Abstract
SMAD6 encodes an intracellular inhibitor of the bone morphogenetic protein (BMP) signalling pathway. Until now, SMAD6-deficiency has been associated with three distinctive human congenital conditions, i.e., congenital heart diseases, including left ventricular obstruction and conotruncal defects, craniosynostosis and radioulnar synostosis. Intriguingly, a similar spectrum of heterozygous loss-of-function variants has been reported to cause these clinically distinct disorders without a genotype–phenotype correlation. Even identical nucleotide changes have been described in patients with either a cardiovascular phenotype, craniosynostosis or radioulnar synostosis. These findings suggest that the primary pathogenic variant alone cannot explain the resultant patient phenotype. In this review, we summarise clinical and (patho)genetic (dis)similarities between these three SMAD6-related conditions, compare published Madh6 mouse models, in which the importance and impact of the genetic background with respect to the observed phenotype is highlighted, and elaborate on the cellular key mechanisms orchestrated by SMAD6 in the development of these three discrete inherited disorders. In addition, we discuss future research needed to elucidate the pathogenetic mechanisms underlying these diseases in order to improve their molecular diagnosis, advance therapeutic strategies and facilitate counselling of patients and their families.
Similar content being viewed by others
The protein SMAD6, encoded by SMAD6 (OMIM: 602931), belongs to the SMAD family of proteins involved in the bone morphogenetic proteins (BMP) signalling cascade. Even though these molecules were initially discovered for their ability to induce bone formation, it is now clear that BMPs are important in the embryogenesis and development of many organ systems, as well as in maintenance of adult tissue homoeostasis. SMAD6 is an intracellular inhibitor of, predominantly, the BMP signalling pathway, yet it cross-talks with the closely related transforming growth factor-β (TGF-β) signalling pathway1,2.
Over the past 10 years, genetic variants in SMAD6 were demonstrated to impinge on the risk of human genetic disorders3,4,5,6,7,8,9,10,11,12,13 such as cardiovascular diseases, including congenital heart defects (CHD), craniosynostosis (CRS) and radioulnar synostosis (RUS). CHD is among the most common birth defects, affecting 6–13:1000 live-born infants14,15,16. In association with SMAD6-deficiency, it encompasses a range of cardiac and outflow tract abnormalities. Complex lesions consisting of multiple defects are often severe, even critical, for which treatment with advanced surgery for definitive correction of malformations or (palliative) medication is imperative17. In addition, adult patients with a sole congenital aortic valve defect associate with more late-onset vascular complications like a pathological widening of the thoracic aorta (~thoracic aortic aneurysm (TAA))18. TAAs are also life-threatening as they are (1) clinically silent19, (2) entail a high risk for acute dissection and/or rupture (mortality rates ≥70%)20, and (3) no therapy currently exists that can stop TAA development or progression21. CRS, which is a skull defect afflicting 1:2100–2500 live births22,23, is a second SMAD6-related disease. Surgical correction is frequently necessary to prevent complications24 such as developmental delay, facial abnormality, sensory, respiratory and neurological dysfunction, anomalies affecting the eye, and psychological disturbances25. Finally, congenital RUS, also referred to as fused forearm bones, is a rare condition with ~500 cases reported in literature9,13,26. This malformation, usually diagnosed before the age of 5 years, is not life-threatening, but corrective surgery and/or medication to control pain might, in some cases, improve the quality of life13.
The therapeutic strategies for SMAD6 mutation-positive patients mainly focus on disease monitoring in order to define the appropriate time for intervention, medication to control pain, and surgical repair19,24,27,28,29,30. Even though surgery is effective, it is associated with risks, requires early detection of at-risk patients, only provides relief late in the disease course, and does not target the underlying driver(s) of the disease. Hence, there is a need for a better (molecular) understanding for early diagnosis, and to empower new therapies to prevent disease progression. With this review, we provide a comprehensive overview on SMAD6-deficiency in human genetic disorders by summarising the clinical, (patho)genetic and cellular (dis)similarities observed in human and mouse models. We conclude with future directions of research needed to improve patient management based on the underlying SMAD6-related molecular disease signature.
