Abstract
The understanding of transport and mixing in fluids in the presence and in the absence of external fields and reactions represents a challenging topic of strategic relevance for space exploration. Indeed, mixing and transport of components in a fluid are especially important during long-term space missions where fuels, food and other materials, needed for the sustainability of long space travels, must be processed under microgravity conditions. So far, the processes of transport and mixing have been investigated mainly at the macroscopic and microscopic scale. Their investigation at the mesoscopic scale is becoming increasingly important for the understanding of mass transfer in confined systems, such as porous media, biological systems and microfluidic systems. Microgravity conditions will provide the opportunity to analyze the effect of external fields and reactions on optimizing mixing and transport in the absence of the convective flows induced by buoyancy on Earth. This would be of great practical applicative relevance to handle complex fluids under microgravity conditions for the processing of materials in space.
Similar content being viewed by others
Introduction
The diffusion of molecules is a fundamental mass transfer process where the random thermal motion of molecules in multi-component mixtures determines the relative motion of one species with respect to the others. The presence of external fields or of chemical reactions can significantly change the dynamics. A typical example is represented by a liquid mixture under the action of a macroscopic temperature gradient that determines a non-equilibrium mass flow of particles through the Soret effect. The presence of gravity can have a strong influence on the stability of a liquid mixture undergoing diffusion. A meaningful example is represented by convective instabilities driven by double diffusion, which can be triggered even when a liquid mixture exhibits an initially stable density profile. The interplay of the effect of gravity, of other external fields and of reactions can give rise to a complex stability diagram which shows that purely diffusive processes in multi-component mixtures can be hardly investigated on earth. For this reason, during the last 20 years, the European Space Agency has largely invested on the realization of Space experiments for the investigation of diffusion and thermodiffusion processes under microgravity conditions1. Within this context, the GRADFLEX project investigated heat and mass transfer diffusion processes at the mesoscopic scale on the FOTON M3 spaceship in 2007, providing convincing evidence that both processes are accompanied by large amplitude non-equilibrium fluctuations whose largest size coincides with that of the sample2,3. The IVIDIL project investigated the interplay between diffusion and vibrations under microgravity conditions on the International Space Station in 2009–2010, with the outcome that high-frequency periodic forcing generates time-averaged flows, which substantially affect the diffusive mass transfer4. The SCCO project involved many experiments performed on several space platforms, jointly supported by ESA and the China National Space Center, and by petrochemical industries including TOTAL, PETROCHINA and RIPED. The aim of the project was the investigation of thermodiffusion of multi-component mixtures under reservoir thermodynamic conditions of interest for the petroleum industry, with a focus on understanding how thermodiffusion affects the stability of the system5,6,7. The DCMIX project was aimed at the investigation of thermodiffusion in ternary liquid mixtures. Four space missions on the International Space Station, supported by ESA and Roscosmos, were performed from 2012 to 2018. The project allowed a systematic investigation of thermal diffusion in several model ternary mixtures, including mixtures selected to extend to ternaries the Fontainebleu benchmark previously developed for binaries8. The CHYPI project has, on the other hand, explored in microgravity chemo-hydrodynamic patterns due to the interplay between molecular diffusion and reactions9,10,11,12 .
In general, all these space projects were motivated by the fact that concentration gradients are associated with density gradients. As a result, any perturbation in the concentration of the mixture can either be stabilized by gravity or amplified by it, leading to the onset of convective instabilities.
Key knowledge gaps
Mixing and transport of mass and/or heat in fluids is at the heart of numerous applications. We discuss a selection of them and explain the advantage of microgravity conditions in answering key questions in their respective fields.
Confinement of fluctuations during diffusion processes
Mass diffusion and heat diffusion in liquid mixtures are triggered by composition and temperature gradients and are accompanied by long-ranged non-equilibrium fluctuations of concentration and temperature, respectively13,14. These fluctuations are limited in amplitude15 and lifetime16 by buoyancy forces. Gravity and diffusion are antagonist effects acting at different time scales that strongly depend on the fluctuation size. For small fluctuations, diffusion is the dominant mechanism while for large ones it is buoyancy. In the absence of gravity, fluctuations become giant—they grow proportional to the fourth power or their size—and they are limited only by the size of the sample container, as it has been demonstrated by the microgravity GRADFLEX experiment both for temperature and concentration fluctuations2,3 (Fig. 1).
False-colour shadowgraph images of non-equilibrium concentration fluctuations induced by the Soret effect in a polystyrene polymer solution on Earth (left) and in space (right) during the GRADFLEX experiment on FOTON M33. Scale bar: 2.0 mm.
The rigid boundaries influence large non-equilibrium fluctuations also in the presence of gravity. This confinement effect has been detected for concentration non-equilibrium fluctuations by dynamic-shadowgraphy and gives rise to a dramatic slowing-down of the dynamics of fluctuations17. For very thin samples, the confinement effect can be so large that the effect of buoyancy is totally cancelled out18. The forces generated on the fluctuations by the boundaries have a counterpart in a net force exerted on the boundaries themselves by the fluctuations. In fact, it has been predicted theoretically that the confinement of non-equilibrium fluctuations should lead to the development of non-equilibrium forces between the confining plates19,20. This phenomenon is similar to the critical Casimir effect, predicted in 1978 by Fisher and de Gennes21 and measured in 2008 by Hertlein et al.22, where equilibrium fluctuations become long-ranged close to the consolute critical point of a liquid mixture. Similar forces acting on colloidal particles dispersed in a critical mixture have been investigated under microgravity conditions in 201223 by means of Near Field Scattering during the SODI-COLLOID experiment of ESA on the ISS.
