the aerobic environment of seawater was not suitable for anaerobic SRB growth until oxygen was depleted in the enclosed systems (without exchange of gas)26. Sulfate consumption was stimulated by crude oil as noted above, but the abundance of major planktonic and attached SRB was not stimulated by crude oil at the same timepoint (Figs. 1D and 5B). This indicates that planktonic and attached SRB in seawater were not the major contributor to the strengthened sulfate consumption. To determine real contributors in oil-amendment simulators, the number of SRB in sediments was further examined. The result showed that the number of SRB in sediment was higher in With Oil group than that in No Oil group (3.15 × 102 vs. 2.74 × 102 cells mL−1) after 85 days of incubation. This clearly implies that increased SRB in sediments instead of that in seawater was one of the contributors to the observed higher sulfate consumption in seawater exposed to oil.
Crude oil increased the corrosion rate of steel by stimulating MIC
To determine the effect of crude oil on steel corrosion, we measured the corrosion rate by measuring the weight loss of steel after 85 days of incubation (Fig. 2). The steel exposed to seawater without crude oil underwent an average corrosion rate of 0.21 mm per year, while that exposed to seawater with crude oil showed an average corrosion rate of 0.29 mm per year. Thus the addition of crude oil significantly stimulated the steel corrosion (P < 0.05). Sterilized groups with/without crude oil were set to evaluate the effect of MIC on steel corrosion. The corrosion rate of both sterilized groups was 0.07 mm per year (P > 0.1). However, it was significantly lower than the corrosion rate of steel in unsterilized groups. Obviously, crude oil couldn’t accelerate steel corrosion unless microorganisms are involved. That is to say, crude oil enhanced steel corrosion mainly through stimulating the growth and activity of microorganisms.
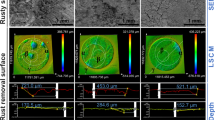
To further describe the characteristics of corrosion products, the surface morphology of corroded steel was analyzed by SEM and CLSM (Fig. 3). Clear differences were observed between No Oil group and With Oil group. At the T1 time point, pitting attack on the steel surface in groups was evident, but the maximum depth of the corrosion pits in No Oil group was 16.1 μm (average depth 10.6 μm), which was higher than that in With Oil group (maximum depth 8.8 μm, average depth 8.1 μm). The value of steel roughness in No Oil group (1.05 μm) was also higher than that in With Oil group (0.92 μm). The latter had more dense pits. At the T2 time point, the maximum depth of corrosion pits increased to 31.7 μm (average depth 27.4 μm) and the tiny pits developed into big holes (391.8 μm) in No Oil group, which were still higher than that in With Oil group (maximum depth 20.1 μm, average depth 17.6 μm, pore size 83.3 μm). The steel in No Oil group (4.12 μm) was rougher than that in With Oil group (1.72 μm). However, the corrosion seemed more severe in With Oil group than No Oil group at the end of the experiment (T3): the depth of the corrosion pits formed in With Oil group (maximum 54.2 μm, average depth 35.8 μm) exceeded that in No Oil group (maximum 49.4 μm, average depth 28.9 μm); the corrosion morphology of steel in With Oil group seemed to turn into more severe uniform corrosion. A similar phenomenon that steel surface showed the most evident localized corrosion was also found in seawater simulators after 14 weeks of exposure to oil34. Compared with the steel in microcosms under anaerobic conditions (> 1000 μm)11, the maximum pitting depth observed in these aerobic microcosms (< 100 μm) was much shallower. Collectively, these results suggested that crude oil enhanced the average corrosion rate, and the localized corrosion seemed to be inhibited at the initial exposed stage but stimulated at later exposed stage ultimately.
Crude oil impacted the composition of corrosion products
In order to analyze the potential effects of oil exposure on the formation of corrosion products, spectroscopic and microscopic techniques were used to observe the morphology and determine the composition of corrosion products formed at the end of incubation (Fig. 4). Rod and spiral-shaped microbes were observed to be associated with the corrosion products, and some were covered by corrosion products (white arrow in Fig. 4A, C) in simulators with and without crude oil. EDS analysis showed that compounds with high oxygen, iron, and sulfur content but with different shapes were observed around these microbes (Fig. 4B, D). This implied that microbes had a tight connection with the formation of corrosion products. Furthermore, manganese was only detected on steel surfaces in No Oil group (Fig. 4B, D), indicating manganese in steel was dissolved. This may be caused by manganese-oxidizing bacteria like Bacillus and Actinobacteria (data not shown) in the attached microbial communities. As manganese-oxidizing bacteria may cooperate with SRB to cause severe steel corrosion35, the higher abundance of SRB in No Oil group may indicate a stronger cooperation with manganese-oxidizing bacteria that accelerate the dissolution of manganese from the steel compared with that in With Oil group.
SEM images with high magnification view of corrosion products and microbes with (A) and without (C) crude oil. EDS spectra of typical corrosion products with (B) and without (D) crude oil. White rectangles represent the corresponding areas for EDS analysis. White arrows represent attached microbes. E XRD analysis of representative corrosion products. Indicative diffraction peaks for key Fe-corrosion phases were noted. “With Oil”: With crude oil amendment; “No Oil”: Without crude oil.
As shown in Fig. 4E, the corrosion products formed in No Oil group were mainly composed of mackinawite (FeS), ferrihydrite (FeO(OH)) iron oxide (Fe2O3, Fe3O4 and FeO), and iron hydroxide (Fe(OH)2). By comparison, the corrosion products in With Oil group were composed of iron carbonate (FeCO3 and Fe2O2CO3) and iron sulfate (Fe2(SO4)3 and FeSO3∙2.5 H2O), in addition to the same corrosion products like iron sulfide (FeS), iron oxide (Fe2O3 and Fe3O4) identified in No Oil group. The presence of iron sulfate might indicate the active activity of APB in the formation of corrosion products, as sulfate/sulfite might be actively produced by APB. Iron sulfate has also been previously observed as a major corrosion product in a naphtha-storage tank36. Moreover, as iron carbonate could be converted to FeS by sulfide37, the identification of iron carbonate in With Oil group rather than No Oil group, indirectly reflected the relatively low content of sulfide on the steel surface exposure to crude oil. Because the presence of iron sulfide could be regarded as an indicator of SRB-influenced corrosion4,38, the identification of iron carbonate in this study (Fig. 4E) might imply a relatively weak influence of SRB on steel corrosion when exposed to crude oil.
