Abstract
Microorganisms impact spaceflight in a variety of ways. They play a positive role in biological systems, such as waste water treatment but can be problematic through buildups of biofilms that can affect advanced life support. Of special concern is the possibility that during extended missions, the microgravity environment will provide positive selection for undesirable genomic changes. Such changes could affect microbial antibiotic sensitivity and possibly pathogenicity. To evaluate this possibility, Escherichia coli (lac plus) cells were grown for over 1000 generations on Luria Broth medium under low-shear modeled microgravity conditions in a high aspect rotating vessel. This is the first study of its kind to grow bacteria for multiple generations over an extended period under low-shear modeled microgravity. Comparisons were made to a non-adaptive control strain using growth competitions. After 1000 generations, the final low-shear modeled microgravity-adapted strain readily outcompeted the unadapted lac minus strain. A portion of this advantage was maintained when the low-shear modeled microgravity strain was first grown in a shake flask environment for 10, 20, or 30 generations of growth. Genomic sequencing of the 1000 generation strain revealed 16 mutations. Of the five changes affecting codons, none were neutral. It is not clear how significant these mutations are as individual changes or as a group. It is concluded that part of the long-term adaptation to low-shear modeled microgravity is likely genomic. The strain was monitored for acquisition of antibiotic resistance by VITEK analysis throughout the adaptation period. Despite the evidence of genomic adaptation, resistance to a variety of antibiotics was never observed.
Similar content being viewed by others
Introduction
Human space travelers experience a unique environment that affects homeostasis and physiologic adaptation. Space flight has been shown to induce varied immune responses, many of them potentially detrimental.1 Impairment of the immune system may lead to higher risk of bacterial and/or viral infection during human space flight missions.2,3,4 In combination with this increased human risk, bacteria exposed to the same environments may become more virulent and/or antibiotic resistant as a result of exposure to the low shear environment induced.5,6,7
To better understand the effect of the space environment on microorganisms, a number of spaceflight8,9,10,11,12,13,14,15,16,17 and laboratory studies utilizing a low-shear modeled microgravity (LSMMG) environment have been undertaken.5,6,7, 13, 18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36 These experiments have shown changes in phenotypic microbial characteristics including microbial growth, morphology, metabolism, genetic transfer, stress susceptibility, and/or virulence in a variety of organisms including Salmonella enterica serovar typhimurium,8, 9, 31, 33, 35 Yersinia pestis,5, 10, 30 Escherichia coli,19, 20, 22, 29, 34, 36 Pseudomonas aeruginosa,12, 16, 17 Streptococcus mutans,37 Mycobacterium marinum 38 and Candida albicans.11, 18, 32 In an earlier study with Escherichia coli (E. coli), we carried out transcriptional studies on the K12 MG1655 strain grown in both rich and minimal medium under LSMMG.34 The results were compared to a normal gravity control. This study revealed reproducible changes in transcription. However, no specific LSMMG responsive genes were identified. Instead, reduction of fluid shear and a randomized gravity vector appeared to cause local extra-cellular environmental changes, which elicit cellular responses. Thus, even in the absence of significant genomic adaptation, an organism may be more virulent simply because it is already expressing genes that facilitate avoidance of the immune system17 or because genes associated with virulence are being expressed.21
LSMMG studies to date have been short-term, thereby providing only limited opportunity for genomic changes that might make observed responses permanent. The objective of the current study was to examine the extent to which extended exposure to the LSMMG environment leads to genomic adaptation. To address this, E. coli MG1655 cells were exposed to simulated microgravity conditions for 1000 generations (1000G) in rich medium. The subsequent analysis used competition assays and genomic sequencing to assess the adaptation of the 1000G strains. In addition, resistance to antibiotics was monitored throughout the adaptation period.
Results
Competition assays
In order to assess whether genomic changes account for a portion of the adaptation, erasure studies were conducted. The 1000G-adapted strain was grown in shake flasks under normal gravity for various periods of time. If the adaptation to the high aspect rotating vessel (HARV) environment, which was used to create the LSMMG conditions, was primarily phenotypic, the favorable changes acquired during HARV growth would be rapidly erased upon return to the shake flask environment. In contrast, genomic adaptations to the HARV environment rather than being erased would persist.
To assess the extent of such genomic adaptation, isogenic strains of E. coli that can be readily distinguished by colony color were used in competition studies. Initially, the two strains were competed against one another by growing them together. After one cycle of growth, the mixture of cells was streaked onto multiple plates. Following incubation, the number of colonies of each type were counted on each plate to determine the ratio of the lac plus and lac minus strains. The ratios seen on individual plates varied widely with large standard deviations (Fig. 1, Supplementary Table 1). Thus, to calculate overall ratios, the colony counts were pooled from all the plates used in multiple trials/sets. Each set (trial) refers to a different experiment. For example, the nine ‘control’ sets, correspond to nine different flasks containing the combined growth of the lac plus and lac minus cultures. Under LSMMG/HARV conditions, the unadapted E. coli MG1655 lac plus strain and the unadapted E. coli MG1655 lac minus strain both grew nearly equally well with a lac plus/lac minus ratio of 0.86. When the long-term-adapted 1000-G strain was competed vs. the unevolved E. coli MG1655 lac minus strain, the lac plus/lac minus ratio increased dramatically to 2.46. However, if the 1000G strain were first grown under shake flask conditions for 10, 20, or 30 generations, the ratio initially declined to 2.19 and subsequently was 2.12 and 1.78 (Fig. 1). Thus, 72% of the adaptive advantage survived the extended erasure (Supplementary Fig. 1). An alternative approach is to first determine ratios for each individual plate and then calculate averages. This allows statistical validation whereas the first method does not.