In the case of non-equilibrium Casimir forces, the system does not need to be in the vicinity of critical conditions, but only out-of-thermodynamic equilibrium, a rather generic situation that is encountered in many practical cases of scientific or technological interest19,20.
The forces generated by the confinement of long-ranged fluctuations are present also between macromolecules thus altering their interactions, like it has been demonstrated in the case of critical Casimir forces. The experimental investigation of confined non-equilibrium fluctuations has both fundamental relevance for the understanding of interactions in non-equilibrium systems at the mesoscopic scale, a problem of quite general interest, and applicative relevance for microfluidic devices, Micro-ElectroMechanical Systems and biological systems. Experimental attempts made on Earth to detect these interactions have been hampered by the presence of buoyancy. As described above, on Earth the range of fluctuations is limited by the gravity force, which limits their amplitude and lifetime at small wave vectors15. These two aspects make the microgravity environment ideally suited for the investigation of forces generated by the confinement of fluctuations. The Giant Fluctuations (NEUF-DIX) and TechNES projects of ESA24,25 investigate non-equilibrium fluctuations in complex fluids under microgravity conditions and include experiments where non-equilibrium fluctuations are confined by colloidal particles dispersed in the sample fluid. From an experimental point of view, temperature gradients can readily be prescribed at the sample boundaries. Concentration gradients can be realized by preparing stratified layers of different compositions16, but a more elegant way is provided by the Soret effect26,27,28. This effect allows one to induce the composition gradient by a temperature gradient starting with a homogeneous equilibrium system that can be stored for almost arbitrary times prior to the actual experiment. Due to the time required to establish stationary convection-free non-equilibrium conditions, ideal Low Earth Orbit (LEO) platforms are represented by the ISS.
Confinement effects on the spatio-temporal behaviour of fluctuating fields are not restricted to a finite size of the sample container. Inside a porous medium, the average effective diffusion process is slowed down by the tortuous pathway of the solid matrix29,30. Recent experimental observations by heterodyne dynamic light scattering31 suggest that the dynamics of small fluctuations in density are dependent on the aspect ratio between the size of the fluctuation to the diameter of the pore.
Non-equilibrium diffusion of particles in the presence and in the absence of external fields
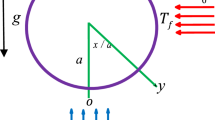
Particles in a fluid respond to gravity in a way that critically depends on their size a and the density mismatch Δρ between particles and fluid (Fig. 2). This is captured by the Péclet number Pe = a/lg, where \(l_g = k_BT/{{\Delta }}\rho V_pg\) is the sedimentation length, kBT is the thermal energy, Vp the volume of the particle and g the acceleration of gravity. Small colloidal particles, for which Pe << 1, and large granular ones, with Pe >> 1, behave differently: for colloids, the fluid acts as a thermal bath continuously exchanging energy with the particles and keeping them in perpetual Brownian motion; for granular systems, the effects of gravity dominate thermal agitation, rendering thermal fluctuations negligible. Accordingly, the granules sediment and accumulate at the bottom of the container (Fig. 2c, f). Inelastic collisions between granules result in energy distribution and frictional energy dissipation. This renders a continuous injection of energy necessary to maintain a fluid state32,33,34. This is achieved by, e.g. a gas or liquid flow or a mechanical or magnetic excitation, which, together with measurement facilities, has been realized for investigations in microgravity35 (Fig. 2b). Fluidized granular media represent an important model system to study mass and energy transport in athermal, intrinsically non-equilibrium conditions. Interestingly, after full equilibration, granules exhibit colloid-like features, including for instance density induced crystallization36,37, a diffusive-like particle dynamics in dilute and dense systems33,35,38. How similar colloids and fluidized granules are, is currently unclear. Microgravity experiments are an invaluable tool to avoid sedimentation, rendering an investigation of other factors easier.
Schematic representation of some key effects of gravity on small (colloidal) or large (granular) particles in a fluid. a In the absence of gravity, thermal fluctuations lead to a uniform distribution of colloidal particles. b For granular particles this process can be prohibitively slow, and hence injection of mechanical energy is required for the system to remain in a fluid state. c Gravity promotes a net flux of particles (sedimentation) along the direction of gravity. d If the mass distribution within each particle is not spherically symmetric, gravity results in a net torque on the particles, which can promote their orientational ordering. e For a diluted colloidal suspension, the steady-state distribution of particles in the presence of gravity is not homogeneous along the direction of gravity but corresponds to an exponential concentration profile. f In a granular medium, the particles densely accumulate at the bottom of the container, where they minimize their gravitational potential energy and form load-bearing frictional contacts.
Colloidal sedimentation is much better understood and investigated, starting with the seminal work of Jean Perrin39. Nevertheless, the presence of long-ranged hydrodynamic interactions still holds surprises: while theoretical models predict a divergence of the correlation length of velocity fluctuations in a sedimenting suspension, this divergence is not confirmed by experiments, leading to the so-called divergence paradox40,41. A possible solution may reside in the theoretical prediction that, during sedimentation, the fluctuations are quenched by gravity due to the coupling between the fluctuations and the concentration gradient42.
