Abstract
The recent incidental discovery of an asymptomatic venous thrombosis (VT) in the internal jugular vein of an astronaut on the International Space Station prompted a necessary, immediate response from the space medicine community. The European Space Agency formed a topical team to review the pathophysiology, risk and clinical presentation of venous thrombosis and the evaluation of its prevention, diagnosis, mitigation, and management strategies in spaceflight. In this article, we discuss the findings of the ESA VT Topical Team over its 2-year term, report the key gaps as we see them in the above areas which are hindering understanding VT in space. We provide research recommendations in a stepwise manner that build upon existing resources, and highlight the initial steps required to enable further evaluation of this newly identified pertinent medical risk.
Similar content being viewed by others
Introduction
A recent study investigating astronaut jugular venous flow inflight on the International Space Station (ISS) reported the incidental finding of a persistent asymptomatic obstructive left internal jugular (IJV) venous thrombosis (VT) in a single crewmember1. Furthermore, an additional suspected case was reported following retrospective analysis of the images from the other 11 crew members who participated in the study2. Venous thrombi can lead to localised pain, redness, distal swelling, venous ischaemia, skin necrosis and even organ dysfunction3. However, the most significant complication is lung embolization of thrombotic fragments, termed pulmonary embolism (PE) that can lead to cardiorespiratory insufficiency, which can be fatal. Thus, the potential identification of VT, a mission-critical medical condition, in such a highly selected, previously considered low-risk population is concerning and warrants urgent investigation.
As a result, the European Space Agency (ESA) supported the creation in 2020 of a ‘Topical Team’ comprising of international subject matter experts in coagulation, venous thrombosis, prophylaxis, and treatment of thrombo-embolic events. The creation of this team was additionally supported by the Cardiovascular, Pulmonary, Renal and Lymphatic Working Group of the ESA SciSpacE roadmap. An operationally focused research strategy is required to define the pathophysiological processes leading to VT presentation in spaceflight, potential clinical presentation, its risk (and the factors that may affect risk), whilst proposing evaluation of candidate diagnostic, mitigation, and management strategies compatible with human spaceflight. The team was supported by individuals familiar with Human Spaceflight medical operations to identify relevant knowledge gaps.
The objectives of the team were to:
-
1.
Identify key knowledge gaps and technological/procedural development needs in the (proposed) domains of:
-
11.
Venous system status, hemodynamics and coagulation cascade in microgravity (Pathophysiology working group);
-
12.
Risk of VT and its clinical presentation in microgravity (Risk working group);
-
13.
VT diagnostic systems compatible with the constraints of spaceflight (Operational Diagnostic working group);
-
14.
Management of VT in microgravity (Operational Management working group).
-
11.
-
2.
To determine a cohesive and appropriate operationally driven research and technological/procedural evaluation strategy that addresses the identified gaps, needs, risks and risk factors, whilst also proposing evaluation pathways of candidate diagnostic, mitigation and management strategies compatible with future long duration/exploration space missions.
Initially four working groups were formed to address the specific domains, however during monthly full group meetings where ongoing research and key findings of the subgroups were discussed, working groups I & II were combined. This paper will discuss the findings of the topical team, key knowledge gaps, and provide recommendations for future research along European and international space research goals.
Venous thrombus in space: what do we know?
VT Pathophysiology and risk factors (working group I)
To date, no symptomatic cases of VT have been identified in 609 astronauts that have undertaken space travel (https://www.worldspaceflight.com/bios/stats.php). In 2019, a cohort study of 11 ISS crew members showed 6 individuals with apparent stagnant or retrograde IJV flow on approximately flight day 50, with one developing occlusive IJV thrombosis2, the management of which is discussed separately by Aunon-Chancellor et al1. An additional potential partial IJV thrombus was identified in another crew member retrospectively1,2.