Ratio of cumulative totals (CT) of lac plus colonies vs. lac minus colonies from all the plates; Average * = Average of ratios of lac plus colonies vs. lac minus colonies from individual plates with SD; G = number of generations; 1000G = lac plus strain adapted to 1000 generations of LSMMG; 1000G-10E, 1000G-20E and 1000G-30E refer to the 1000G strain subjected to 10, 20 and 30 generations of ‘erasure’ on shaker flask conditions, respectively; UP unadapted lac plus, UM unadapted lac minus
Using this method, the respective ratios were 0.96 (with a robust standard deviation (SD) of 0.4 a coefficient of variation (CV) of 42%, and a confidence interval (CI) of 0.12), 3.71 (SD = 2.81, CV = 76%, CI = 0.9), 2.65 (SD = 1.52, CV = 57%, CI = 0.63), 1.95 (SD = 1.01, CV = 51%, CI = 0.46) and 1.83 (SD = 1.14, CV = 62%, CI = 0.49). The corresponding sample CV corrected for bias are 5.2, 8, 4.4, 2.9, and 3.9, respectively (Supplementary Table 1). Scatter plots performed on each of the datasets showed robust trend lines (Supplementary Fig. 2). In the overall analysis, approximately 50% of the advantage persists.
To further determine the variance differences, we applied the F-test to the lac plus vs lac minus ratio for two data sets. These were the unadapted lac plus strain vs. the unadapted lac minus strain and the 1000G-adapted lac plus strain vs. the unadapted lac minus strain. Given the vast variance differences between the two data sets as observed in the F-statistic value of 0.02 compared to the F Critical one-tail value of 0.6, we performed the t-test assuming unequal variances for this same pair of data sets. The t-test results show that the two-tailed and one-tailed p-values (9.2 × 10−15 and 1.8 × 10−14, respectively) are well under the statistical threshold of 0.05, thus clearly indicating the dominance of the 1000G plus strain over the unadapted lac minus strain when grown under LSMMG.
The paired t-test was then performed on each of the three data sets from the 1000G strain grown under shake flask conditions for 10, 20, or 30 generations (competed with the unadapted lac minus strain) against data from the 1000G-adapted strain grown with the unadapted lac minus strain. The results in all three cases show consistent two-tailed and one-tailed p-values of 0.5 and 1, respectively, thus, supporting our inference that the adaptation is only partially lost. A linear fit for the ratios (data sets) of 1000G strain grown under shake flask conditions for 10, 20, or 30 generations (competed with the unadapted lac minus strain) showed a trend consistent with our observations (Supplementary Fig. 3a). Similarly, a scatter plot of the same data set with the unadapted lac plus and minus ratio strain showed a correlation consistent with our observations (Supplementary Fig. 3b). The statistical values are provided in Supplementary Table 2.
In summary, extended exposure to the HARV conditions gave the MG1655 lac plus strain a significant adaptive advantage when the two strains were competed in the HARV. When the adapted strain was first exposed to shake flask conditions, the advantage was partially lost. However, even after three cycles of erasures, over 50% of the adaptation remained.
Genome sequencing
The genomic data gave no indication of contamination and the 16 S rRNA sequences were as expected for E. coli. Overall, 16 changes were found in the genome of the evolved 1000G E. coli MG1655 strain, compared to the control strain (Table 1). This included eight point mutations (six in genes surA, betA, ygfK, trkH, fimH, ylbE, and two intergenic (cspH-cspG, and ppiC-yifN), two deletions (one in the gene fhuA and the other intergenic insB-insA), and six insertions. Five of the insertions are mediated by transposons (cspC, insA-uspC, gatY-fbaB, yhhZ, fimE), while one is in the gene ylbE. There were five point mutations, resulting in amino acid changes, none of which were neutral. One large deletion eliminates 776 bases from an intergenic region (insB). A second one base deletion disrupts the reading frame of fhuA. In another case, a single base addition in combination with a single base point mutation converts a pseudogene (ylbE) into a potentially viable coding region. Five major transposon insertions occur at various loci, two of which are in intergenic non-coding regions, while three occur within coding genes. Of the three intragenic insertions, one (IS1) occurs within two genes involved in stress response and motility (Table 1). The description of the insertions as predicted by breseq is given in more detail in Fig. 2.