Crude oil changed the composition and diversity of planktonic and surface microbial community
α diversity
In total, ~1,440,567 effective tags of bacterial 16S rRNA genes with an average length of ~412 bp were obtained for subsequent analysis and 38,184 operational taxonomic units (OTUs) were acquired. To identify differences in α diversity, the species richness revealed by ACE and Chao1 index, the diversity revealed by Shannon and Simpson index among these four groups (planktonic microbial community without crude oil, planktonic microbial community with crude oil, surface microbial community without crude oil, surface microbial community with crude oil) in different stages were calculated (Supplementary Fig. 2). For planktonic microbial communities, With Oil group had a higher average value of ACE and Chao1 index, but a lower value of Shannon and Simpson index. Since Shannon and Simpson index are comprehensive diversity estimators giving both species richness and evenness, the contrast value of higher ACE/Chao1 but lower Shannon/Simpson could be explained by the lower species evenness in With Oil group. For the attached microbial community, the species richness of the With Oil group was higher (ACE, 2180; Chao1, 2040) in the initial stage, but it decreased to a lower level in the last stage (ACE, 2054; Chao1, 1959) than No Oil group (ACE, 2587; Chao1, 2488). The whole microbial diversity shown by Shannon and Simpson index had a similar trend. The phenomenon that oil exposure induced a reduction in bacterial diversity was in agreement with previous studies, in which hydrocarbon-degrading bacteria became the dominant responders to oil contamination in oil-contaminant environments such as mudflat beaches and soil39,40. In summary, the oil treatment reduced both the planktonic and attached bacterial diversity after 85-days simulation, which can be accounted for by the strong selection for specialist hydrocarbon-degrading bacteria22,39 such as Alcanivorax sp. and Marinobacter sp. (Fig. 5B) in the present study.
A Phylum (or class) level and B genus level composition analysis of top bacterial 16S rRNA gene sequences for seawater and surface samples. The cladogram of discriminatory taxa identified in the groups with/without crude oil from day 85 (T3) by LEfSe analysis (Linear Discriminant Analysis (LDA), log score > 3.5, P = 0.05). C Planktonic microbial communities from day 0 (T0) and day 85 (T3), and D the attached microbial communities from day 25 (T1), day 55 (T2), and day 85 (T3) were determined in laboratory microcosms with/without crude oil. The relative abundance of each taxonomy was the average value of that in triplicates. “With Oil”: With crude oil amendment; “No Oil”: Without crude oil.
Composition of planktonic microbial community
Regarding taxonomic assignment at the phylum (or class) level shown in Fig. 5A, Gammaproteobacteria (38.8%) and Alphaproteobacteria (36.0%) were the two predominant planktonic classes in the original seawater (T0). After immersion of X70 plates in seawater for 85 days (T3) without crude oil, the relative abundance of Gammaproteobacteria increased slightly from 38.8% to 43.0% and Alphaproteobacteria decreased from 36.0% to 22.2%. The relative abundance of Gammaproteobacteria in With Oil group increased significantly from 38.8% to 61.2% compared with that in No Oil group, while the contents of Alphaproteobacteria decreased obviously from 36.0% to 14.7%. Similar results were also observed at the genus level as shown in Fig. 5B. The original coastal seawater only contained a small proportion of hydrocarbon-degrading microbes. After being exposed to crude oil for 85 days, the relative abundance of oil-degrading microorganisms belonging to Gammaproteobacteria increased significantly, especially Alcanivorax from 3.2% to 27.2% and Marinobacter from 1.3% to 5.9%, respectively. Alcanivorax has a widespread occurrence across the ocean due to their ability of using a variety of saturated alkanes in crude oil (e.g., n-alkanes and iso-alkanes shown in Supplementary Fig. 1) more effectively than do other hydrocarbon-degrading bacteria. Thus, this genus usually acts as the early colonizer and become the predominant one after the amendment of crude oil in natural marine environments41. This phenomenon has been confirmed in this study (Fig. 5B). Marinobacter is also one of the known oil-degraders in the oil-polluted ocean and shows moderate amounts of oil degradation if compared to the high oil degradation potential of Alcanivorax18. The rapid response of marine hydrocarbon-degrading bacteria to oil implied that their role in steel corrosion should be taken into consideration given the growing oil-contamination cases in coastal sea areas.
LEfSe was further employed to identify specific planktonic taxa that were enriched in each group in the last culture stage (T3). The results are shown in Fig. 5C. The exposure to oil induced an increased abundance of the order Oceanospirillales (35%) in With Oil group (T3 with Oil). It belongs to Gammaproteobacteria and includes genera Alcanivorax (the above discussed), Marinobacterium, Oleibacter and Oleiphilus identified in the current study. Oceanospirillales members can move by chemotaxis degrade alkanes, enabling them to actively aggregate and increase in numbers rapidly under oil-polluted marine environments42. The genus Cycloclasticus (3%) was another specific planktonic taxa in With Oil group (Fig. 5B, C). Cycloclasticus has a significant role in the removal of aromatic hydrocarbons which were also major components of crude oil (Supplementary Fig. 1B). It usually appears in the later stage of oil-contaminated cases, at which time most saturated alkanes had been degraded and persistent aromatic hydrocarbon left. Cycloclasticus was only detected at a high concentration after 85 days (Fig. 5B), and also detected after a similar sampling period in some other oil-spill sea areas43.