This quenching only occurs for length scales beyond a critical value, which may explain why large-scale fluctuations are not observed experimentally. The underlying theory42 also describes the occurrence of giant fluctuations during diffusion processes in molecular and macromolecular fluids13,15, whose properties have been extensively studied and confirmed also in microgravity during the GRADFLEX experiment3,43,44. More recently, giant fluctuations have also been confirmed in ground-based experiments with dilute16 and dense45 colloidal suspensions. However, the role of the coupling between fluctuations and concentration gradients for the divergence paradox needs to be clarified and requires experiments in which a homogeneous colloidal suspension is suddenly exposed to gravity and the spatio-temporal correlation of the fluctuations is measured as a function of time during the sedimentation. Such experiments are extremely challenging on ground because an artefact-free initial state is practically impossible to prepare. In contrast, microgravity platforms such as FLUMIAS, in which a microscope is coupled to a centrifuge, represent a unique opportunity: in microgravity a colloidal sample is left undisturbed to reach a homogenous state (Fig. 2a) and subsequently the centrifuge is started to induce sedimentation and the fluctuations are monitored (Fig. 2c) before an equilibrium concentration profile is attained (Fig. 2e). Varying the effective gravity between 0.01 and 1.1 g, the contribution of gravity can be singled out and hence the predictions42 tested.
The above examples focus on particles undergoing translational motion. Gravity can also induce rotation if a particle has an inhomogeneous or anisotropic density distribution (Fig. 2d). This includes particles used in self-assembly and some active Brownian particles, e.g. Janus particles. The effects of gravity46 are reduced in quasi-two-dimensional geometry. It is, however, desirable to extend experiments to three-dimensional situations47 for at least two reasons: first, to clarify the dependence of phenomena such as motility-induced phase separation on dimensionality48; second, to avoid direct or hydrodynamic interactions between the particles and the confining walls which might introduce spurious effects. Thus, microgravity experiments can offer opportunities for the investigation of non-equilibrium effects in a range of systems and various situations.
Transport in multi-scale soft matter systems under external forces; interfacial dynamics and particle transport in forced fluid systems
Many soft matter systems of practical relevance exhibit a high level of complexity. There is a large knowledge gap in situations where different factors interact, like the simultaneous presence of small molecules, entangled polymers and large particles, in conjunction with an external temperature gradient or other external fields49. Transport mechanisms in multi-scale soft matter are not governed by a single “pure” effect, and an understanding of cross-interactions and their coupling is essential. These could include, for example, the interplay between mass and heat diffusion, viscoelasticity and volume flow. Complexity can originate not only from the number of constituents and driving forces but also from an intrinsic asymmetry of the components. A deep understanding of such complex dynamics could be achieved by starting with the investigation of low-concentration systems with one external driving force, increasing the complexity level towards crowded systems and biological aspects. The obtained results could allow the development of novel detection techniques for in situ sensing.
The behaviour of interfaces50 plays an important role in forced multi-phase systems (Fig. 3), which arise in numerous applications in physics, engineering and biology. In particular, the capture of small particles dispersed in water (or brine) by viscoelastic fluids, such as mucus, is an issue of critical concern due to the growing problem of micro-plastic contamination in marine environments and their uptake by living organisms51. A systematic investigation of particle transport in the presence of more realistic flows, with time-dependent forcing representative of swimming, breathing through gills and wave motion, can be expected to provide a better understanding of the influence of fluid dynamics and interfacial phenomena on particle capture and to allow for more informed decisions about micro-plastics management and related problems such as filter design. Microgravity is particularly appealing for the analysis of inertial effects without the complications of buoyancy or sedimentation. Density matching (on ground) is undesirable, since it reduces inertial effects, and is impractical for realistic particle distributions. The exploration of interfacial dynamics and their role in particle uptake requires relatively long microgravity times (as on the ISS or Sounding Rockets) since the relevant phenomena develop and evolve slowly, depending on applied amplitude and frequency. With the low frequencies characteristic of swimming, breathing, or waves, sufficient particle capture events accumulate over many minutes. A better understanding of interface dynamics and particle transport has broad relevance for Human Space Exploration due to the importance of fluid management in life support and propulsion systems. Multi-phase systems also arise in numerous applications including resource extraction, which will be an important part of long-term Space Exploration.
Complex interfacial dynamics with FC-40 and 20 cSt silicone oil vibrated at 50 Hz in microgravity50.
Diffusion of fluid mixtures across the Widom line—CO2 sequestration
Until a decade ago, the supercritical region was believed to be a featureless, homogeneous fluid-phase. However, contrary to popular belief, physically observable liquid- and gas-like subdomains can be found in the supercritical region52. The transition between these two subdomains occurs at the so-called Widom line, which can be considered as a continuation of the vapour pressure curve up to ten times its critical value53. There is still a large knowledge gap in the research field targeting diffusion of mixtures in the critical region and crossing the Widom line which requires significant development of experimental and theoretical approaches. In fact, although several industrial processes operate just above the critical point of the fluid, i.e. 1 < T/Tc < 1.1 and 1 < p/pc < 1.5, where Tc and pc are the critical temperature and pressure, respectively, reliable experimental data on mass diffusion, and particularly, on thermodiffusion of mixtures are scarce at these conditions54. An attempt to characterize the Widom line has also been reported by investigating the fluid density through the refractive index55.