An insertion of the transposon IS1 to the left of position 1,905,401 in the forward orientation (+) is predicted (coordinate as annotated in NC_000913.2). At the point of insertion, it duplicates nine base pairs (1905401-1905409). Thus, the sequence goes up to 1,905,409 on the left side and then goes into the IS1 element, which is inverted, and then comes out at 1,905,401 and continues to higher coordinates. All of this occurs within the “cspC” gene, thereby likely making it dysfunctional. Likewise, transposon insertions have been predicted for positions 3,580,708 (four base pairs) and 4,540,601 (nine base pairs), disrupting the genes yhhZ (conserved protein) and fimE (tyrosine recombinase/inversion of on/off regulator of fimA), respectively. The JC (New junction) evidence thus shows the two new junctions (one on each side) that the mutations were predicted from. Two other transposon insertions occur in intergenic regions and may not have any impact on the actual genome itself. This figure was prepared by Quyen Tran
Antibiotic susceptibility—VITEK results
No significant changes in antibiotic resistance were observed when comparing the unevolved E. coli lac plus strain to the same strain grown under LSMMG conditions for 100, 200, 300, 400, 500, 600, 700, 800, 900, and 1000 generations (Supplementary Table 3). The initial strain and all the evolved strains were sensitive to all the antibiotics used in the VITEK analysis. The antibiotics used in this study included ampicillin, amoxicillin/clavulanic acid, piperacillin/tazobactam, cefalotin, cefazolin, cefuroxime, cefuroxime axetil, cefoxitin, cefpodoxime, ceftazidime, ceftriaxone, cefepime, gentamicin, tobramycin, ciprofloxacin, levofloxacin, norfloxacin, tetracycline, nitrofurantoin, and trimethoprim/sulfamethoxazole. Thus, no increase in resistance was observed in the evolved as compared to the unevolved control strain (Supplementary Fig. 4, Supplementary Table 3).
Discussion
The unevolved E. coli MG1655 lac plus strain does not initially possess a natural competitive (growth) advantage over the lac minus strain under LSMMG conditions. However, exposure to LSMMG over 1000 generations increases the fitness level of the lac plus strain. This is evident from the competition experiments in which the lac plus strain significantly outcompetes the unadapted E. coli MG1655 lac minus strain when both are grown together under LSMMG conditions. A portion of this adaptation is transient, which is clearly seen when the ability of the 1000G lac plus strain to outcompete the unadapted lac minus strain is decreased following erasure by overnight growth in the different environment found in shake flasks. However, repeated cycles of erasure have minimal additional effect, implying that there is a residual genomic component to the adaptation. Genomic adaptation to any new environment is in fact expected. In previous long term studies of E. coli, it was observed that the initial mutations were frequently adaptive rather than neutral.22, 23
Consistent with this, in the present case, genome re-sequencing revealed five changes in coding regions, none of which were neutral. In the case of surA (Q224K), ygfK (G412R), and trkH (G25R) a non-charged amino acid is converted to a basic residue. The gene betA undergoes a mutation changing a non-charged residue to an acidic residue (G9D). The fifth mutation in the gene fimH changes V43L with no net charge change. surA, fimH, and betA are of particular interest from an LSMMG adaptation perspective.
The protein encoded by surA plays a key role in pili biogenesis39 as well as the folding and assembly of fimbrial proteins40,41,42 on gram-negative bacterial surfaces, which enable attachment to surfaces of host cells and tissues for colonization and infection.43, 44 Thus surA indirectly influences bacterial adhesion. Furthermore, the SurA protein is conferred protection by the periplasmic acid stress chaperone HdeA,45 which was shown to be overexpressed in E. coli MG1655 when grown short-term under LSMMG.34 Thus, the surA mutation in the 1000G strain may have a key role in the response to LSMMG.
The gene fimH encodes a bacterial adhesion protein. FimH (along with other receptor/adhesion proteins) has been well-characterized for key structural and functional properties, as adaptation responses to increased shear stress on surfaces subject to differential flow dynamics.46,47,48,49 The V43L mutation is in the N-terminal lectin domain of FimH (N-FimH). Two residues, N46 and D47, in the vicinity of V43L, are important for the N-FimH domain’s binding and polymerization functions.50 Thus, it is possible that the V43L change in the N-FimH gene may influence its function.
The third gene, betA, is involved in osmotic stress response.51, 52 Surface-exposed fimbrial subunits are classified under the LSMMG regulon in S. enterica serovar Typhimurium, with direct implications for host immune system-pathogen interactions.7 The five observed non-synonymous mutations may be part of a regulon that facilitates adaptation of E. coli MG1655 strain to the LSMMG environment. Such adaptation may influence the bacterium’s ability to colonize the interior surfaces of the space station or spacecraft, including life support systems.
In addition, five changes are associated with insertion sequence (IS) elements. Such elements are known to increase the fitness of E. coli strains by mediating major transpositions in a genome, generating extensive genome rearrangements, and thereby increasing mutation rates and thus facilitating genome evolution.53, 54 The two insertions observed, IS1 and IS5, have both been well-studied in natural isolates of E. coli (the ECOR collection) with respect to their distribution and abundance.55 IS1 and IS5 are reported to be poorly regulated transposons and an increase in their copy number is correlated with a moderate to strong decrease in fitness.56 On the other hand, the IS1 element is also reported to increase mutational supply57 and occasionally generate variants with especially large phenotypic effects, such as host SOS response.58
Overall, the changes appear to be associated primarily with interaction with the environment. In other words, the cells are likely learning how to best interact with the low shear environment presented by the HARV. Thus, we see changes that are associated with transport, outer membrane interactions, and stress response. However, a 1000G non-LSMMG control is not available. Thus, the unadapted lac plus strain was used as the control for comparison of the sequencing results. Future studies are therefore needed to assess whether the mutations observed were specifically induced by exposure to the modeled microgravity environment.
Although adaptation to the LSMMG environment likely occurred, this did not impact antibiotic sensitivity. Previous experiments examined bacterial drug resistance in space flight samples. In these studies, a significant increase in the minimum inhibitory concentration levels of a few antibiotics was seen when tested against E. coli isolated from cosmonauts.59,60,61 However, bacteria, grown in culture for 4 months on Space Station Mir typically became less resistant to antibiotics.14 The present results suggest that antibiotic resistance strains likely did not develop as the result of adaptation to the low shear microgravity environment. Instead, the observed resistance to antibiotics seen in some earlier studies may reflect prior exposure of the natural microflora of the space station or the cosmonauts themselves.