Clinical phenotype of patients with SMAD6-deficiency
The clinical presentation of heterozygous SMAD6 variant-positive patients is extremely heterogeneous as illustrated by the different affected organ systems, the varying degree of severity, and the range of associated complications. Table 1 summarises the clinical phenotype of probands with disease-causative SMAD6 variants. All disease-related clinical definitions are summarised in Table 2.
Cardiovascular diseases
The cardiovascular phenotype (cases, N = 31) (probands, N = 28, Table 1) include left ventricular outflow tract defects (N = 21/28, 75%)4,5,7,8,10, conotruncal defects (N = 4/28, 14%)5, and defects defined as “others” (N = 3/28, 11%)5 as they cannot be categorised into the traditional subgroups that arise from disruption of shared embryonic processes. Left ventricular outflow tract defect refers to hypoplastic left heart syndrome (HLHS) (N = 1/21, 5%), coarctation of the aorta (CoA) (N = 2/21, 10%), and bicuspid aortic valve (BAV), which is associated with congenital CoA and/or late-onset TAA (N = 17/21, 81%) in all patients, except for one toddler with isolated BAV (N = 1/21, 5%; 1.5 years old). Conotruncal defects include Tetralogy of Fallot (N = 3/4, 75%) and D-loop transposition of the Great Arteries (N = 1/4, 25%). The remaining three patients presented with either a vascular ring, partially anomalous pulmonary veins combined with sinus venosus septal defect, or stenotic pulmonary valve and stenotic left main coronary artery accompanied with dilated cardiomyopathy.
Craniosynostosis
The clinical outcome of CRS (cases, N = 49) (probands, N = 43, Table 1)3,11,12 involves syndromic (N = 7/43, 16%) and non-syndromic presentations (N = 36/43, 84%) in which single and multiple fusion events of almost all sutures have been identified. Most common presentation was metopic synostosis (N = 27/43, 63%), followed by sagittal synostosis (N = 9/43, 21%), right unicoronal synostosis (N = 2/43, 5%), combined metopic and sagittal synostosis (N = 2/43, 5%), combined sagittal and bicoronal synostosis (N = 2/46, 4%), and combined sagittal and left unicoronal synostosis (N = 1/43, 2%). Remarkably, raised intracranial pressure following cranial reconstruction, which is usually a rather infrequent complication in simple synostosis of midline sutures, should be specifically monitored in SMAD6 variant-positive patients3. Other recurrent brain or skull anomalies in (non-)syndromic subjects comprise ventriculomegaly and absent corpus callosum, macrocephaly, and mild microcephaly, and mild-to-moderate neurodevelopmental delay (consisting of speech, educational and global delay)3,11,12. More subtle learning difficulties were observed in 36% of the non-syndromic patients (N = 14)11, while gross and fine motor delays were only observed occasionally3,11. In syndromic cases3, cardiac defects were common (N = 5/7, 71%), but seem to have another pattern which is further discussed in the following section on the clinical overlap.
Congenital radioulnar synostosis
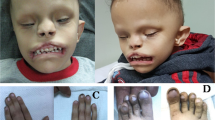
Patients with RUS9,13 (cases, N = 93) (probands, N = 77, Table 1) are most frequently characterised by bilateral RUS (69%; isolated (N = 42/61), familial (N = 22/32)), followed by unilateral left-sided RUS in sporadic patients (N = 15/42, 36%) versus right-sided RUS (N = 4/42, 10%), while no susceptibility for left- or right-sided RUS was observed in families (N = 5/32, 16%). Affected individuals within a single pedigree can show both bilateral and unilateral RUS. No syndromic cases have been reported thus far, yet some subordinate clinical findings have been described in 14 families9,13: three families presented with axial skeletal deformities (including cervical fusion, rib malformation, caudal vertebrae dysplasia and vertebral malformations), two families had polydactyly (pre- and pro-axial type), five families exhibited CHD (encompassing patent ductus arteriosus, mitral/tricuspid/aortic regurgitation, atrial septal defect, BAV, left ventricular hypertrophy and mitral/pulmonary valve insufficiency), and four families showed skull-related defects (containing frontal bossing, plagiocephaly and premature closure of anterior fontanel, but no unequivocal description of CRS). Remarkably, in six out of the 14 families a variant-positive SMAD6 carrier without RUS but with skeleton-, skull-, or CHD-related features9,13 was reported: two affected individuals from two families displayed cervical fusion or caudal vertebrae dysplasia, two patients from two families exhibited with premature closure of anterior fontanel, frontal bossing and plagiocephaly or solely plagiocephaly, one subject presented with polydactyly and, finally, one affected had patent ductus arteriosus together with mitral regurgitation.