The focus of this research theme lies on the quantification and understanding of the anomalies in mass- and thermal diffusion mechanisms related to the critical point and the Widom line in fluid mixtures. At the Widom line, thermophysical properties of fluids show an anomalous behaviour where a small temperature or pressure variation can lead to drastic changes of density, enthalpy, viscosity, etc. At the critical plait point theoretical studies predict singularities, i.e. a vanishing value of the mass diffusion coefficient as well as a diverging Soret coefficient and strong component segregation. Furthermore, the steady state may need an infinite amount of time to settle. An increase of the Soret coefficient with a critical slow-down of mass diffusion towards another type of criticality, i.e. the continuous line of consolute (demixing) points in ternary mixtures, has been observed in experiments on the ISS (DCMIX2 and DCMIX4)1,56. At the Widom line and near critical regions, fluid mixtures are very sensitive to any external perturbation, even small deviations from linearity of the temperature gradient may easily induce convection, which may disturb or erase the separation of components that is the hallmark of the Soret effect. In microgravity conditions, natural convection disappears in liquids even if a temperature gradient is applied. This creates an excellent possibility for the measurement and overall understanding of mass diffusion and thermodiffusion of fluid mixtures across the Widom line and approaching the critical plait point. Furthermore, high-quality convection-free thermodiffusion reference data are necessary for the development of theoretical models for the prediction of Fick diffusion and Soret coefficients of multi-component fluid mixtures.
Of particular relevance is the investigation of carbon dioxide (CO2) mixtures in the context of carbon capture and sequestration. Indeed, one of the possible routes to reduce the level of CO2 in the atmosphere, following the GIEC recommendations57, is to capture CO2 at plants or directly from the air, and inject it into deep saline aquifers that are spread all over the world and specifically under the sea and can provide storage for multi-billion tons of CO258,59,60. Deep saline aquifers are huge reservoirs made of porous rock with moderately low permeability sandwiched by layers of rock with very low permeability, the cap-rock, sealing the container especially on the top. In the early phase after injection, the near-well zone is particularly impacted with strong geochemical reactivity associated with intense mass and heat exchanges61. CO2 sequestrated is therefore subject to high temperature gradients and may be found in states across the Widom line and close to the critical point (Fig. 4). The unavoidable presence of impurities under these conditions may lead to a variety of non-equilibrium processes, each being important to perform a detailed quantitative reservoir risk assessment. For instance, mass- and thermodiffusion may result in the accumulation of chemically active species near the top of the cap-rock provoking CO2 leakage. A systematic investigation and understanding of the thermal transport of impurities in CO2 reservoirs can be expected to reduce the uncertainties related to risk assessment of CO2 geological storage.
Schematic representation of carbon capture and sequestration in reservoirs. The inherent temperature gradient induces thermal diffusion of impurities from the bottom to the cap-rock of the reservoir. During injection, sequestration and storage, CO2 goes through different thermodynamic states as depicted in the right panel. The temperature and pressure dependence of the Widom line for pure CO2 was determined by the maxima of the isobaric heat capacity. The area between the blue and red lines represents the transition region between the liquid-like and gas-like subdomains of the supercritical fluid. Image of thermal-power plant by vectorpocket on Freepik.com (https://www.freepik.com/free-vector/heavy-industry-factory-working-thermal-power-plant-station-with-electricity_4758475.htm).
The understanding of the dissolution process of CO2 into the brine is then of major relevance for the geological storage of large quantities of CO2. So far, the process has been largely investigated at the macroscopic scale to understand how convective motions on Earth could enhance the efficiency of the mixing process62. The progression and reactivity of CO2 at the microscopic level of the pores or at the mesoscopic level of the porous matrix still needs to be fully understood. In particular, the investigation of the role of fluctuations at the mesoscopic scale and of possible capillarity effects at the pore level would be of relevance for the understanding of real systems. The identification of pure Marangoni flows requires microgravity conditions to discriminate surface tension effects from buoyancy-driven progression of the CO2 in the host phase porous matrix, which contains both gas and liquid phases. Studies of convective dissolution of CO2 in model porous systems would therefore benefit from investigation in microgravity.
Reactive flows
In the presence of reactions, the dynamics can become even more complex because chemical reactions can change a physical property of the solutions at hand and thereby induce convective motions63. Gradients of composition can induce gradients of density, which are prone, on earth, to trigger buoyancy-driven currents. In the presence of an interface between two immiscible solvents, gradients in concentration can also initiate gradients of surface tension inducing Marangoni flows. On earth, it is challenging to disentangle buoyancy effects from Marangoni effects. Microgravity has played an important role in that respect, allowing to isolate Marangoni convection triggered by reactions along the interface between two immiscible solvents64. In such two-phase layers, the dynamics of A + B → C fronts, where reactant A diffuses out of one solvent into the other immiscible solvent containing reactant B, have been studied in parabolic flights9. It has been shown that a modulation of gravity induces acceleration/deceleration of the front and increases product formation9. Similarly, deformation of an autocatalytic front due to convective flows induced by self-sustained surface tension gradients could be analyzed in detail in sounding rocket flights thanks to the absence of buoyancy effects (Fig. 5)10,11.