Complex dynamics are often involved in long-term evolution of genomes.62, 63 Thus, 1000 generations is exceedingly unlikely to represent the end of adaptation. It can be expected that organisms on the space station or other long term missions would continue adapting indefinitely as has been observed in long-term studies of E. coli.62, 64,65,66,67,68,69 This process would possibly be accelerated by the elevated radiation levels experienced in space. During such true long-term adaptation, neutral mutations would likely become more common. At the very least, the initial focus of the adaptation would likely continue to be on the direct interaction with the low shear/microgravity environment and hence the types of changes seen here, rather than unrelated changes. However, the observation that there is enduring adaptation to the LSMMG environment supports the concern that organisms exposed to microgravity may as a by-product evolve undesirable properties such as predisposition to a growth stage that facilitates avoidance of the immune system.17
Monitoring microbial response to real time space conditions over extended periods is constrained by operational difficulty as well as costs, and thus, simulated microgravity offers a cheaper prototype analog of space conditions. Whether the results seen here reflect peculiarities of the HARV as a model system or an actual adaptation to microgravity is uncertain and would be best resolved by extended studies on the International Space Station. It is, however, reassuring that despite ongoing genomic adaptation to the LSMMG environment, increased antibiotic resistance was not observed.
Methods
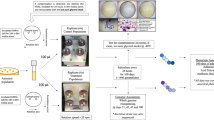
An overview of the experimental procedures is provided as Figs. 3 and 4.
Cell culture
An isogenic pair of E. coli strains, namely NCM520 (designated genotype F−, Δ(lacA-lacZ)880(FRT), λ −, rph-1; CGSC # 8252) a lac minus strain derived from MG1655 in which the entire lac operon was deleted and the well characterized MG1655 (a lac plus strain; CGSC # 6300)70 were obtained from the E. coli Genetic Stock Center at Yale University.71 The MG1655 strain was selected for analysis because its genome has been completely sequenced and is well annotated.72 The two strains can be easily differentiated on MacConkey agar medium by colony color.73 Both strains were immediately and separately inoculated into Luria Broth (LB) medium74 and incubated at 37 °C, 150 rpm on a rotary shaker. All shaker culture growth studies were conducted at these standard conditions. At saturation, the cultures were maintained as glycerol stocks at −70 °C for later use.
Simulated microgravity/LSMMG
In order to obtain simulated microgravity, cells were grown in HARVs (HARVs) (Synthecon Inc., Houston, TX) at 37 °C in 50 ml of LB medium. The LSMMG environment was obtained by HARV rotation at 25 rpm on a horizontal axis.6, 7 The aerobic environment is maintained by gas exchange which occurs by perfusion through a permeable membrane. Cell growth was monitored spectrophotometrically (DU530 spectrophotometer; Beckman Instruments, Fullerton, CA) at 600 nm by removing 500-µl samples every 20–45 min without interrupting culture agitation; no more than 5% of the culture was removed prior to harvesting of cells for DNA isolation.
Preparation of HARV vessels
The HARV-vessels were thoroughly rinsed first under running water. The entire set of different parts of the HARV unit was placed in a tub, and were treated with 25% bleach solution and left standing for 10–15 min. The bleach-treated parts were then rinsed thoroughly under running water and loosely reassembled, wrapped in aluminum foil and autoclaved at 117 °C for 15 min. The sterile HARVs were allowed to cool under sterile conditions, the vessels completely reassembled and then used for growth studies (Fig. 3a).
Growth of the E. coli MG1655 lac plus strain over 1000 generations of LSMMG exposure
A single colony was picked from an overnight LB agar plate and inoculated into LB medium for growth in an autoclaved HARV. Initially, growth curves were obtained for the E. coli MG1655 lac plus strain in the HARV (Supplementary Fig. 5) and the number of generations were inferred to be approximately ten for each cycle, Each cycle of growth reached saturation over 8–12 h, and the cells were then removed and treated as follows. One aliquot of 500 µl was preserved as glycerol stock. A second aliquot of 10 µl was diluted appropriately and plated on LB agar plates for growth in an incubator at 37 °C to rule out contamination. If contamination was detected, the growth was reverted back to the previous stage/cycle to ensure contamination free adaptation. A third aliquot of 500 µl was used to subculture cells into LB medium for growth in a sterile HARV. This procedure was continued for approximately 100 growth cycles. Each cycle represented approximately ten generations of growth resulting in approximately 1000 generations of growth for each original culture (Fig. 3b). Plating out was done as follows: the cell cultures were serial diluted and 50 µl of the same added to the center of the plate. Sterile glass beads numbering 6–7 were added to the plate and the plate was placed on a flat surface and shaken to spread the 50 µl of the diluted culture.