Clinical overlap?
Studies have, in addition to the clinical indication for study enrolment, to some extent also assessed the presence of other SMAD6-related clinical associations. Patients with cardiovascular disease did not exhibit CRS and/or RUS. A child, from a consanguineous family harbouring a pathogenic homozygous SMAD6 variant, was reported to present with CoA, suspected tricuspid aortic valve, bilateral RUS, renal anomalies, facial dysmorphism and global development delay6. In view of consanguinity, homozygosity at other loci as an explanation for these multisystemic features cannot be excluded. CRS cases did not exhibit RUS, but five syndromic CRS cases presented with a CHD3 of which none seem to mimic the more severe conotruncal and outflow tract defects seen in the cardiovascular disease cohorts. For example, defects in three patients resolved spontaneously. One patient with atrioventricular septal defect required surgery at the age of three years, yet his variant-positive mother had a normal echocardiogram. The fifth patient had BAV (N = 1/46, 2%) with right bundle branch block. This observation exactly matches the epidemiological number of 2% for BAV in the general population though31. None of the seven extra screened asymptomatic parents of SMAD6 variant-positive children with CRS showed any evidence for BAV or TAA3. Finally, no BAV or TAA has been identified so far in the non-syndromic CRS cohort (personal communication with A. Wilkie, Oxford). Hence, no clinical overlap of a variant-positive SMAD6 carrier with cardiovascular disease or CRS with any abnormality affecting the other organ systems has been observed to date.
Finally, the phenotypic picture in 14 families with RUS is more complicated as both CHD as well as skull and skeletal abnormalities have been observed occasionally (N = 12/93, 13%)9,13. Although based on their nature and incidence, we cannot rule out an alternative cause for some abnormalities (e.g., valve insufficiency, left ventricular hypertrophy and rib/vertebral malformation), the occurrence of skeletal- (N = 4/93, 4%), skull- (N = 9/93, 10%), or CHD-related (N = 3/93, 3%) abnormalities in families with RUS does hint to some clinical overlap. For example, two variant-positive SMAD6 carriers from two families without RUS presented with axial skeletal deformities, either cervical fusion or caudal vertebrae dysplasia. Extra skull features were observed in another five families, including frontal bossing (N = 4/93, 4%), plagiocephaly (N = 3/93, 3%), and premature fusion of the anterior fontanel (N = 2/93, 2%). Plagiocephaly and premature fusion of the anterior fontanel was reported in a variant-positive family member without RUS. And finally, three families had CHD too, namely patent ductus arteriosus (N = 1/93, 1%), atrial septal defect (N = 1/93, 1%), and BAV (N = 1/93, 1%).
Genetic (dis)similarities between SMAD6-related disorders
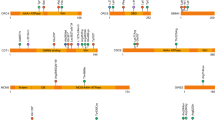
Intriguingly, similar, or even identical, heterozygous loss-of-function variants in SMAD6 cause these three distinct disorders (Fig. 1 and Supplementary Table 1)3,4,5,6,7,8,9,10,11,12,13. The variant spectrum includes rare truncating and missense variants locating in the functional MH1- and MH2-domain of the protein with no phenotypic correlation with respect to variant type nor location. Identical nucleotide changes (N = 6) have been described in patients with cardiovascular disease (N = 3), CRS (N = 5) or RUS (N = 10). Moreover, the phenotype within these families are, predominantly, restricted to one affected organ system. For example, the p.(Gly156Valfs*23) variant causes BAV-related aortopathy (N = 1), sagittal synostosis (N = 1), and non-syndromic RUS (N = 4), for which no clinical overlap has been documented except for frontal bossing in one family with left-sided RUS. Hence, the molecular finding cannot predict the clinical presentation of a patient, and, as such, it is likely that (a) factor(s) inherited together with the primary SMAD6 mutation drives the resultant patient phenotype. The latter seems likely as within one family concordance of the phenotype is frequently observed.