More recently, reactive fronts have been studied in flow-driven out-of-equilibrium conditions, which can be used to control the spatio-temporal distribution and amount of product C formed in time. Theoretical studies have shown that the temporal scaling of the properties of A + B → C fronts become then dependent on the injection rate65. On earth, testing these scalings is challenging because buoyancy-driven currents perturb the dynamics66. Microgravity conditions have here again allowed to test these scalings in absence of gravity (Fig. 6) and hence, have contributed to enlighten the influence that such density effects have on the behaviour of the front12.
Radial reaction-diffusion-advection front generated by a simple bimolecular reaction A + B → C when a reactant A is injected at a constant flow rate in a quasi-2D reactor prefilled with the reactant B. In the absence of buoyancy-driven currents under microgravity, the dark product C spreads as an expanding circle12. Scale bar: 10 mm.
A vast class of chemo-hydrodynamic patterns and instabilities remains, however, largely unexplored, namely the one involving a phase change like in precipitation fronts. In such systems, reactions locally generate a solid product, which may change the local density, viscosity or permeability of the medium. In addition, sedimentation can in some cases trigger a local clogging in confined reactors. Such dynamics have been recently studied for applications in CO2 sequestration techniques67,68, polymorph selection69,70, frontal polymerization71 or new material synthesis, to name a few. The specificity of precipitation reactions is that the solid product undergoes sedimentation and always changes the density of the solution, which naturally induces buoyancy-driven flows on earth. Disentangling such density-driven phenomena from other effects like viscosity or permeability related convective dynamics is crucial to optimize the processes but remains quite challenging72. Microgravity conditions will here play a crucial role in the future, providing substantial insights on the product properties, the yield of the reaction and the spatial distribution of the precipitate products. A further step advancing the knowledge on complex reactive flows includes precipitation processes in the vicinity of fluidic interfaces. The presence of the interface changes the nucleation of the precipitate particles, makes it necessary to consider Marangoni effects by changing reactant concentration, and can even lead to precipitate adsorption at the interface. These phenomena are strongly coupled to buoyancy effects so that a fundamental understanding can only be gained by microgravity experiments.
Perspectives
The investigation of transport and mixing in fluids under microgravity is needed to achieve a deep fundamental understanding of the behaviour of multi-component fluids at the mesoscopic scale in the presence and in the absence of external fields. This kind of investigation will shed new light on the transport and mixing processes occurring in the Universe under various gravity conditions. A strong priority of the investigation of diffusion processes in mixtures is its relevance for Space Exploration. In fact, the sustainability of long-duration space travel will rely critically on the ability to process materials in the fluid state on site73, to mix fluids for the preparation of food, heating or cooling living chambers homogeneously, processing the CO2 released by the astronauts, etc. All these processes involve the transport and mixing of fluids such as gases or liquids under microgravity, or under gravity levels different from those of Earth. The processing will involve complex fluid materials such as fuels, food and construction materials. A suitable platform for these types of studies will be represented by the Deep Space Gateway, and by other platforms used to reach the Moon and Mars. We envision that after a conceptual phase, the development of suitable experiments will be driven by the practical experience collected during long-term missions, where complex fluids will be stored and processed under microgravity and reduced gravity conditions. Mixing processes may occur over a very wide range of time scales. This is because, under microgravity conditions, mixing processes do not exhibit a typical length scale, besides the microscopic size of the molecules and macroscopic size of the container where the mixing is taking place. For these reasons, diffusion in mixtures at the mesoscopic scale can be also investigated by performing experiments lasting for times as short as a few minutes, which can be executed on LEO facilities.
The general idea of the investigation of diffusion in mixtures is thus to understand the main factors which control the mixing of fluids in microgravity and, on this basis, to develop efficient approaches for the optimization of the transport of species and/or heat during mixing under various conditions relevant to space exploration.
Benefit for Earth and industrial relevance
Besides space exploration, the understanding of transport and mixing in fluids is also a topic of great strategic and practical relevance for applications on earth. A huge variety of industrial processes are currently based on the processing of multi-component and multi-phase materials in a fluid state. Notable examples include the processing of foods, homogenization of blends in complex fluids like polymers, cements, metallic mixtures, as well as fluid control of chemical engineering processes. In porous media, transport is of tantamount importance in chemical engineering, oil recovery techniques, pollution remediation and the storage of CO2 into saline aquifers, to name a few examples. Depending on the goal, mixing between the displacing and the displaced fluids is either to be welcomed or avoided. Moreover, complex fluid materials structured at the mesoscopic scale are attracting increasing interest, due to the relevance of this range of length scales for living things. Of relevance are applications in the biomedical and pharmaceutical sectors, and reducing the pollution of micro-plastics. On Earth, mixing between reactive or non-reactive fluids of different composition and/or at different temperatures often involves various processes including buoyancy-driven flows. Microgravity conditions can help to isolate the other effects and allow a better understanding of their role in the control and efficiency of mixing and transport.