Competition growth studies of the isogenic pair
When grown on MacConkey agar, the MG1655 lac plus strain produces pink colonies and the NCM520 lac minus strain produces colorless colonies. A single colony of each strain was picked from MacConkey agar plates streaked with the strain(s) overnight from the glycerol stocks and inoculated separately in LB and set for growth. At growth saturation (O.D of 1.25 at A600), 5 μl each of the two strains were combined and inoculated into 50 ml of LB for growth in five sets of flasks. The growth of the strains was quantified by plating them out at saturation on MacConkey agar. The cultures were allowed to grow until they reached the end of the log phase, which based on growth studies (Supplementary Fig. 5) is equivalent to ten generations. The culture was only then plated out. The plates were placed in a 37 °C incubation chamber overnight. The ratio of the lac plus and lac minus colonies was determined by counting the numbers of colonies of each color on each plate. Because the number of colonies per plate is highly variable, we calculated the ratio based on the total counts from all the plates. Alternatively, the ratio for each plate was determined and these could then be averaged.
Competition growth studies of the isogenic pair under microgravity conditions
The lac plus strain evolved over 1000 generations (1000G) under microgravity (LSMMG) in HARV were directly inoculated into LB medium and set for growth under appropriate LSMMG conditions for several cycles to reactivate growth. The reactivated lac plus strain was co-inoculated with an equal amount of the lac minus strain grown in LB medium in a flask at 37 °C overnight. Care was taken to ensure both cultures had grown to similar OD levels, in order to obtain equal amounts of cells. LB medium was inoculated with this mixed population grown under LSMMG conditions in the HARV. The ratio of the lac plus strain (pink colonies) to the lac minus strain (white colonies) was again determined.
Competition growth studies of the unevolved isogenic pair under microgravity conditions
The above was repeated, but instead of the 1000G lac plus strain, the unevolved E. coli MG1655 lac plus (WT) strain grown in LB under shaker flask conditions was co-inoculated with an equal amount of the lac minus strain that was grown in LB medium in a flask at 37 °C overnight.
Adaptation ‘erasure’ experiment
The LSMMG 1000G-adapted strain and the lac minus strain were grown in LB medium in a flask under rotary conditions at 37 °C overnight. The two strains were co-inoculated and the competition assay was repeated under LSMMG at 37 °C, and the ratio of lac plus and lac minus was calculated (Fig. 4). This procedure was repeated after exposing the 1000G LSMMG adapted strain to shake flask conditions for two and three cycles (one cycle referring to ten generations of growth), respectively to determine if repeated exposure of the lac plus strain to shake flask conditions would erase the adaptation.
VITEK analysis for measuring antibiotic sensitivity
The antibiotic sensitivity of the E. coli strain(s) exposed to different conditions were assayed using the VITEK® 2 Compact instruments and VITEK® 2 PC software (BioMérieux, Inc., Hazelwood, MO). The antibiotics used in the VITEK analysis included ampicillin, amoxicillin/clavulanic acid, piperacillin/tazobactam, cefalotin, cefazolin, cefuroxime, cefuroxime axetil, cefoxitin, cefpodoxime, ceftazidime, ceftriaxone, cefepime, gentamicin, tobramycin, ciprofloxacin, levofloxacin, norfloxacin, tetracycline, nitrofurantoin, trimethoprim/sulfamethoxazole.
Genome sequencing
The genome of the 1000G strain was examined for mutations using the Illumina Genome Analyzer System (Illumina®, Hayward, CA). Briefly, the genomic DNA sample preparation kit was used to build DNA libraries for single-read sequencing. Random size-selected fragments of DNA were created, polished, and prepared for the addition of unique adapters to each end of the DNA fragments. Standard Cluster Generation Kit reagents were used to bind DNA library samples to complementary adapter oligomers that were next grafted on the flow cell surface. This allowed amplification of the attached DNA fragments to create clonal clusters of roughly 1000 copies each. An Illumina Sequencing Kit was used to determine the DNA sequence of Clusters on the flow cell. All the sequencing steps were performed following the manufacturer’s specifications (Illumina®, Hayward, CA).
Analysis of genome sequencing data
The individual reads were assembled de novo using Velvet.75 They were next aligned against the wild type E. coli MG1655 (CGSC7740) genome sequence using Mosaik76 and breseq.77 Mutation discovery was performed with MUMmer’s nucmer tool78 and gigaBayes.79 Each candidate mutation was visually checked and confirmed using Samtools’ tview80 to visualize the Mosaik alignment. As a result, a list of mutations within the 1000G HARV genome was created and further analyzed in order to evaluate the likely impact of the mutation at the protein level. Mutations found to be located within a gene were translated to assess any change at the amino acid level as compared to the original protein sequence using BioEdit (Hall, Ibis Biosciences, Carlsbad, CA). Online protein domain annotation tools such as SMART (version 7) or InterProScan were used to predict the location of the mutated amino acid position in the protein’s conserved domain(s)/motif(s).
References
Aponte, V. M., Finch, D. S. & Klaus, D. M. Considerations for non-invasive in-flight monitoring of astronaut immune status with potential use of MEMS and NEMS devices. Life Sci. 79, 1317–1333 (2006).
Crucian, B. E., Stowe, R. P., Pierson, D. L. & Sams, C. F. Immune system dysregulation following short- vs long-duration spaceflight. Aviat. Space Environ. Med. 79, 835–843 (2008).
Gueguinou, N. et al. Could spaceflight-associated immune system weakening preclude the expansion of human presence beyond Earth’s orbit? J. Leukoc. Biol. 86, 1027–1038 (2009).
Blaber, E., Marcal, H. & Burns, B. P. Bioastronautics: the influence of microgravity on astronaut health. Astrobiology 10, 463–473 (2010).