Interestingly, the No Oil group (T3 without Oil) showed an increase of discriminatory taxa that have a high iron requirement for growth, such as Magnetovibrio and Nitrosomonas44,45. Magnetovibrio (6%) belongs to Alphaproteobacteria and is a type of typical magnetotactic genus with intracellular magnetosomes. Some members within Magnetovibrio exhibit chemolithoautotrophic growth on sulfide with oxygen as terminal electron acceptor and/or chemo-organoheterotrophic growth on various organic acids44. These groups of microorganisms were also detected in this study (Fig. 1C). Their motility by means of polar flagellum enables them to consume the carbon and energy substances in seawater initiatively.
Collectively, these results suggest that the addition of crude oil changed the planktonic microbial community around the steel and significantly stimulated the growth of oil-degrading marine microorganisms, while the only introduction of steel into seawater stimulated microbes that have a high requirement for iron.
Composition of surface microbial community
The dominant microbial groups of the microbial community attached to the steel were different from that of the planktonic one. Compared with the dominance of Gammaproteobacteria in planktonic microbial communities, Deltaproteobacteria had a higher abundance in surface microbial communities and even became the most abundant phylum in the surface microbial community in With Oil group (Fig. 5A). In contrast, the abundance of predominant Deltaproteobacteria decreased over time in No Oil group, while Deltaproteobacteria increased gradually in With Oil group. This observation was different from a previous study, which showed that the community shift from Gammaproteobacteria to Alphaproteobacteria was observed after 2 weeks of exposure to oil34. The discrepancy may be due to the presence of sediment as one of the sources of taxa in the present experimental design.
At the genus level, Desulfovibrio belonging to Deltaproteobacteria was detected as the most abundant genus in No Oil group, accounting for 65.8%, 34.7%, 25.9% at T1, T2, and T3, respectively (Fig. 5B). Desulfovibrio is one of the representative SRB usually observed as the predominant genus in the corrosion rust formed in the marine environments28,46. It has been proven to be the main cause of marine bacterial corrosion47,48. However, the amendment of crude oil into seawater changed the dominance of Desulfovibrio into Alcanivorax in the initial stages (T1 and T2 with Oil, Fig. 5). As iron is a significant component for Alcanivorax cells to synthesize a variety of oxygenases responsible for alkane activation and iron-containing heme to protect cells against oxidative stress, Alcanivorax cells possess a highly performing iron-uptake system consisting of siderophore and an extracellular type of iron-chelating molecule to provide necessary iron for the cells49,50. The capacity for iron-uptake and biofilm formation51, may explain why Alcanivorax dominated the attached bacterial communities. For the first time, this study revealed that the oil-degraders, Alcanivorax and Marinobacter, dominated the surface microbial community of steel and may greatly contribute to the corrosion process in the oil-polluted seawater.
LEfSe was used to identify the specific surface colonizers at the last incubation phases. As shown in Fig. 5D, the family Marinobacteraceae including genus Marinobacter and family Alcanivoracaceae including Alcanivorax were significantly enriched in With Oil group, while family Desulfurivibrionaceae including Desulfovibrio and family Flavobacteriaceae including Lutibacter were enriched in No Oil group. A previous study showed that members of Flavobacteriaceae displayed highly competitive Fe-uptake systems among marine prokaryotes52, which may explain its enrichment in steel surface microbial community herein.
Taken together, crude oil induced different but sustained impacts on the composition of surface microbial community compared to the planktonic microbial community. The added crude oil provided a major source of carbon and energy in an otherwise nutrient-starved seawater environment, which stimulated the growth of the oil-degrading microbes attached to the steel surface initially rather than SRB. Along with the degradation and consumption of crude oil, the abundance of oil-degraders began to decrease and SRB began to increase over time.
Crude oil changed the potential functions of planktonic and attached microbial community
To better identify the functions of the planktonic and attached microbial community under the exposure of crude oil, we analyzed the functional genes using PICRUSt prediction. The results are shown in Fig. 6. It was found that the surface microbial community had a different response to the crude oil amendment compared with the planktonic microbial community (Fig. 6A, B). For the planktonic microbial community (Fig. 6A), genetic information processing like ribosome biogenesis, chaperones and folding catalysis, protein folding and translation proteins were strongly stimulated by crude oil. Differently, amino acid metabolism (including arginine, proline, lysine and beta-alanine metabolism, valine, leucine, and isoleucine degradation), lipid metabolism (including fatty acid metabolism and biosynthesis of unsaturated fatty acids) and carbohydrate metabolism (including butanoate and propanoate metabolism) of the attached microbial community on the steel surfaces were stimulated by crude oil (Fig. 6B). Additionally, xenobiotics biodegradation and metabolism including degradation of benzoate, aminobenzoate, naphthalene, chloroalkane, and chloroalkene were promoted in the surface samples with crude oil addition. This indicated that crude oil had a certain effect on the functional pathways of both planktonic and attached microbial communities in a different manner.
A The functional pathways of planktonic microbial community and B attached microbial community that significantly enriched in groups with/without crude oil for 85 days (T3). The left column showed the relative abundance of each pathway and the right column showed the difference between groups. C The abundance of important functional genes of the planktonic and attached microbial communities from day 0 (T0), day 25 (T1), day 55 (T2), and day 85 (T3). Significant differences (P < 0.05) were analyzed by the T-test; “▾” and “*” indicated the significantly different genes between groups with/without crude oil from planktonic and attached samples for 85 days (T3), respectively. “With Oil”: With crude oil amendment; “No Oil”: Without crude oil.