Outlook and summary
The investigation of the diffusive mass transport in liquid mixtures in the past and present years has largely benefitted from the opportunity of performing experiments under reduced gravity conditions by using sounding rockets and Low Earth Orbit platforms, such as the International Space Station. The reduced gravity conditions allowed to perform experiments under purely diffusive conditions, in the absence of convective instabilities driven by the gravitational acceleration, and to investigate the interplay between diffusion and chemical reactions, between vibrations and diffusion, to perform accurate measurements of the transport coefficients of multi-component mixtures, and to elucidate the role of fluctuations at the mesosocopic scale during diffusion processes. Although microgravity experiments are still important for a fundamental understanding of the diffusive mass transfer in complex fluids, the significance of these studies is gradually shifting towards the understanding of the behaviour of multi-component mixtures under hypergravity and reduced gravity conditions. This topic is of great relevance for long-duration space exploration missions, where the ability of processing complex liquids represents a strategic habilitating factor.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
References
Braibanti, M. et al. European Space Agency experiments on thermodiffusion of fluid mixtures in space. Eur. Phys. J. 42, 86 (2019).
Takacs, C. J. et al. Thermal fluctuations in a layer of liquid CS2 subjected to temperature gradients with and without the influence of gravity. Phys. Rev. Lett. 106, 244502 (2011).
Vailati, A. et al. Fractal fronts of diffusion in microgravity. Nat. Commun. 2, 290 (2011).
Shevtsova, V. et al. Experimental and theoretical study of vibration-induced thermal convection in low gravity. J. Fluid Mech. 767, 290 (2015).
Legros, J. C., Van Vaerenbergh, S., Decroly, Y. & Montel, F. Expériences en microgravité étudiant l’effet Soret: SCM, SCCO et MBIS. Entropie 198, 1 (1994).
Van Vaerenbergh, S., Srinivasan, S. & Saghir, M. Z. Thermodiffusion in multicomponent hydrocarbon mixtures: Experimental investigations and computational analysis. J. Chem. Phys. 131, 114505 (2009).
Galliero, G. et al. Thermodiffusion in multicomponent n-alkane mixtures. npj Microgravity 3, 20 (2017).
Platten, J. K. et al. Benchmark values for the Soret, thermal diffusion and diffusion coefficients of three binary organic liquid mixtures. Philos. Mag. 83, 1965 (2003).
Eckert, K., Rongy, L. & De Wit, A. A+ B→ C reaction fronts in Hele-Shaw cells under modulated gravitational acceleration. Phys. Chem. Chem. Phys. 14, 7337–7345 (2012).
Horváth, D. et al. Convective dynamics of traveling autocatalytic fronts in a modulated gravity field. Phys. Chem. Chem. Phys. 16, 26279–26287 (2014).
Bába, P. et al. Interaction of pure Marangoni convection with a propagating reactive interface under microgravity. Phys. Rev. Lett. 121, 024501 (2018).
Stergiou, Y. et al. Effects of gravity modulation on the dynamics of a radial A + B → C reaction front. Chem. Eng. Sci. 257, 117703 (2022).
Ortiz de Zarate, J. M. & Sengers, J. V. Hydrodynamic Fluctuations in Fluids and Fluid Mixtures (Elsevier, 2006).
Croccolo, F., Ortiz de Zárate, J. M. & Sengers, J. V. Colloquium: Non-local fluctuation phenomena in liquids. Eur. Phys. J. E 39, 125 (2016).
Vailati, A. & Giglio, M. Giant fluctuations in a free diffusion process. Nature 390, 262 (1997).
Croccolo, F., Brogioli, D., Vailati, A., Giglio, M. & Cannel, D. S. Effect of gravity on the dynamics of nonequilibrium fluctuations in a free-diffusion experiment. Ann. N. Y. Acad. Sci. 1077, 365 (2006).
Giraudet, C. et al. Slowing-down of non-equilibrium fluctuations in confinement. Europhys. Lett. 111, 60013 (2015).
Croccolo, F. & Bataller, H. Microgravity in a thin film: how confinement kills gravity. Eur. Phys. J. E 39, 132 (2016).
Kirkpatrick, T. R., Ortiz de Zárate, J. M. & Sengers, J. V. Giant Casimir effect in fluids in nonequilibrium steady states. Phys. Rev. Lett. 110, 235902 (2013).
Kirkpatrick, T. R., Ortiz de Zárate, J. M. & Sengers, J. V. Nonequilibrium Casimir-like forces in liquid mixtures. Phys. Rev. Lett. 115, 035901 (2015).
Fisher, M. E. & de Gennes, P. G. Phenomena at the walls in a critical binary mixture. C. R. Acad. Sci. Paris B 287, 207–209 (1978).
Hertlein, C., Helden, L., Gambassi, A., Dietrichand, S. & Bechinger, C. Direct measurement of critical Casimir forces. Nature 451, 172 (2008).
Veen, S. J. et al. Colloidal aggregation in microgravity by critical Casimir forces. Phys. Rev. Lett. 109, 248302 (2012).
Baaske, P. et al. Giant fluctuations induced by thermal diffusion in complex liquids. Eur. Phys. J. E 39, 119 (2016).
Vailati, A. et al. Giant fluctuations induced by thermal diffusion in complex liquids. Microgravity Sci. Technol. 32, 873–887 (2020).
Vailati, A. & Giglio, M. q divergence of nonequilibrium fluctuations and its gravity induced frustration in a temperature stressed liquid mixture. Phys. Rev. Lett. 77, 1484 (1996).