Rosenzweig, J. A., Ahmed, S., Eunson, J. Jr. & Chopra, A. K. Low-shear force associated with modeled microgravity and spaceflight does not similarly impact the virulence of notable bacterial pathogens. Appl. Microbiol. Biotechnol. 98, 8797–8807 (2014).
Nickerson, C. A. et al. Microgravity as a novel environmental signal affecting Salmonella enterica serovar Typhimurium virulence. Infect. Immun. 68, 3147–3152 (2000).
Wilson, J. W. et al. Microarray analysis identifies Salmonella genes belonging to the low-shear modeled microgravity regulon. Proc. Natl. Acad. Sci. USA 99, 13807–13812 (2002).
Wilson, J. W. et al. Media ion composition controls regulatory and virulence response of Salmonella in spaceflight. PLoS One 3, e3923 (2008).
Wilson, J. W. et al. Space flight alters bacterial gene expression and virulence and reveals a role for global regulator Hfq. Proc. Natl. Acad. Sci. USA 104, 16299–16304 (2007).
Rosenzweig, J. A. & Chopra, A. K. The effect of low shear force on the virulence potential of Yersinia pestis: new aspects that space-like growth conditions and the final frontier can teach us about a formidable pathogen. Front. Cell Infect. Microbiol. 2, 107 (2012).
Crabbe, A. et al. Spaceflight enhances cell aggregation and random budding in Candida albicans. PLoS One 8, e80677 (2013).
Crabbe, A. et al. Transcriptional and proteomic responses of Pseudomonas aeruginosa PAO1 to spaceflight conditions involve Hfq regulation and reveal a role for oxygen. Appl. Environ. Microbiol. 77, 1221–1230 (2011).
Huang, B., Liu, N., Rong, X., Ruan, J. & Huang, Y. Effects of simulated microgravity and spaceflight on morphological differentiation and secondary metabolism of Streptomyces coelicolor A3(2). Appl. Microbiol. Biotechnol. 99, 4409–4422 (2015).
Juergensmeyer, M. A., Juergensmeyer, E. A. & Guikema, J. A. Long-term exposure to spaceflight conditions affects bacterial response to antibiotics. Microgravity Sci. Technol. 12, 41–47 (1999).
Kacena, M. A., Manfredi, B. & Todd, P. Effects of space flight and mixing on bacterial growth in low volume cultures. Microgravity Sci. Technol. 12, 74–77 (1999).
Kim, W. et al. Effect of spaceflight on Pseudomonas aeruginosa final cell density is modulated by nutrient and oxygen availability. BMC Microbiol. 13, 241 (2013).
Kim, W. et al. Spaceflight promotes biofilm formation by Pseudomonas aeruginosa. PLoS One 8, e62437 (2013).
Altenburg, S. D., Nielsen-Preiss, S. M. & Hyman, L. E. Increased filamentous growth of Candida albicans in simulated microgravity. Genom. Proteom. Bioinform. 6, 42–50 (2008).
Arunasri, K. et al. Effect of simulated microgravity on E. coli K12 MG1655 growth and gene expression. PLoS One 8, e57860 (2013).
Baker, P. W., Meyer, M. L. & Leff, L. G. Escherichia coli growth under modeled reduced gravity. Microgravity Sci. Technol. 15, 39–44 (2004).
Chopra, V. et al. Alterations in the virulence potential of enteric pathogens and bacterial-host cell interactions under simulated microgravity conditions. J. Toxicol. Environ. Health A 69, 1345–1370 (2006).
Ciferri, O., Tiboni, O., Di Pasquale, G., Orlandoni, A. M. & Marchesi, M. L. Effects of microgravity on genetic recombination in Escherichia coli. Naturwissenschaften 73, 418–421 (1986).
Demain, A. L. & Fang, A. Secondary metabolism in simulated microgravity. Chem. Rec. 1, 333–346 (2001).
Fang, A., Pierson, D. L., Koenig, D. W., Mishra, S. K. & Demain, A. L. Effect of simulated microgravity and shear stress on microcin B17 production by Escherichia coli and on its excretion into the medium. Appl. Environ. Microbiol. 63, 4090–4092 (1997).
Fang, A., Pierson, D. L., Mishra, S. K., Koenig, D. W. & Demain, A. L. Secondary metabolism in simulated microgravity: beta-lactam production by Streptomyces clavuligerus. J. Ind. Microbiol. Biotechnol. 18, 22–25 (1997).
Gao, H., Liu, Z. & Zhang, L. Secondary metabolism in simulated microgravity and space flight. Protein Cell 2, 858–861 (2011).
Gao, Q., Fang, A., Pierson, D. L., Mishra, S. K. & Demain, A. L. Shear stress enhances microcin B17 production in a rotating wall bioreactor, but ethanol stress does not. Appl. Microbiol. Biotechnol. 56, 384–387 (2001).
Kacena, M. A., Leonard, P. E., Todd, P. & Luttges, M. W. Low gravity and inertial effects on the growth of E. coli and B. subtilis in semi-solid media. Aviat. Space Environ. Med. 68, 1104–1108 (1997).
Kim, H. W. & Rhee, M. S. Influence of low-shear modeled microgravity on heat resistance, membrane fatty acid composition, and heat stress-related gene expression in Escherichia coli O157:H7 ATCC 35150, ATCC 43889, ATCC 43890, and ATCC 43895. Appl. Environ. Microbiol. 82, 2893–2901 (2016).