Crude oil stimulated energy metabolism of both planktonic and attached metabolism. Thus, the key functional genes related to terminal electron acceptation and hydrocarbon degradation were further identified. As shown in Fig. 6C, it was observed that the genes related to oxygen respiration (cox and cco), denitrification (nar and nap), iron (mtr), and sulfate reduction (dsr) were identified in both seawater and surface samples. But only cox and nar genes were significantly enriched (P < 0.05) in surface samples exposed to crude oil. In contrast, the cox gene in seawater was significantly reduced (P < 0.05) because of oil amendment. This observation indicated that crude oil stimulated the microbial oxygen respiration and denitrification process of the attached microbial community, but weakened the oxygen respiration of planktonic microbial communities. The enhancement of both aerobic and anaerobic respiration modes on steel surface may be because of the formation of heterogeneous aerobic and anaerobic microenvironments within the thickening and complicated corrosion products, where both aerobic and anaerobic areas existed. The microbial denitrification process could happen under such anaerobic conditions, which was also previously observed in anoxic zones with oil in marine environments53,54. The dominant attached microbes, such as Alcanivorax sp. and Marinobacter sp. with the ability of anaerobic growth via denitrification process55,56, were most likely responsible for the denitrification process of the attached microbial community.
Not surprisingly, the key gene dsr related to dissimilatory sulfate-reduction process was not significantly stimulated in With Oil group compared to that No Oil group (P > 0.5) (Fig. 6C), which was consistent with the taxonomic analysis (Fig. 5). In contrast, key genes like cys involved in assimilatory sulfate-reduction were significantly enriched in With Oil group (P < 0.5). As more sulfate was consumed in With Oil group than that in No Oil group, we proposed that dissimilatory sulfate-reduction by SRB was not the major cause of sulfate consumption. Microbial assimilatory sulfate-reduction may greatly promote sulfate consumption in sediments.
Genes related to aerobic hydrocarbon degradation for both planktonic and attached microbial communities were enhanced by crude oil, especially genes related to alkane (alk), alcohol (adh), and fatty acid (fad) degradation (P < 0.5) (Fig. 6C). Genes related to aldehyde degradation (aldh) were only significantly enriched in steel surfaces (P < 0.5), suggesting the potential formation of fatty acid. Although at a low abundance, genes related to anaerobic hydrocarbon degradation were also observed (Fig. 6C). This is also expected because anaerobic degradation of hydrocarbons is usually considered to be several orders of magnitude slower than aerobic degradation of hydrocarbons57. Despite of the low abundance, the benzoate-relating gene bcr, which is important for the anaerobic degradation of aromatic compounds58, was significantly facilitated by crude oil on the steel surface (P < 0.5). The anaerobic fermentation of hydrocarbon usually produces a range of low-molecular-weight organic acid, which was observed at higher concentrations in surrounding seawater with crude oil than that in control groups (Fig. 1C). This implies that anaerobic fermentation also contributes to the accelerated steel corrosion.
Corrosion mechanisms under oil-amendment seawater conditions
Previous laboratory experiments performed with coastal seawater have shown that the amendment of hydrocarbon including both petroleum fuels24 and alternative fuels11,13 could accelerate steel corrosion. Higher sulfate consumption and sulfide production were observed along with the increased corrosion rate in those studies. Thus, the steel corrosion was believed to be the result of biogenic sulfide production stimulated by anaerobic or aerobic degradation of hydrocarbons11,23,24,26. In the present study, similar phenomenon of higher sulfate consumption and more severe corrosion were also observed in microcosms with crude oil amendment seawater than that without crude oil. However, the lower abundance of SRB and the key corresponding gene dsr in both attached and planktonic microbial communities exposed to the crude oil implied that the role of microbial sulfide production on steel corrosion was not as important as that in anaerobic environments. The higher sulfate consumption and weak microbial sulfate-reduction in seawater seem contrary. This can be explained by the aerobic environment during all the incubation stages and the usage of both seawater and sediment as inocula. Because the aerobic seawater was not suitable for the growth of SRB, the sediment in the simulators provided the anaerobic environments for SRB which induced the reduction of sulfate in seawater. The increased number of SRB in sediments in the crude oil exposure group confirmed this. The significantly stimulated gene cys by crude oil in seawater and surface, suggests that assimilatory sulfate reduction may also contribute to sulfate consumption in seawater. Sulfate consumption data are therefore not necessarily supportive of the assumption that microbial dissimilatory sulfate reduction was the major cause of steel MIC corrosion under crude oil contamination marine conditions.
We hence propose that hydrocarbon-degrading bacteria rather than SRB is the major contributor of MIC in the early stage of crude oil pollution in surface seawater. Although their abundance was low in the original coastal seawater, hydrocarbon-degrading bacteria like Alcanivorax and Marinobacter increased rapidly once the intrusion of crude oil, and dominated both the planktonic and surface microbial community after 25 days of exposure. These taxa can attach to the steel surfaces and contribute to biofilm formation. In the initial period (0–55 day), aerobic hydrocarbon degradation was the major activity performed by the dominant oil-degraders. The end-product of complete microbial aerobic hydrocarbon degradation is CO2 which generates less threat to pitting corrosion than corrosive sulfide produced by SRB6,26 compared with that in No Oil group. This may explain why the pitting corrosion seems to be inhibited in the initial stage.
Along with the maturation of biofilm and formation of corrosion products, heterogeneous microenvironments with oxic and anoxic areas formed on steel surfaces. It is further confirmed by the strengthened O2 respiration-related genes and anaerobic NO3− reduction-related genes. The heterogeneity of surface microenvironments primarily formed by these hydrocarbon-degrading bacteria may lead to the formation of corrosion zones, which is called oxygen concentration cell containing cathodic (high concentrations of dissolved oxygen) and anodic (low concentrations of dissolved oxygen) areas59. The stimulated microbial O2 respiration implied that O2 also plays roles in accelerating corrosion. In the presence of O2, surface-bound iron-sulfide is oxidized and more surface reactions take place, whereas in the absence of oxygen corrosion will be slow as surface ions are derivatized60. These factors accelerate steel corrosion in oil-containing seawater.