SMAD6 protein has several domains: MH1-domain (grey; inhibitory effect on signalling), PY motif (orange), PLDLDS motif (yellow), MH2-domain (grey; inhibitory effect on signalling) and L3-loop (blue; determines specificity for interaction with type I receptors)88,89. Of note: (1) all variants were called “pathogenic” in the original publications but there is different criteria for calling “pathogenicity” (e.g., by applying different allele frequency thresholds, and/or using specific functional tests); (2) identical protein changes (underlined) are described in patients with distinctive phenotypes.
Cardiovascular disease
The aetiology of CHD is multifactorial, involving genetic and environmental factors such as smoking, alcohol abuse and infection transmitted by the mother during pregnancy32. Familial studies have demonstrated that the CHD recurrence risk in family members of affected individuals depends on the type of lesion33. Pathogenic variants cause autosomal dominant, autosomal recessive, or X-linked traits with variable penetrance and clinical expressivity. About 132 definitive and strong candidate genes for CHD in numerous functional classes like chromatin modification, transcription factors and signal transduction, amongst others, have been reported. The predominant disease-causative effect is through loss-of-function34. To date, 50% of the patients remain molecularly undiagnosed though, and the yield is even lower in non-syndromic cases34. Interestingly, pathogenic SMAD6 variants have been shown to be enriched in isolated paediatric and adult CHD patients, in which most patients exhibited left ventricular outflow tract defects (Table 3). So far, patients with recessive variants do not seem to present with a more severe cardiovascular phenotype as compared to subjects harbouring heterozygous variants. However, this observation is based on only two cases, and no functional analyses have been performed6. A SMAD6 genetic uptake of 4.6% was reached in more severely affected BAV-related aortopathy patients, i.e., BAV patients who underwent surgical repair for aneurysmal disease before the age of 50, and with a positive family history for cardiovascular disease. The estimated penetrance for the disease was 82.4%. SMAD6 is the most important BAV/TAA gene identified thus far, as none of the approximately 30 definitive and candidate genes for BAV and/or TAA explain more than 1% of these patients7. The emerging BAV/TAA disease-related pathways include impaired cardiac transcription factor activity (e.g., GATA5)35,36, perturbed extracellular matrix homoeostasis (e.g., LOX)37, aberrant TGF-β (e.g., TGFBR1)19 and NOTCH (e.g., NOTCH1)38 signalling, deficiency of the vascular smooth muscle cell contractile apparatus (e.g., ACTA2)39, and altered endothelial cell function (e.g., ROBO4)40. Taken together, carrying a pathogenic SMAD6 might be insufficient to definitively cause cardiovascular disease in all cases, and, as such, more research is required to identify the missing information, and to understand how it contributes to disease.
A clinical and genetic association between BAV, HLHS, and CoA have already been thoroughly discussed in familial studies41,42, and some examples of monozygotic twins with discordant phenotypes, i.e., one has BAV while the other present with HLHS, have been described43,44. As SMAD6-deficiency results in a spectrum of, mainly, left ventricular outflow tract defects, one could hypothesise the existence of additional genetic hits in families. Particular emphasis might be given to ascertain essential cardiac transcription complexes, and to investigate the accessibility of these factors onto DNA in patient-derived material in order to reveal novel crucial clues on the pathogenesis of CHD disease.