Croccolo, F., Bataller, H. & Scheffold, F. A light scattering study of non-equilibrium fluctuations in liquid mixtures to measure the Soret and mass diffusion coefficient. J. Chem. Phys. 137, 234202 (2012).
Köhler, W. & Morozov, K. I. The Soret effect in liquid mixtures—a review. J. Non-equilib. Thermodyn. 41, 151 (2016).
Giraudet, C. et al. Thermodiffusion of the tetrahydronaphthalene and dodecane mixture under high pressure and in porous medium. C. R. Mécanique 341, 340 (2013).
Reich, S. J. et al. Hindrance factor expression for diffusion in random mesoporous adsorbents obtained from pore-scale simulations in physical reconstructions. Ind. Eng. Chem. Res. 57, 3031 (2018).
Giraudet, C. et al. Diffusion of gold nanoparticles in inverse opals probed by heterodyne dynamic light scattering. Transp. Porous Media 131, 723 (2020).
Falcon, E. et al. Collision statistics in a dilute granular gas fluidized by vibrations in low gravity. Europhys. Lett. 74, 830 (2006).
Tatsumi, S., Murayama, Y., Hayakawa, H. & Sano, M. Experimental study on the kinetics of granular gases under microgravity. J. Fluid Mech. 641, 521 (2009).
Sack, A., Heckel, M., Kollmer, J. E., Zimber, F. & Pöschel, T. Energy dissipation in driven granular matter in the absence of gravity. Phys. Rev. Lett. 111, 018001 (2013).
Born, P. et al. Soft matter dynamics: a versatile microgravity platform to study dynamics in soft matter. Rev. Sci. Instrum. 92, 124503 (2021).
Messina, R. et al. Quantitatively mimicking wet colloidal suspensions with dry granular media. Sci. Rep. 5, 10348 (2015).
Sakaï, N., Moulinet, S., Lechenault, F. & Adda-Bedia, M. Experimental evidence of thermal-like behavior in dense granular suspensions. Phys. Rev. Lett. 122, 168001 (2019).
Born, P., Schmitz, J. & Sperl, M. Dense fluidized granular media in microgravity. npj Microgravity 3, 27 (2017).
Piazza, R. Settled and unsettled issues in particle settling. Rep. Progr. Phys. 77, 056602 (2014).
Guazzelli, E. & Hinch, J. Fluctuations and instability in sedimentation. Ann. Rev. Fluid Mech. 43, 97 (2011).
Segrè, P. N. In Fluids, Colloids and Soft Materials: An Introduction to Soft Matter Physics, 43–58 (Wiley, 2016).
Vailati, A. & Giglio, M. Nonequilibrium fluctuations in time-dependent diffusion processes. Phys. Rev. E 58, 4361 (1998).
Cerbino, R., Sun, Y., Donev, A. & Vailati, A. Dynamic scaling for the growth of non-equilibrium fluctuations during thermophoretic diffusion in microgravity. Sci. Rep. 5, 14486 (2015).
Croccolo, F., Giraudet, C., Bataller, H., Cerbino, R. & Vailati, A. Shadowgraph analysis of non-equilibrium fluctuations for measuring transport properties in microgravity in the GRADFLEX experiment. Microgravity Sci. Technol. 28, 467 (2016).
Giavazzi, F., Savorana, G., Vailati, A. & Cerbino, R. Structure and dynamics of concentration fluctuations in a non-equilibrium dense colloidal suspension. Soft Matter 12, 6588 (2016).
Campbell, A. I., Wittkowski, R., ten Hagen, B., Löwen, H. & Ebbens, S. J. Helical paths, gravitaxis, and separation phenomena for mass-anisotropic self-propelling colloids: Experiments versus theory. J. Chem. Phys. 147, 084905 (2017).
Keßler, R. et al. Direct-imaging of light-driven colloidal Janus particles in weightlessness. Rev. Sci. Instrum. 91, 013902 (2020).
Stenhammar, J., Marenduzzo, D., Allen, R. J. & Cates, M. E. Phase behaviour of active Brownian particles: the role of dimensionality. Soft Matter 10, 1489 (2014).
Jiang, H.-R., Wada, H., Yoshinaga, N. & Sano, M. Manipulation of colloids by a nonequilibrium depletion force in a temperature gradient. Phys. Rev. Lett. 102, 208301 (2009).
Salgado Sánchez, P. et al. Interfacial phenomena in immiscible liquids subjected to vibrations in microgravity. J. Fluid Mech. 865, 850–883 (2019).
Bourguignon, D. Plastics in a circular economy: opportunities and challenges. Eur. Parliam. Brief. PE 603, 940 (2017).
Simeoni, G. et al. The Widom line as the crossover between liquid-like and gas-like behaviour in supercritical fluids. Nat. Phys. 6, 503 (2010).
Banuti, D., Raju, M. & Ihme, M. Between supercritical liquids and gases—reconciling dynamic and thermodynamic state transitions. J. Supercrit. Fluids 165, 104895 (2020).
Guevara-Carrion, G., Ancherbak, S., Mialdun, A., Vrabec, J. & Shevtsova, V. Diffusion of methane in supercritical carbon dioxide across the Widom line. Sci. Rep. 9, 8466 (2019).