Another cause of accelerated steel corrosion is the produced organic acids. Some members of hydrocarbon-degraders such as Alcanivorax sp. may perform anaerobic fermentation of oil55 within the anoxic areas of the surface in the later period (55–85 day). This was supported by the detection of anaerobic hydrocarbon degradation-related genes (Fig. 6C) and the iron-carbonate corrosion products (Fig. 4). Microbial anaerobic fermentation produces a series of low-molecular-weight organic substances as end-products, such as lactate and butyrate, which were detected in high concentrations in seawaters (Fig. 1C). These organic acids can accelerate steel corrosion by destroying the protective rust layer based on the local acidic microenvironments61, and provide carbon sources for the growth of SRB which were observed increasing over the incubation time. The above factors contribute to the accelerated uniform and localized corrosion observed in the later incubation stages. These findings provide guidance to substantial studies on steel corrosion in petroleum-hydrocarbon contaminated seawaters.
Methods
Experimental design
Coastal surface seawater that used as both medium and inoculum sources from an oil wharf (120.24°E, 35.98°N) in Qingdao Port, China were collected and stored in sterilized bottles. Since marine sediment could be the main source of taxa in biofilms formed on steel surfaces proven in previous field studies62, surface sediments of the same sampling sites were also collected using a bottom grab. The seawater and sediment samples were kept in ice boxes before transporting to the laboratory as soon as possible and were used for further treatment within 24 h. The major components of the used crude oil were n-alkane (66.7%), iso-alkane (14.1%), naphthalene (8.23%), aromatics (7.14%) and cycloalkane (3.77%) (Supplementary Fig. 1). A commonly used marine steel, X70 steel was fabricated to 40 × 20 × 5 mm plates. The composition of X70 was as follows (wt %): C ≤ 0.16, Si ≤ 0.45, Mn ≤ 1.70, P ≤ 0.02, S ≤ 0.01, V ≤ 0.06, Nb ≤ 0.05, Ti ≤ 0.06, B ≤ 0.45, bal. Fe. The surface of X70 plates was polished using P120 to P800 grit SiC papers and cleaned with ethanol before drying in a baking oven. The plates were weighed and then stored in a sealed desiccator. Afterwards, they were further sterilized by irradiation under a UV lamp for 30 min before starting the experiments.
To examine the impact of crude oil exposure on steel corrosion, crude oil amendment (With Oil) and unamendment groups (No Oil) with fresh seawater and sediments were set up. 1.5 L of fresh seawater and 0.5 L of sediments were put into 2 L sterilized bottles which had sampling ports located at the seawater and sediment phases for subsequent sampling. Steel plates were suspended at a depth of 5 cm below the seawater surfaces. Five milliliter crude oil and the same volume of sterilized water were added aseptically into the seawater phase of With Oil and No Oil groups using sterile syringes, respectively. The bottles were sealed by parafilm to permit gas out and in but prevent microbial contamination. Crude oil amendment and unamendment groups with autoclaved inocula (121 °C for 20 min; 20 psi) were set as sterile controls. Triplicates were performed in each experimental group. All the incubations were kept in dark environments at room temperature (25 ± 2 °C) for 85 days. Time series (25, 55, 85 days) were analyzed for the treatments to detect dynamic geochemical factors and bacterial communities in response to oil addition.
Analysis of geochemical factors
For detection of hydrocarbons in original seawater, seawater samples were pretreated before determination by GC-MS (Agilent 7980A GC; MS: 5975C) (Agilent Technologies, Santa Clara, USA) as previously described6. The pretreatment process was as followed: 20 mL of seawater was acidified to pH 2 by 6 N HCl and was performed extraction using the equal volume of ethyl acetate. The extracted hydrocarbons were concentrated to 100 μL, which were then derivated using N,O-bis (trimethylsilyl) trifluoroacetamide (BSTFA). pH and sulfate concentration of the seawater in the treatments were detected after 25, 55, and 85 day’s incubation, respectively. pH of each treatment was determined in a laminar flow after sampling 3 mL seawaters from the simulators. For detection of sulfate, 1 mL seawater in each treatment was filtered through 0.22 μm filter membranes and then measured using inductively coupled plasma optical emission spectroscopy (ICP-OES) (PerkinElmer, Waltham, MA, USA). Concentrations of low-weight organic substances in filtered seawaters after 85 days incubation were measured using LC-20AT high-performance liquid chromatography (HPLC) (Shimadzu Corporation, Kyoto, Japan).
Analysis of corrosion morphology, mineralogy, and corrosion rate
At the end of the incubation, the tested plates were recovered. The surface morphology and element composition were visualized by field emission scanning electron microscopy (FESEM, Zeiss Ultra 55, Germany) with an energy-dispersive spectrometer (EDS, INCAx, Oxford, UK). Sample preparation for SEM and EDS was performed according to the previous study63,64. For further determination of the crystal minerals, corrosion products were collected from the steel surfaces, dried and crushed to a fine powder. The samples were analyzed by X-ray diffraction (XRD, Rigaku/max-Ultima IV, Japan) under the following conditions: 40 kV, 30 mA, graphite-filtered Cu Ka radiation (λ = 0.1542 nm). To measure the corrosion rate, corrosion products of triplicates plates were removed according to GB/T16545-2015 standard and the plates were weighed again to determine the total weight loss. The corresponding general corrosion rate (mm per year) was calculated from weight loss data based on ASTM Standard G1-03. To characterize the localized corrosion damage, the cleaned plates were scanned using SEM and confocal laser scanning microscope (CLSM) (LSM 510, Carl Zeiss, Jena, Germany).