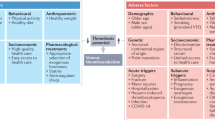
These findings suggest that the underlying VT incidence in space may not be dissimilar to that in the general population. For instance, the average rate of venous thrombus in the United States is 1–2 per 1000 people4. A calculation of the 95%CI of the proportion states that a symptomatic VT rate of 0/609 astronauts places the upper bound of the 95%CI at 0.6%, similar to the symptomatic VT rate in postoperative patients (at 0.3–0.7%)5. Concerningly, VT in neck veins may be associated with relatively poor outcomes6, yet are rare on Earth in the absence of specific risk factors7, accounting for 4–10% of overall VT or ~4–10 per 100,000 persons per year8. Currently, astronauts are a carefully selected cohort that lack many classical VT risk factors (detailed in Table 1), however, studies suggest that up to 50% of VT on Earth have no identifiable risk factors9.
There may be space- or microgravity-specific risk factors, such as the removal of tissue compressive forces along with cephalad fluid shifting that may also cause pathologies such as Spaceflight Associated Neuro-ocular Syndrome (SANS)10,11, which is suggested by the finding of flow abnormalities in the aforementioned studies. However, whether IJV flow was correctly identified as stagnant or retrograde in the initial cohort study, considering the technological limitations and the relative inexperience of crew as sonographers, is unknown12. However, a recent update by Pavela et al. 13 also documented retrograde IJV flow in 2/11 astronauts undergoing routine venous ultrasound surveillance in flight.
A recent systematic review reported that microgravity and its ground-based analogues, chiefly 6° head down tilted bed rest (HDTBR), may induce an enhanced coagulation state, most prominently in the venous system due to cephalad fluid shifting during gravitational unloading14. Some evidence of changes in venous flow, distension, pressures, endothelial damage, and possibly hypercoagulability exists, suggesting that all aspects of Virchow’s Triad may be modulated but the data is not conclusive14. While unexplored by Kim et al. 14, the radiation environment in Low Earth Orbit (LEO) may also have an effect, particularly as radiotherapy is associated with a significant increase in the risk of VT, including cerebral bleeding15. However, whether such changes precipitate an actual increased VT risk in spaceflight remains to be determined, given the lack of VT in ground-based analogues and limited sample of VT in spaceflight. Additionally, the association of radiotherapy with VT on the ground is often within the context of malignancy, which may be a risk.
Following spaceflight, a return to activity in a 1 G gravitational field may also constitute a VT risk factor. Indeed, changes in G-loading have been demonstrated to be high risk periods, with brief periods of hypergravity activating haemostasis in healthy volunteers16. However, the only documented VT to date in spaceflight was undetectable upon landing, and was discovered months after the hypergravity of launch1. While a return to 1 G after prolonged exposure to 0 G may induce haemostasis due to the relative hypergravity of the changed gravitational field, this has not been thoroughly explored.
One set of VT risk factors of increasing relevance are those related to sex17,18 given that whilst to date (as of March 2022), only 67 women have flown into space19, representing <10% of the astronaut population20. This situation is evolving, with 50% (9/18) of those selected by NASA in 2020 to prepare for the Artemis programme (NASA Artemis. at https://www.nasa.gov/specials/artemis/) being females. Indeed, whilst there are some physiological differences that should be considered21, females may also present operational physiological advantages for extended spaceflight due to decreased (on average) body size and energy consumption22 compared to their male counterparts23. Sex-based risks include the optional but frequent use of hormonal contraception in female astronauts to suppress menstruation17,18,24,25. The elevated risk is highest during the first months but remains significantly increased compared to non-users, depending on the type of hormonal contraceptive prescribed (ie. 1st or 2nd generation, (2–3x risk), versus 3rd or 4th generation (2–5x risk))18,26,27. Moreover, increased risk persists up to 3 months following discontinuation18. The presence of thrombophilia further increases VT risk. There is currently no indication for hereditary or acquired thrombophilia screening in astronauts without a personal or family history of VT or without any other comorbidity. Whether this needs to be re-considered remains to be determined—particularly as thrombophilia testing has little relevance for terrestrial VT management28,29.