Giraudet, C., Marlin, L., Bégué, D., Croccolo, F. & Bataller, H. Concentration dependent refractive index of CO2/CH4 mixture in gaseous and supercritical phase. J. Chem. Phys 144, 134304 (2016).
Mialdun, A., Ryzhkov, I., Khlybov, O., Lyubimova, T. & Shevtsova, V. Measurement of Soret coefficients in a ternary mixture of toluene-methanol-cyclohexane in convection-free environment. J. Chem. Phys. 148, 044506 (2018).
Pathak, M. et al. Intergovernmental Panel on Climate Change, Working Group III Contribution to the IPCC Sixth Assessment Report (AR6): Technical Summary. https://www.ipcc.ch/report/ar6/wg3/downloads/report/IPCC_AR6_WGIII_TS.pdf. (2021).
Bachu, S. & Adams, J. J. Sequestration of CO2 in geological media in response to climate change: capacity of deep saline aquifers to sequester CO2 in solution. Energy Convers. Manag. 44, 3151–3175 (2003).
Bader, A. G. et al. CO2 storage capacity evaluation in deep saline aquifers for an industrial pilot selection. methodology and results of the France Nord Project. Energy Procedia 63, 2779–2788 (2014).
Michael, K. et al. Geological storage of CO2 in saline aquifers—a review of the experience from existing storage operations. Int. J. Greenh. Gas. Control 4, 659 (2010).
De Silva, G. P. D., Ranjith, P. G. & Perera, M. S. A. Geochemical aspects of CO2 sequestration in deep saline aquifers: a review. Fuel 155, 128–143 (2015).
Thomas, C., Dehaeck, S. & De Wit, A. Convective dissolution of CO2 in water and salt solutions. Int. J. Greenh. Gas. Control 72, 105–116 (2018).
De Wit, A. Chemo-hydrodynamic patterns and instabilities. Annu. Rev. Fluid Mech. 52, 531–555 (2020).
Eckert, K., Acker, M., Tadmouri, R. & Pimienta, V. Chemo-Marangoni convection driven by an interfacial reaction: Pattern formation and kinetics. Chaos Interdiscip. J. Nonlinear Sci. 22, 037112 (2012).
Brau, F., Schuszter, G. & De Wit, A. Flow Control of A + B → C Fronts by Radial Injection. Phys. Rev. Lett. 118, 134101 (2017).
Tóth, Á. et al. Effects of radial injection and solution thickness on the dynamics of confined A + B → C chemical fronts. Phys. Chem. Chem. Phys. 22, 10278–10285 (2020).
Schuszter, G., Brau, F. & De Wit, A. Calcium carbonate mineralization in a confined geometry. Environ. Sci. Technol. Lett. 3, 156–159 (2016).
Bere, K. V. et al. Enhancing the yield of calcium carbonate precipitation by obstacles in laminar flow in a confined geometry. Phys. Chem. Chem. Phys. 23, 15515–15521 (2021).
Bohner, B., Schuszter, G., Berkesi, O., Horváth, D. & Tóth, Á. Self-organization of calcium oxalate by flow-driven precipitation. Chem. Commun. 50, 4289–4291 (2014).
Ziemecka, I. et al. Polymorph selection of ROY by flow-driven crystallization. Crystals 9, 351 (2019).
Pojman J. A. In Polymer Science: A Comprehensive Reference, 957–980 (Elsevier, 2012).
Cartwright, J. H. E., Escribano, B., Sainz-Díaz, C. I. & Stodieck, L. S. Chemical-garden formation, morphology, and composition. II. Chem. Gard. Microgravity 27, 3294–3300 (2011).
Hessel, V., Sarafraz, M. M. & Tran, N. N. The resource gateway: microfluidics and requirements engineering for sustainable space systems. Chem. Eng. Sci. 225, 115774 (2020).
Acknowledgements
We are indebted to O. Minster, and M. Braibanti for a fruitful collaboration that led to the development of the ideas discussed in this publication. We acknowledge financial support from the European Space Agency.
Author information
Authors and Affiliations
Contributions
A.V. and A.D. drafted the manuscript and coordinated its critical revision. All the authors provided inputs for the drafting of the manuscript and contributed to its critical revision and to the literature survey: Section 1: A.V., M.C., W.K., H.B., F.C. and C.G.; Section 2: R.C., S.E. and F.G; Section 3: V.S., M.B.-A., W.K., A.M., J.P. and G.G.-C.; Section 4: V.S., M.B.-A., A.M., G.G.-C., H.B., F.C. and C.G.; Section 5: A.D., D.H. and K.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vailati, A., Bataller, H., Bou-Ali, M.M. et al. Diffusion in liquid mixtures. npj Microgravity 9, 1 (2023). https://doi.org/10.1038/s41526-022-00246-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41526-022-00246-z
This article is cited by
-
Unraveling dispersion and buoyancy dynamics around radial A + B → C reaction fronts: microgravity experiments and numerical simulations
npj Microgravity (2024)
-
Electromagnetic levitation containerless processing of metallic materials in microgravity: rapid solidification
npj Microgravity (2023)
-
Method of Taking into Account Influence of Thermal Shock on Dynamics of Small Satellite and its Use in Analysis of Microaccelerations
Microgravity Science and Technology (2023)