Enumeration of functional microorganisms
The numbers of culturable SRB and APB in seawater were counted using the most-probable number (MPN) dilution test. Enumeration of SRB was performed according to GB/T14643.5-2009 standard with a modified SRB medium. The SRB enumeration medium contained: 0.5 g KH2PO4, 0.1 g CaCl2·6H2O, 2.0 g MgSO4·7H2O, 25 g NaCl, 0.3 g (NH4)2Fe(SO4)2·6H2O, 0.1 g vitamin C, 3.5 mL sodium lactate and 1.0 g yeast extract in 1 L distilled water with pH value of 7.2 ± 0.2. APB was enumerated using phenol-red dextrose broth as previously reported65. The medium for APB contained 10.0 g peptone, 1.0 g beef extract, 5.0 g glucose, and 0.018 g phenol-red in 1 L distilled water with pH value of 7.4 ± 0.1. All MPN enumerations were performed in triplicates by serially diluting 1 mL of water sample in 9 mL of the sterilized medium. These anaerobic tubes filled with inoculated enumeration medium were kept at 30 °C for 30 days and 3 days, respectively. Individual tubes were scored positive for the growth of SRB if the medium color turned black or for the growth of APB if the medium color turned from red to yellow.
Characterization of planktonic and attached microbial community
16S rRNA gene sequencing was used to characterize the planktonic and attached microbial communities. For detection of planktonic microbial community, 400 mL seawater samples in With Oil and No Oil simulators were collected and concentrated on 0.22 μm filter membranes at the end of incubation. For detection of attached microbial community, triplicate biofilms (with some rust) on the surface of steel at all three time points (25, 55, and 85 days) were sampled by sterile scrapers. Once finishing sampling, genomic DNA was extracted within 24 h using a FastDNA SPIN kit for soil (MoBio Laboratories, Carlsbad, CA, USA) according to the manufacturer’s instructions. The quality of extracted DNA was checked using NanoDrop One spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA) and agarose gel (1%) electrophoresis. After construction of sequencing libraries as previously reported63, the biofilm and seawater samples were pair-end sequenced on Illumina Novaseq PE250 platform.
The raw data were merged using FLASH (V1.2.7)66, removed chimera using UCHIME67, and quality-filtered using QIIME (V1.9.1)68 subsequently. UPARSE (V7.0.1001)69 was used to cluster the obtained effective tags into operational taxonomic units (OTUs) at 97% similarity. Representative sequences were picked and assigned to SSU rRNA database57 for taxonomy annotation (threshold 0.8–1) using Mothur (V1.40.45)58. For further diversity analysis between different samples, the sequencing data were normalized to the same number of reads in each sample. Alpha-diversity indexes including Chao1, ACE, Shannon, and Simpson were calculated in QIIME platform. The functional genes were annotated against KEGG database70 based on the sequencing data using PICRUSt71. Significantly different taxa (biomarker) enriched in each group were indicated by the linear discriminant analysis (LDA) based on Kruskal−Wallis tests using LEfSe software (V1.0)39 with LDA score >3.5. T-test was used to determine the difference in individual functional genes between different treatments. P < 0.05 was regarded as significant.
Data availability
Sequencing data of samples from the microcosm systems were deposited in NCBI Short Read Archive (SRA) database under Bioproject accession number PRJNA438021, with Biosample numbers SAMN22141952-22141979.
References
Hou, B. et al. The cost of corrosion in China. npj Mater. Degrad. 1, 4 (2017).
Bonifay, V. et al. Metabolomic and metagenomic analysis of two crude oil production pipelines experiencing differential rates of corrosion. Front. Microbiol. 8, 99 (2017).
Vigneron, A., Head, I. M. & Tsesmetzis, N. Damage to offshore production facilities by corrosive microbial biofilms. Appl. Microbiol. Biotechnol. 102, 2525–2533 (2018).
Jack, T. ASM Handbook: Failure Analysis and Prevention, Vol. 11 (ASM International, 2021).
Little, B. J. et al. Microbially influenced corrosion-Any progress? Corros. Sci. 170, 108641 (2020).
Aktas, D. F. et al. Anaerobic metabolism of biodiesel and its impact on metal corrosion. Energy Fuels 24, 2924–2928 (2010).
Lyles, C. N. et al. Impact of organosulfur content on diesel fuel stability and implications for carbon steel corrosion. Environ. Sci. Technol. 47, 6052–6062 (2013).
Gieg, L. M., Jack, T. R. & Foght, J. M. Biological souring and mitigation in oil reservoirs. Appl. Microbiol. Biotechnol. 92, 263–282 (2011).
Davidova, I. A., Duncan, K. E., Choi, O. K. & Suflita, J. M. Desulfoglaeba alkanexedens gen. nov., sp nov., an n-alkane-degrading, sulfate-reducing bacterium. Int. J. Syst. Evol. Microbiol. 56, 2737–2742 (2006).
Lyles, C. N., Le, H. M., Beasley, W. H., McInerney, M. J. & Suflita, J. M. Anaerobic hydrocarbon and fatty acid metabolism by syntrophic bacteria and their impact on carbon steel corrosion. Front. Microbiol. 5, 114 (2014).
Liang, R. X. et al. Anaerobic biodegradation of alternative fuels and associated biocorrosion of carbon steel in marine environments. Environ. Sci. Technol. 50, 4844–4853 (2016).
Hasegawa, R., Toyama, K., Miyanaga, K. & Tanji, Y. Identification of crude-oil components and microorganisms that cause souring under anaerobic conditions. Appl. Microbiol. Biotechnol. 98, 1853–1861 (2014).
Little, B. J. & Lee, J. S. Microbiologically influenced corrosion: an update. Int. Mater. Rev. 59, 384–393 (2014).
Dong, Y. et al. Severe microbiologically influenced corrosion of S32654 super austenitic stainless steel by acid-producing bacterium Acidithiobacillus caldus SM-1. Bioelectrochemistry 123, 34–44 (2018).
Prince, R. C. et al. The primary biodegradation of dispersed crude oil in the sea. Chemosphere 90, 521–526 (2013).
Cheng, F. Monitor safety of aged fuel pipelines. Nature 529, 156–156 (2016).
Kvenvolden, K. A. & Cooper, C. K. Natural seepage of crude oil into the marine environment. Geo-Mar. Lett. 23, 140–146 (2003).