Finally, as space travel is opening to a wider population, conventional risk factors associated with VTs may become more relevant to the practice of aerospace medicine. While the medical requirements for private spaceflight participants are currently less stringent than space agency crews, they will evolve30. Consideration of pre-existing conditions and comorbidities such as age, obesity, personal history of VT (VT or PE), family history of unprovoked VT and cancer (and its treatment) will become even more important for longer duration spaceflight participants.
VT diagnosis
Given the limited resources31 and absence of clinical presentation, VT diagnostic testing is not performed in space. However, in symptomatic patients a range of approaches are taken.
On Earth, B-mode compression ultrasound (CUS) is reported to be a sensitive and specific diagnostic method for acute VT in the peripheral veins of both the lower and upper limbs in addition to the jugular veins32,33,34,35. The technique relies on venous compressibility to ‘rule-out’ the presence of VT, with non-compressibility combined with the direct imaging of the thrombus confirming a VT diagnosis. Colour Doppler Ultrasound is an adjunct to B-mode CUS. By evaluating venous blood flow, it helps to delineate the thrombus if it is partially obstructing the vein, or show no flow when the thrombus is completely occlusive32,34,35.
Slow, stagnant, or retrograde venous flow may be important clues to understanding if a clot is formed, in the process of forming or even predict where one may in the future. The combination of Colour Doppler Ultrasound for the direct investigation of central vein thromboses (abdominal, pelvic and intrathoracic veins) with the Doppler signal patterns at the level of the peripheral veins appears to provide high diagnostic accuracy, although this technique has not been subject to the same level of validation compared to peripheral veins in the legs32,33.
In spaceflight, the diagnostic accuracy of conventional B-mode US/ CUS/ Colour Doppler US/ Doppler US is unknown. Furthermore, reduced, stagnant and/or retrograde venous flow may occur not due to VT, but rather—particularly in the upper-limb and jugular veins—due to volume overload and elevated venous pressures secondary to the classic cephalad fluid shift36. In fact, the ultrasound used to measure venous blood flow velocity on ISS, and subsequently qualitatively to determine the presence of turbulent, slow, stagnant or retrograde venous flow is intended to be applied to the high velocities associated with arterial flow12. That said, discerning the presence or absence of flow is sufficient in most cases, if the pressure applied at the probe is appropriate and consistent.
Hence, whilst the compression ultrasound (CUS) test can rule-out the diagnosis of thrombosis in a peripheral vein if it is compressible, it may lack specificity if the vein segment is not compressible in spaceflight. This lack of specificity due to higher pressures needed to compress, and thus collapse a vein may potentially be alleviated by simply using an adjacent artery as a visual ‘pressure gauge’. For instance, by increasing the pressure applied at the ultrasound probe to that required to almost close the adjacent artery, the visualised vein segment should be completely compressed37.
It must be noted however that manual control of applied pressure in microgravity is difficult even if combined with the use of the Crew Medical Restraint System (CMRS). Currently, safety concerns relating to pressure control preclude the use of autonomous probes. Thus, even with remote guidance (from ground-based sonographers), the crew, despite being inexperienced sonographers, must generate application pressure and thus significantly affect image acquisition. This is particularly important in, and around the neck as inadvertent activation of the carotid sinus baroreceptors could precipitate hypotension and bradycardia in individuals with carotid sinus hypersensitivity38. Thus, poor technique and image quality could translate into flow measurement error, particularly in the case of no-flow characterisation.
Furthermore, ultrasonic flow evaluation requires several assumptions including linear flow with respect to the vessel lumen and probe positioning12. This, assumption is in fact known to be violated in the IJV39. However, angle-independent methods are being developed in addition to technologies such as ultra-fast US and vector projectile imaging that show promise in facilitating more accurate evaluation of turbulent, slow, stagnant, and retrograde flow40.