Craniosynostosis
CRS is a heterogeneous disease influenced by mechanical and extrinsic forces as well as genetic components affecting the intrinsic properties of the suture45. In families, an autosomal dominant mode of transmission is mostly observed, but in about half of the cases a de novo variant is found. The genetic uptake is highest in syndromic cases, while isolated cases (i.e., 75% of all patients) largely remain molecularly undiagnosed45,46. Approximately one quarter of CRS cases harbour a disease-causative variant in one of the known genes, mostly in FGFR2, FGFR3, TCF12, ERF, EFNB1, or TWIST1, causing either a loss- or gain-of-function. These gene products are involved in signal transduction pathways like FGF signalling (FGR2, FGFR3), Eph/ephrin signalling (EFNB1) and ERK-MAPK activity (ERF) or they bind DNA to regulate gene expression (TWIST1, TCF12). As SMAD6 variants account for 5.8% of all (non-)syndromic patients with metopic synostosis, it became, by far, the largest monogenic contributor to metopic synostosis yet identified. Furthermore, SMAD6 variants seem less commonly associated with other types of suture fusion3, making it in particular relevant to screen patients with metopic synostosis for SMAD6 deficiency.
SMAD6-related CRS has been associated with reduced penetrance (overall penetrance, 16–24%)3,11,12. As such, a two-locus inheritance model for CRS (i.e., metopic, sagittal and combined metopic and sagittal) was proposed by Timberlake et al., in which near complete-penetrance was reached upon co-occurrence with a common BMP2 SNP risk allele (C) (rs1884302)11. Upon merger of datasets, this association still holds true, yet the initial signal has weakened due to non-replication in an independent cohort. One explanation might be the underrepresentation of the risk allele (frequency ~0.33, gnomAD: European non-Finnish) in non-penetrant SMAD6 variant harbouring individuals in the discovery studies, which was not observed in a third study3,11,12. Additionally, rs1884302 was found to strongly associate with sagittal synostosis47, and more recent data for metopic synostosis reveal no equivalent association for this SNP48. Extra work is necessary to explore on such relationship between SMAD6 variant-positive patients with sagittal synostosis, and a larger sample size is needed to dissect whether this interaction is truly digenic inheritance or is merely an additive effect of the GWAS signal, modifying the penetrance of SMAD6 pathogenic variants. Additional light was shed onto this digenic inheritance model by revealing the presence of this common SNP in SMAD6 mutation-positive patients with either BAV-related aortopathy (N = 4)7 or radioulnar synostosis (N = 7)13 but in the absence of any sign of CRS. Altogether, current data suggest that the pathogenic SMAD6 variant alone might be insufficient to definitively cause CRS in all cases, and it still remains to be further investigated what the extra hits, and what the underlying mechanisms are.
Radioulnar synostosis
Since the 70’s, congenital RUS is recognised as an inheritable disease segregating in an autosomal dominant manner49,50. In total, 10% of the RUS patients were identified with a monogenetic cause (e.g., NOG) or with aneuploidy syndromes, in which the syndromic subjects presented with additional abnormalities in the skeleton, heart, urinary tract, blood and males had extra X and Y chromosomes51. At present, SMAD6 deficiency is, by far, the most important known disease gene for non-syndromic RUS, as it explains 42% of familial cases and 16% of sporadic patients9,13. The penetrance of disease is incomplete, and has been reported around 20–25%9,13. Other genetic causes include two pathogenic variants in NOG, explaining less than 1% of the patients13. NOG encodes noggin, which is a major BMP antagonist. Dysregulation of BMP signalling due to NOG deficiency in mice showed interference with hedgehog signalling for BMP-induced interdigital cell death52, and for axial skeleton development53. The contribution of genetic variability in SMAD6 and NOG to syndromic RUS is yet unexplored. In sum, literature indicates that radioulnar synostosis is not exclusively caused by one pathogenic SMAD6 variant in all cases. Again, more investigation is needed to fill our gap in knowledge about the extra hits and underlying mechanisms.