Kostka, J. E. et al. Hydrocarbon-degrading bacteria and the bacterial community response in Gulf of Mexico beach sands impacted by the Deepwater Horizon oil spill. Appl. Environ. Microbiol. 77, 7962–7974 (2011).
Acosta-Gonzalez, A., Rossello-Mora, R. & Marques, S. Characterization of the anaerobic microbial community in oil-polluted subtidal sediments: aromatic biodegradation potential after the Prestige oil spill. Environ. Microbiol. 15, 77–92 (2013).
Fowler, S. J., Toth, C. R. A. & Gieg, L. M. Community structure in methanogenic enrichments provides insight into syntrophic interactions in hydrocarbon-impacted environments. Front. Microbiol. 7, 562 (2016).
Hu, P. et al. Simulation of Deepwater Horizon oil plume reveals substrate specialization within a complex community of hydrocarbon degraders. Proc. Natl Acad. Sci. USA 114, 7432–7437 (2017).
Head, I. M., Jones, D. M. & Roling, W. F. M. Marine microorganisms make a meal of oil. Nat. Rev. Microbiol. 4, 173–182 (2006).
Lee, J. S. et al. Sulphide production and corrosion in seawaters during exposure to FAME diesel. Biofouling 28, 465–478 (2012).
Mugge, R. L., Lee, J. S., Brown, T. T. & Hamdan, L. J. Marine biofilm bacterial community response and carbon steel loss following Deepwater Horizon spill contaminant exposure. Biofouling 13, 870–882 (2019).
Goñi-Urriza, M. & Duran, R. Microbial Communities Utilizing Hydrocarbons and Lipids: Members, Metagenomics and Ecophysiology. Handbook of Hydrocarbon and Lipid Microbiology 19–35 (Springer, 2019).
Aktas, D. F. et al. Effects of oxygen on biodegradation of fuels in a corroding environment. Int. Biodeterior. Biodegrad. 81, 114–126 (2013).
Xu, D. et al. Accelerated corrosion of 2205 duplex stainless steel caused by marine aerobic Pseudomonas aeruginosa biofilm. Bioelectrochemistry 113, 1–8 (2017).
Zhang, Y. et al. Analysis of marine microbial communities colonizing various metallic materials and rust layers. Biofouling 35, 429–442 (2019).
Postma, D. & Jakobsen, R. Redox zonation: equilibrium constraints on the Fe(III)/SO4-reduction interface. Geochim. Cosmochim. Acta 60, 3169–3175 (1996).
Fauville, A., Mayer, B., Frommichen, R., Friese, K. & Veizer, J. Chemical and isotopic evidence for accelerated bacterial sulphate reduction in acid mining lakes after addition of organic carbon: laboratory batch experiments. Chem. Geol. 204, 325–344 (2004).
Fan, W., Yang, Y. S., Du, X. Q., Lu, Y. & Yang, M. X. Finger-printing biodegradation of petroleum contamination in shallow groundwater and soil system using hydro-bio-geochemical markers and modelling support. Water Air Soil Pollut. 220, 253–263 (2011).
Hazen, T. C., Prince, R. C. & Mahmoudi, N. Marine oil biodegradation. Environ. Sci. Technol. 50, 2121–2129 (2015).
Moreno, R. & Rojo, F. Aerobic Utilization of Hydrocarbons, Oils, and Lipids. Handbook of Hydrocarbon and Lipid Microbiology 117–142 (Springer, 2019).
Mugge, R. L., Lee, J. S., Brown, T. T. & Hamdan, L. J. Marine biofilm bacterial community response and carbon steel loss following Deepwater Horizon spill contaminant exposure. Biofouling 35, 870–882 (2019).
Dickinson, W. H. & Lewandowski, Z. Manganese biofouling and the corrosion behavior of stainless steel. Biofouling 10, 79–93 (1996).
Rajasekar, A. et al. Bacterial degradation of naphtha and its influence on corrosion. Corros. Sci. 47, 257–271 (2005).
Remazeilles, C., Leveque, F., Conforto, E. & Refait, P. Long-term alteration processes of iron fasteners extracted from archaeological shipwrecks aged in biologically active waterlogged media. Corros. Sci. 181, 109231 (2021).
Pourbaix, M. Atlas of Electrochemical Equilibria in Aqueous Solution (Pergamon Press, 1966).
Rabus, R. Petroleum Microbiology. (ASM Press, 2005).
Su, H., Mi, S. F., Peng, X. W. & Han, Y. The mutual influence between corrosion and the surrounding soil microbial communities of buried petroleum pipelines. RSC Adv. 9, 18930–18940 (2019).
Hara, A., Syutsubo, K. & Harayama, S. Alcanivorax which prevails in oil-contaminated seawater exhibits broad substrate specificity for alkane degradation. Environ. Microbiol. 5, 746–753 (2003).
Mason, O. U. et al. Metagenome, metatranscriptome and single-cell sequencing reveal microbial response to Deepwater Horizon oil spill. ISME J. 6, 1715–1727 (2012).
Kasai, Y. et al. Predominant growth of Alcanivorax strains in oil-contaminated and nutrient-supplemented sea water. Environ. Microbiol. 4, 141–147 (2002).
Bazylinski, D. A. et al. Magnetovibrio blakemorei gen. nov., sp nov., a magnetotactic bacterium (Alphaproteobacteria: Rhodospirillaceae) isolated from a salt marsh. Int. J. Syst. Evol. Microbiol. 63, 1824–1833 (2013).
Wei, X., Vajrala, N., Hauser, L., Sayavedra-Soto, L. A. & Arp, D. J. Iron nutrition and physiological responses to iron stress in Nitrosomonas europaea. Arch. Microbiol. 186, 107–118 (2006).
Li, X. et al. Analysis of bacterial community composition of corroded steel immersed in Sanya and Xiamen seawaters in China via method of Illumina MiSeq sequencing. Front. Microbiol. 8, 1737 (2017).