Lawal, A. et al. The effects of modeled microgravity on growth kinetics, antibiotic susceptibility, cold growth, and the virulence potential of a Yersinia pestis ymoA-deficient mutant and its isogenic parental strain. Astrobiology 13, 821–832 (2013).
Pacello, F., Rotilio, G. & Battistoni, A. Low-Shear modeled microgravity enhances salmonella enterica resistance to hydrogen peroxide through a mechanism involving KatG and KatN. Open Microbiol. J. 6, 53–64 (2012).
Searles, S. C., Woolley, C. M., Petersen, R. A., Hyman, L. E. & Nielsen-Preiss, S. M. Modeled microgravity increases filamentation, biofilm formation, phenotypic switching, and antimicrobial resistance in Candida albicans. Astrobiology 11, 825–836 (2011).
Soni, A. et al. Conservation of the low-shear modeled microgravity response in Enterobacteriaceae and analysis of the trp genes in this response. Open Microbiol. J. 8, 51–58 (2014).
Tucker, D. L. et al. Characterization of Escherichia coli MG1655 grown in a low-shear modeled microgravity environment. BMC Microbiol. 7, 15 (2007).
Wilson, J. W. et al. Low-Shear modeled microgravity alters the Salmonella enterica serovar typhimurium stress response in an RpoS-independent manner. Appl. Environ. Microbiol. 68, 5408–5416 (2002).
Xu, B. et al. Simulated microgravity affects ciprofloxacin susceptibility and expression of acrAB-tolC genes in E. coli ATCC25922. Int. J. Clin. Exp. Pathol. 8, 7945–7952 (2015).
Orsini, S. S., Lewis, A. M. & Rice, K. C. Investigation of simulated microgravity effects on Streptococcus mutans physiology and global gene expression. NPJ Microgravity 3, 4 (2017).
Abshire, C. F. et al. Exposure of Mycobacterium marinum to low-shear modeled microgravity: effect on growth, the transcriptome and survival under stress. NPJ Microgravity 2, 16038 (2016).
Justice, S. S. et al. Periplasmic peptidyl prolyl cis-trans isomerases are not essential for viability, but SurA is required for pilus biogenesis in Escherichia coli. J. Bacteriol. 187, 7680–7686 (2005).
Palomino, C., Marin, E. & Fernandez, L. A. The fimbrial usher FimD follows the SurA-BamB pathway for its assembly in the outer membrane of Escherichia coli. J. Bacteriol. 193, 5222–5230 (2011).
Rego, A. T., Chandran, V. & Waksman, G. Two-step and one-step secretion mechanisms in gram-negative bacteria: contrasting the type IV secretion system and the chaperone-usher pathway of pilus biogenesis. Biochem. J. 425, 475–488 (2010).
Waksman, G. & Hultgren, S. J. Structural biology of the chaperone-usher pathway of pilus biogenesis. Nat. Rev. Microbiol. 7, 765–774 (2009).
Gerlach, R. G. & Hensel, M. Protein secretion systems and adhesins: the molecular armory of Gram-negative pathogens. Int. J. Med. Microbiol. 297, 401–415 (2007).
Kline, K. A., Falker, S., Dahlberg, S., Normark, S. & Henriques-Normark, B. Bacterial adhesins in host-microbe interactions. Cell Host Microbe 5, 580–592 (2009).
Zhang, M. et al. A genetically incorporated crosslinker reveals chaperone cooperation in acid resistance. Nat. Chem. Biol. 7, 671–677 (2011).
Choudhury, D. et al. X-ray structure of the FimC-FimH chaperone-adhesin complex from uropathogenic Escherichia coli. Science 285, 1061–1066 (1999).
Hung, C. S. et al. Structural basis of tropism of Escherichia coli to the bladder during urinary tract infection. Mol. Microbiol. 44, 903–915 (2002).
Nilsson, L. M. et al. The cysteine bond in the Escherichia coli FimH adhesin is critical for adhesion under flow conditions. Mol. Microbiol. 65, 1158–1169 (2007).
Fink, R., Oder, M., Rangus, D., Raspor, P. & Bohinc, K. Microbial adhesion capacity. Influence of shear and temperature stress. Int. J. Environ. Health Res. 25, 656–669 (2015).
Munera, D., Palomino, C. & Fernandez, L. A. Specific residues in the N-terminal domain of FimH stimulate type 1 fimbriae assembly in Escherichia coli following the initial binding of the adhesin to FimD usher. Mol. Microbiol. 69, 911–925 (2008).
Osteras, M., Boncompagni, E., Vincent, N., Poggi, M. C. & Le Rudulier, D. Presence of a gene encoding choline sulfatase in Sinorhizobium meliloti bet operon: choline-O-sulfate is metabolized into glycine betaine. Proc. Natl. Acad. Sci. USA 95, 11394–11399 (1998).
Lamark, T., Rokenes, T. P., McDougall, J. & Strom, A. R. The complex bet promoters of Escherichia coli: regulation by oxygen (ArcA), choline (BetI), and osmotic stress. J. Bacteriol. 178, 1655–1662 (1996).
Biel, S. W. & Hartl, D. L. Evolution of transposons: natural selection for Tn5 in Escherichia coli K12. Genetics 103, 581–592 (1983).
Chao, L., Vargas, C., Spear, B. B. & Cox, E. C. Transposable elements as mutator genes in evolution. Nature 303, 633–635 (1983).