D-dimer (product of fibrin, a protein fragment present in blood following fibrinolytic clot degradation) is a test used terrestrially to rule out lower limb VT and PE3 although it is less established for the diagnosis of upper-limb VT. The key utility is that a low D-dimer in conjunction with a low Wells score can be used to exclude lower limb VT/PE without further imaging41. However, given the lack of clarity regarding sensitivity to VT in the upper body and the ‘normal range’ in spaceflight it is unclear whether D-dimer assessment would be a valid diagnostic approach. Additionally, D-dimer is not currently available or validated for the spaceflight context, nor is the Wells score, and therefore is of limited interest for future diagnosis of VT during spaceflight.
Management
Thus far, the experience in managing IJV thrombosis in space is limited to the single asymptomatic individual. The astronaut was anticoagulated with once daily enoxaparin (1.5 mg/kg, then 1 mg/kg to extend supplies) transitioning to apixaban 5 mg twice daily at 42 days, with dose reduction at 3 months. To negate the risk of excessive bleeding due to traumatic injury on re-entry treatment, the anticoagulant was stopped 4 days prior to return to Earth1. Thought was also given to the provision of an antidote in the event of anticoagulation-related bleeding1.
On Earth, oral anticoagulants including direct factor Xa inhibitors (rivaroxaban, apixaban, edoxaban), direct thrombin inhibitors (dabigatran), vitamin K antagonists (warfarin), and subcutaneous anticoagulants such as low molecular weight heparins or fondaparinux are approved for VT management. Current international guidelines or consensus documents42 recommend direct oral anticoagulants (DOACs) as the first line treatment in the absence of contraindications.
Furthermore, antidotes are available for rivaroxaban and apixaban (andexanet alfa), dabigatran (idarucizumab) and warfarin (vitamin K and prothrombin complex concentrate)43 but not for LMWH, though there is partial efficacy of protamine for this purpose44. The availability of an antidote is a key advantage as rapid reversibility may be required in the case of traumatic injury and may reduce the need for monitoring. However, costs in replacing medications with limited shelf life may be prohibitive and impractical in the face of the low likelihood of VT and subsequent anticoagulant-related bleeding.
On Earth, anticoagulation medication is usually prescribed for 3 months in patients with proximal VT if the risk of recurrence is low (i.e., major transient/reversible risk factors)28. Longer term anticoagulation medication can be considered for those with low bleeding risk and high recurrence rate (i.e., major thrombophilia, persistent or non-identified risk factors). A further important consideration is the risk of embolization from VT in spaceflight. On Earth, upper extremity VT is associated with a significantly (5–8%) lower embolization risk than lower extremity VT45. However, theoretically, IJV VTs could propagate to more proximal vein segments and cause superior vena cava thrombosis and cannot be simply extrapolated from Earth to space.
Gap analysis and recommendations
Over the course of the topical team’s tenure, we have published on the likelihood of flow abnormalities in contributing to VT formation12, endothelial disruption related risks of VT46, and diagnostic modalities for VT in space (in review). We found that current tools on board the ISS are insufficient to characterize blood flow patterns that may contribute to clot formation and that there are no validated biomarkers that reliably predict increased likelihood of VT formation in otherwise healthy people. Based upon these publications and through the meetings of the topical team, knowledge gaps in each of the working group areas were identified. These key knowledge gaps are shown in Tables 2–5, along with recommendations for future research to address said gaps.
While the topical team was subdivided into four initial subgroups, the main objective of the team, and space medicine, is to prevent medical emergencies from occurring, and developing a reasonable management plan for the rare instances they do occur. To capture this goal and show the interdependencies of our findings, a visual representation of the above recommendations and the pathways in which they enable the primary prevention and management of VT in space is shown in Fig. 1, considering the recommendations which must be addressed first to enable the implementation of the other recommendations.
Discussion
There continue to be many unknowns regarding the pathophysiology, risk, prevention, diagnosis, management, and follow up of VT occurrences during spaceflight. With upcoming missions to the Moon and Mars, differences in medical resources and crew autonomy will further complicate the priority of each of the above recommendations. For example, advanced diagnostics that require from-ground input are unlikely to be undertaken for missions with significant communication delays, while initial investigation into the nature of clotting in microgravity may be undertaken preferentially in the low Earth orbit environment. The VT Topical Team suggest that space agencies and commercial operators consider the recommendations of this report to inform, and mitigate the potential risk posed by VT in future spaceflight.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
No datasets were generated as a result of this research.