Duan, J. et al. Corrosion of carbon steel influenced by anaerobic biofilm in natural seawater. Electrochim. Acta 54, 22–28 (2008).
Yu, L., Duan, J. Z., Du, X. Q., Huang, Y. L. & Hou, B. R. Accelerated anaerobic corrosion of electroactive sulfate-reducing bacteria by electrochemical impedance spectroscopy and chronoamperometry. Electrochem. Commun. 26, 101–104 (2013).
Kem, M. P., Zane, H. K., Springer, S. D., Gauglitz, J. M. & Butler, A. Amphiphilic siderophore production by oil-associating microbes. Metallomics 6, 1150–1155 (2014).
Denaro, R. et al. Alcanivorax borkumensis produces an extracellular siderophore in iron-limitation condition maintaining the hydrocarbon-degradation efficiency. Mar. Genomics 17, 43–52 (2014).
Cappello, S. & Yakimov, M. M. Handbook of Hydrocarbon and Lipid Microbiology (Springer, 2010).
Debeljak, P., Toulza, E., Beier, S., Blain, S. & Obernosterer, I. Microbial iron metabolism as revealed by gene expression profiles in contrasted Southern Ocean regimes. Environ. Microbiol. 21, 2360–2374 (2019).
Zedelius, J. et al. Alkane degradation under anoxic conditions by a nitrate-reducing bacterium with possible involvement of the electron acceptor in substrate activation. Environ. Microbiol. Rep. 3, 125–135 (2011).
Scott, N. M. et al. The microbial nitrogen cycling potential is impacted by polyaromatic hydrocarbon pollution of marine sediments. Front. Microbiol. 5, 108 (2014).
Yakimov, M. M., Golyshin, P. N., Crisafi, F., Denaro, R. & Giuliano, L. Taxonomy, Genomics and Ecophysiology of Hydrocarbon-Degrading Microbes. Handbook of Hydrocarbon and Lipid Microbiology 167–179 (Springer, 2019).
Grossi, V., Cravo-Laureau, C., Guyoneaud, R., Ranchou-Peyruse, A. & Hirschler-Rea, A. Metabolism of n-alkanes and n-alkeries by anaerobic bacteria: a summary. Org. Geochem. 39, 1197–1203 (2008).
Heider, J. & Schühle, K. The Prokaryotes: Prokaryotic Physiology and Biochemistry (Springer, 2013).
Schmitz, R. A., Daniel, R., Deppenmeier, U. & Gottschalk, G. The Prokaryotes (Springer, 2006).
Picioreanu, C. & van Loosdrecht, M. C. M. A mathematical model for initiation of microbiologically influenced corrosion by differential aeration. J. Electrochem. Soc. 149, B211–B223 (2002).
Hamilton, W. A. Microbially influenced corrosion as a model system for the study of metal microbe interactions: a unifying electron transfer hypothesis. Biofouling 19, 65–76 (2003).
Xu, D., Li, Y. & Gu, T. Mechanistic modeling of biocorrosion caused by biofilms of sulfate reducing bacteria and acid producing bacteria. Bioelectrochemistry 110, 52–58 (2016).
Mugge, R. L. et al. Deep-sea biofilms, historic shipwreck preservation and the Deepwater Horizon spill. Front. Mar. Sci. 6, 48 (2019).
Zhang, Y. et al. Metagenomic resolution of functional diversity in copper surface-associated marine biofilms. Front. Microbiol. 10, 2863 (2019).
Tang, H. Y. et al. Stainless steel corrosion via direct iron-to-microbe electron transfer by Geobacter species. ISME J. 15, 3084–3093 (2021).
Struchtemeyer, C. G. & Elshahed, M. S. Bacterial communities associated with hydraulic fracturing fluids in thermogenic natural gas wells in North Central Texas, USA. FEMS Microbiol. Ecol. 81, 13–25 (2012).
Magoč, T. & Salzberg, S. L. FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27, 2957–2963 (2011).
Edgar, R. C., Haas, B. J., Clemente, J. C., Quince, C. & Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27, 2194–2200 (2011).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 7, 335–336 (2010).
Edgar, R. C. UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 10, 996–998 (2013).
Kanehisa, M., Furumichi, M., Tanabe, M., Sato, Y. & Morishima, K. KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. 45, D353–D361 (2017).
Langille, M. G. I. et al. Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat. Biotechnol. 31, 814–823 (2013).
Acknowledgements
This work was funded by the Natural Science Foundation of Shandong Province (No. ZR2021QD099), the China Postdoctoral Science Foundation (No. 2021M690152) and the National Natural Science Foundation of China (No. 42076044, No. 41806090).
Author information
Authors and Affiliations
Contributions
Y.Z.: Conceptualization, Methodology, Software, Investigation, Writing—Original Draft; X.Z., F.G., X.D., J.S., B.Z.: Resources, Visualization, Investigation; R.Z.: Methodology, Writing—Original Draft, Project administration; J.D.: Conceptualization, Supervision, Project administration, Funding acquisition; B.H.: Supervision
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, Y., Zhai, X., Guan, F. et al. Microbiologically influenced corrosion of steel in coastal surface seawater contaminated by crude oil. npj Mater Degrad 6, 35 (2022). https://doi.org/10.1038/s41529-022-00242-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41529-022-00242-4
This article is cited by
-
Improving the efficiency of DNA extraction from iron incrustations and oilfield-produced water
Scientific Reports (2024)
-
Metagenomic insights into nutrient and hypoxic microbial communities at the macrofouling/steel interface leading to severe MIC
npj Materials Degradation (2023)
-
Biodegradation of materials: building bridges between scientific disciplines
npj Materials Degradation (2023)
-
Steel rust layers immersed in the South China Sea with a highly corrosive Desulfovibrio strain
npj Materials Degradation (2022)
-
A comparison study of crevice corrosion on typical stainless steels under biofouling and artificial configurations
npj Materials Degradation (2022)