Sawyer, S. A. et al. Distribution and abundance of insertion sequences among natural isolates of Escherichia coli. Genetics 115, 51–63 (1987).
Mahillon, J. & Chandler, M. Insertion sequences. Microbiol. Mol. Biol. R. 62, 725–774 (1998).
Feher, T. et al. Competition between transposable elements and mutator genes in bacteria. Mol. Biol. Evol. 29, 3153–3159, doi:10.1093/molbev/mss122 (2012).
Lane, D., Cavaille, J. & Chandler, M. Induction of the SOS response by IS1 transposase. J. Mol. Biol. 242, 339–350 (1994).
Liu, C. The theory and application of space microbiology: China’s experiences in space experiments and beyond. Environ. Microbiol. doi:10.1111/1462-2920.13472 (2016).
Lapchine, L. et al. Antibiotic activity in space. Drugs Exp. Clin. Res. 12, 933–938 (1986).
Ilyin, V. K. Microbiological status of cosmonauts during orbital spaceflights on Salyut and Mir orbital stations. Acta Astronaut. 56, 839–850 (2005).
Tenaillon, O. et al. Tempo and mode of genome evolution in a 50,000-generation experiment. Nature. doi:10.1038/nature18959 (2016).
Lenski, R. E. & Travisano, M. Dynamics of adaptation and diversification: a 10,000-generation experiment with bacterial populations. Proc. Natl. Acad. Sci. USA 91, 6808–6814 (1994).
Lenski, R. E. et al. Sustained fitness gains and variability in fitness trajectories in the long-term evolution experiment with Escherichia coli. Proc. Biol. Sci. 282, 20152292 (2015).
Turner, C. B., Blount, Z. D. & Lenski, R. E. Replaying evolution to test the cause of extinction of one ecotype in an experimentally evolved population. PLoS One 10, e0142050 (2015).
Maddamsetti, R. et al. Synonymous genetic variation in natural isolates of escherichia coli does not predict where synonymous substitutions occur in a long-term experiment. Mol. Biol. Evol. 32, 2897–2904 (2015).
Wiser, M. J. & Lenski, R. E. A comparison of methods to measure fitness in escherichia coli. PLoS One 10, e0126210 (2015).
Raeside, C. et al. Large chromosomal rearrangements during a long-term evolution experiment with Escherichia coli. MBio 5, e01377–01314 (2014).
Barrick, J. E. & Lenski, R. E. Genome dynamics during experimental evolution. Nat. Rev. Genet. 14, 827–839 (2013).
Jensen, K. F. The Escherichia coli K-12 “wild types” W3110 and MG1655 have an rph frameshift mutation that leads to pyrimidine starvation due to low pyrE expression levels. J. Bacteriol. 175, 3401–3407 (1993).
Berlyn, M. B. & Letovsky, S. Genome-related datasets within the E. coli Genetic Stock Center database. Nucleic Acids Res. 20, 6143–6151 (1992).
Blattner, F. R. et al. The complete genome sequence of Escherichia coli K-12. Science 277, 1453–1462 (1997).
Macconkey, A. Lactose-fermenting bacteria in faeces. J. Hyg. (Lond) 5, 333–379 (1905).
Neidhardt, F. C., Bloch, P. L. & Smith, D. F. Culture medium for enterobacteria. J. Bacteriol. 119, 736–747 (1974).
Zerbino, D. R. & Birney, E. Velvet: algorithms for de novo short read assembly using de Bruijn graphs. Genome Res. 18, 821–829 (2008).
Smith, D. R. et al. Rapid whole-genome mutational profiling using next-generation sequencing technologies. Genome Res. 18, 1638–1642 (2008).
Barrick, J. E. et al. Identifying structural variation in haploid microbial genomes from short-read resequencing data using breseq. BMC Genomics 15, 1039 (2014).
Marth, G. T. et al. A general approach to single-nucleotide polymorphism discovery. Nat. Genet. 23, 452–456 (1999).
Kurtz, S. et al. Versatile and open software for comparing large genomes. Genome Biol. 5, R12 (2004).
Li, H. et al. The sequence alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Acknowledgements
This work was made possible by the loan of HARV vessels to the Fox group by NASA JSC. Funding was provided by the Institute of Space Systems Operations at the University of Houston and the NASA URC at Texas Southern University under Cooperative Agreement NNX08B4A47A.
Author information
Authors and Affiliations
Contributions
M.R.T. and F.K. each conducted a major portion of the experimental work and participated in writing the finished manuscript. M.R.T. performed the competition assays, maintained the cultures, conducted the statistical analysis and prepared Figs. 3 and 4. V.G.S. prepared samples for sequencing and with Q.T. and M.R.T. analyzed the resulting genome sequence data needed to prepare figures and tables. Q.T. prepared Fig. 2. R.J.B. conducted the VITEK experiments. C.M.O. oversaw maintenance and use of the HARVs and assisted in the analysis of the VITEK experiments. D.L.P. participated in planning experiments and evaluating VITEK results. G.E.F. and D.L.P. conceived the study, G.E.F. provided day-to-day oversight. F.K., M.R.T., C.M.O., and G.E.F. wrote the paper; all authors discussed the results in the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tirumalai, M.R., Karouia, F., Tran, Q. et al. The adaptation of Escherichia coli cells grown in simulated microgravity for an extended period is both phenotypic and genomic. npj Microgravity 3, 15 (2017). https://doi.org/10.1038/s41526-017-0020-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41526-017-0020-1