References
Aunõn, S. M., Pattarini, J. M., Moll, S. & Sargsyan, A. Venous thrombosis during spaceflight. N. Engl. J. Med. 382, 89–90 (2020).
Marshall-Goebel, K. et al. Assessment of jugular venous blood flow stasis and thrombosis during spaceflight. JAMA Netw. Open 2, e1915011 (2019).
Mazzolai, L. et al. Diagnosis and management of acute deep vein thrombosis: a joint consensus document from the European Society of Cardiology working groups of aorta and peripheral vascular diseases and pulmonary circulation and right ventricular function. Eur. Heart J. 39, 4208–4218 (2018).
Beckman, M. G., Hooper, W. C., Critchley, S. E. & Ortel, T. L. Venous thromboembolism. Am. J. Prev. Med. 38, S495–S501 (2010).
De Martino, R. R. et al. Impact of screening versus symptomatic measurement of deep vein thrombosis in a national quality improvement registry. J. Vasc. Surg. 56, 1045–1051.e1 (2012).
Ascher, E., Salles-Cunha, S. & Hingorani, A. Morbidity and mortality associated with internal jugular vein thromboses. Vasc. Endovasc. Surg. 39, 335–339 (2005).
Spencer, F. A., Emery, C., Lessard, D. & Goldberg, R. J. Upper extremity deep vein thrombosis: a community-based perspective. Am. J. Med. 120, 678–684.e1 (2007).
Heil, J., Miesbach, W., Vogl, T., Bechstein, W. O. & Reinisch, A. Deep vein thrombosis of the upper extremity. Dtsch. Arztebl. Int. https://doi.org/10.3238/arztebl.2017.0244 (2017).
Heit, J. A. The epidemiology of venous thromboembolism in the community: implications for prevention and management. J. Thromb. Thrombolysis 21, 23–29 (2006).
Lee, A. G., Mader, T. H., Gibson, C. R., Brunstetter, T. J. & Tarver, W. J. Space flight-associated neuro-ocular syndrome (SANS). Eye 32, 1164–1167 (2018).
Buckey, J. C. et al. Microgravity-induced ocular changes are related to body weight. Am. J. Physiol. Integr. Comp. Physiol. 315, R496–R499 (2018).
Harris, K. M. et al. Going against the flow: are venous thromboembolism and impaired cerebral drainage critical risks for spaceflight? J. Appl. Physiol. https://doi.org/10.1152/japplphysiol.00425.2021 (2021).
Pavela, J. et al. Surveillance for jugular venous thrombosis in astronauts. Vasc. Med. https://doi.org/10.1177/1358863X221086619 (2022).
Kim, D. S. et al. The effect of microgravity on the human venous system and blood coagulation: a systematic review. Exp. Physiol. https://doi.org/10.1113/EP089409 (2021).
Guy, J.-B. et al. Venous thromboembolism in radiation therapy cancer patients: findings from the RIETE registry. Crit. Rev. Oncol. Hematol. 113, 83–89 (2017).
Limper, U. et al. Simulated hypergravity activates hemostasis in healthy volunteers. J. Am. Heart Assoc. 9, e016479 (2020).
Zwart, S. R., Auñón-Chancellor, S. M., Heer, M., Melin, M. M. & Smith, S. M. Albumin, oral contraceptives, and venous thromboembolism risk in astronauts. J. Appl. Physiol. https://doi.org/10.1152/japplphysiol.00024.2022 (2022)
Jain, V., Ploutz-Snyder, R., Young, M., Charvat, J. M. & Wotring, V. E. Potential venous thromboembolism risk in female astronauts. Aerosp. Med. Hum. Perform. 91, 432–439 (2020).
NASA. Women’s History Month: Most Recent Female Station Crew Members. https://www.nasa.gov/mission_pages/station/research/news/whm-recent-female-astronauts (2022).
Smith, M. G., Kelley, M. & Basner, M. A brief history of spaceflight from 1961 to 2020: an analysis of missions and astronaut demographics. Acta Astronaut. 175, 290–299 (2020).
Mark, S. et al. The impact of sex and gender on adaptation to space: executive summary. J. Women’s Heal 23, 941–947 (2014).
Scott, J. P. R., Green, D. A., Weerts, G. & Cheuvront, S. N. Body size and countermeasure exercise: implications upon resource utilization during human space exploration missions with female astronauts. Sci. Rep. 10, 13836 (2022).
Scott, J. P. R., Green, D. A., Weerts, G. & Cheuvront, S. N. Body size and its implications upon resource utilization during human space exploration missions. Sci. Rep. 10, 13836 (2020).
Jain, V. & Wotring, V. E. Medically induced amenorrhea in female astronauts. npj Microgravity 2, 16008 (2016).
Steller, J. G. et al. Menstrual management considerations in the space environment. REACH 23–24, 100044 (2021).
Steller, J. G. et al. Gynecologic risk mitigation considerations for long-duration spaceflight. Aerosp. Med. Hum. Perform. 91, 543–564 (2020).
de Bastos, M. et al. Combined oral contraceptives: venous thrombosis. Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD010813.pub2 (2014).
Ortel, T. L. et al. American Society of Hematology 2020 guidelines for management of venous thromboembolism: treatment of deep vein thrombosis and pulmonary embolism. Blood Adv. 4, 4693–4738 (2020).
National Institute for Health and Care Excellence. Venous thromboembolic diseases: diagnosis, management and thrombophilia testing. NICE Guidel. 158, 12–15 (2015).
Bogomolov, V. V. et al. International space station medical standards and certification for space flight participants. Aviat. Space Environ. Med. 78, 1162–1169 (2007).
Scott, J. P. R., Weber, T. & Green, D. A. Introduction to the frontiers research topic: optimization of exercise countermeasures for human space flight—lessons from terrestrial physiology and operational considerations. Front. Physiol. 10, 173 (2019).
Bhatt, M. et al. Diagnosis of deep vein thrombosis of the lower extremity: a systematic review and meta-analysis of test accuracy. Blood Adv. 4, 1250–1264 (2020).
Patel, P. et al. Diagnosis of deep vein thrombosis of the upper extremity: a systematic review and meta-analysis of test accuracy. Blood Adv. 4, 2516–2522 (2020).
Goodacre, S., Sampson, F., Thomas, S., van Beek, E. & Sutton, A. Systematic review and meta-analysis of the diagnostic accuracy of ultrasonography for deep vein thrombosis. BMC Med. Imaging 5, 6 (2005).
Needleman, L. et al. Ultrasound for lower extremity deep venous thrombosis. Circulation 137, 1505–1515 (2018).
Harris, K. M., Petersen, L. G. & Weber, T. Reviving lower body negative pressure as a countermeasure to prevent pathological vascular and ocular changes in microgravity. npj Microgravity. https://doi.org/10.1038/s41526-020-00127-3 (2020).
Martin, D. S. et al. Internal jugular pressure increases during parabolic flight. Physiol. Rep. 4, e13068 (2016).
Strasberg, B. et al. Carotid sinus hypersensitivity and the carotid sinus syndrome. Prog. Cardiovasc. Dis. 31, 379–391 (1989).
Ogoh, S. et al. Coupling between arterial and venous cerebral blood flow during postural change. Am. J. Physiol. Regul. Integr. Comp. Physiol. 311, R1255–R1261 (2016).
Au, J. S. et al. Ultrasound vector projectile imaging for detection of altered carotid bifurcation hemodynamics during reductions in cardiac output. Med. Phys. 47, 431–440 (2020).
Johnson, E. D., Schell, J. C. & Rodgers, G. M. The D‐dimer assay. Am. J. Hematol. https://doi.org/10.1002/ajh.25482 (2019).
Yip, G. & Hull, R. Overview of the Treatment of Lower Extremity Deep Vein Thrombosis (DVT)—UpToDate. https://www.uptodate.com/contents/overview-of-the-treatment-of-lower-extremity-deep-vein-thrombosis-dvt?search=vtetreatment&source=search_result&selectedTitle=2~150&usage_type=default&display_rank=2#H119624123 (2022).
Tomaselli, G. F. et al. 2020 ACC expert consensus decision pathway on management of bleeding in patients on oral anticoagulants. J. Am. Coll. Cardiol. 76, 594–622 (2020).
Drugs.com. Protamine Monograph for Professionals. https://www.drugs.com/monograph/protamine.html#uses (2022).
Muñoz, F. J. et al. Clinical outcome of patients with upper-extremity deep vein thrombosis. Chest 133, 143–148 (2008).
Harris, K. et al. Search for venous endothelial biomarkers heralding venous thromboembolism in space: a qualitative systematic review of terrestrial studies. Front. Physiol. 13, 1–10 (2022).
Acknowledgements
The authors would like to acknowledge the members of the topical team who contributed to the ideas and recommendations provided in this manuscript: ESA VT Topical Team Members. A.B. A.P.B. Andrew Winnard. Angelique Van Ombergen. A.E. Bianca Brix. B.G. D.K.G. D.A.G. D.S.K. Gerhard Cvirn. Giovanni Baldasarre. Guy Meyer. Hanns-Christian Gunga. James Pavela. J.J. Jens Tank. Jutta Gärtner. K.M.H. Kristina Kofler. L.R. L.G.P. L.M. L.Z. Myles George Harris. Patrick De Boever. P.z.E. Rado Pišot. Robert Riddell. Roopen Arya. Sargsyan Ashot. Sergi Vaquer. Setfan Du Plessis. T.W. T.H.K. Ulrich Limper. We are thankful to the European Space Agency and NASA for financial support of this project. The European Space Agency provided funding to cover the open access cost of this publication. Publication costs were covered specifically from the ESA-sponsored Topical Team on “Pathophysiology, risk and clinical presentation of venous thromboembolism (VTE) and its evaluation of its prevention, diagnosis, mitigation and management strategies in spaceflight” (Grant number 4000131108/20/NL/PG/pt) and NASA Grant number 80NSSC19K0020, PI. The project was funded by the European Space Agency and KBR GmbH. The funder (KBR GmbH) provided support in the form of salaries for the authors T.W. and D.A.G. but did not have any additional role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
K.M.H., R.A., L.R., T.W. organized the contributions of all subgroups and formed the final version of the manuscript. A.E., D.S.K., D.A.G., T.W., R.A., L.R. finalized the individual sections of literature review for the four subgroups. D.A.G., A.P.B., A.C., R.A., D.K.G., L.G.P., P.z.E, L.Z., L.M., A.E. edited the written manuscript and generated the tables. K.M.H., R.A., T.W., D.A.G., D.K.G, L.G.P., A.E., L.R., T.H.K., L.M., A.P.B., D.S.K., R.H.O.E., P.z.E., B.G., L.Z., G.B., K.T., S.B., JJ, A.P.B., A.C., T.R., N.G. contributed to the initial literature reviews, the writing of the sections, the generation of the recommendations and gaps, and reviewed the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Harris, K.M., Arya, R., Elias, A. et al. Pathophysiology, risk, diagnosis, and management of venous thrombosis in space: where are we now?. npj Microgravity 9, 17 (2023). https://doi.org/10.1038/s41526-023-00260-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41526-023-00260-9
This article is cited by
-
Effects of whole-body vibration or resistive-vibration exercise on blood clotting and related biomarkers: a systematic review
npj Microgravity (2023)
-
Retinal venular vessel diameters are smaller during ten days of bed rest
Scientific Reports (2023)