Current challenges in SMAD6-related diagnosis and counselling
Patient management for SMAD6-related disorders is challenging as rare pathogenic loss-of-function variants associate with (1) reduced penetrance, (2) extreme variability in phenotypical expression, and (3) distinctive clinical entities without genotype–phenotype correlation, as outlined above. Hence, every single case should be discussed thoroughly in a multidisciplinary team based on phenotype, family history, inheritance pattern, and pathogenicity of the variant. Given the possibility of a devastating cardiovascular outcome, echocardiographic evaluation is currently indicated in a SMAD6 variant-positive proband, irrespective of the clinical indication for referral. A genetic test is best offered to family members of SMAD6 variant-positive patients with cardiovascular disease or RUS as some clinical overlap with the cardiovascular disease might exist. In contrast, variant-positive SMAD6 carriers in CRS cohorts are frequently unaffected making a genetic test uninformative. There is currently some preliminary evidence that phenotypes are quite consistent in a single family. Nevertheless, more insight is needed before we can abandon echocardiographic evaluation in relatives of SMAD6 variant-positive probands with CRS. Another counselling challenge is caused by the observation that the general population well-tolerates loss-of-function SMAD6 variants (pLI = 0, gnomAD v2.1.1), despite the overwhelming overrepresentation of such variants in disease cohorts as compared to this control population3,4,5,11,12,13. This is in particular challenging for CRS given the low penetrance of CRS in individuals heterozygous for pathogenic SMAD6 variants3,11.
Lastly, diagnostic and research laboratories also encounter difficulties for variant interpretation, in particular for missense variants. In this regard, Calpena et al.3 have provided a filtering strategy able to discriminate high-penetrant rare pathogenic missense variants, as proven in functional tests assessing protein stability and/or impaired BMP signalling activity. Even though very useful, this approach will not classify all type of variants (e.g., 5′ untranslated region), and current bio-informatic tools are not sufficient sensitive to assess variants with moderate effects, which are likely to explain, to some extent, the variability in expressivity and unpredictable penetrance. Nonetheless, implementation of flexible, preferably high-throughput, functional assays for variant interpretation, combined with further refinement of bio-informatic tools, is necessary to address this challenge.
Lessons from mouse models
Genetically modified mouse models have, with success, been used to interrogate the pathomechanisms underlying rare human disorders. At present, three mouse models lacking the murine orthologue of SMAD6, i.e., Madh6, have been studied (Table 4). The Madh6-mutant mice were produced by a LacZ/neomycin resistance cassette inserted into the 5´ terminus of the exon encoding the MH2-domain of Smad654. Each model is unique by its respective genetic background as all models were generated using embryonic stem cells created by Galvin et al.
In the model on a mixed 129/SvEv × BALB/cBy background54, homozygous animals exhibited hyperplasia of the cardiac valves, with the mitral and pulmonary valve being more extremely affected, septation defects, and lethality. The latter was observed due to an underrepresentation of homozygotes at the time of weaning. Surviving animals developed aortic ossification with notable cartilaginous metaplasia and trabeculation of the aortic media (from 6 weeks of age), decreased vasodilation and hypertension. Subsequent in-depth characterisation revealed an excess of mesenchymal cells in the cardiac valves in all homozygotes, while the following was only observed in a subset of the animals: (1) abnormal septation of the outflow tract, i.e., a severely narrowed ascending aorta and an enlarged pulmonary trunk or the reverse, (2) thrombotic lesions and ischaemia in lung, liver, and kidney, (3) subepicardial vascular malformations in the ventricular wall with loss of multiple smooth muscle cell layers in large vessels, and (4) thickening of the endocardium. Interestingly, a background sensitivity for the survival of homozygotes up to weaning was observed by comparing mouse models on different genetic backgrounds (i.e., 129/SvEv × BALB/cBy, 129/SvEv × C57Bl/6, inbred 129/SvEv), which corresponded to the severity of cardiac defects. Heterozygotes were not further studied, and no gender-specific analyses were performed. Even though similar anomalies were described in humans, it is still unanswered whether these mice also present BAV, aortic valve calcification, hypoplastic left heart and what the relative position of the aorta and pulmonary artery is. No gross non-cardiovascular anomalies were described, yet this has not been investigated into detail.
The next-studied knock-out mouse model55, on a C57BL/6J × BALB/c background, was generated to investigate the consequences of Smad6 loss during cartilage development. Homozygotes displayed craniofacial anomalies like a domed skull and shortened snout, but no defects in cranial sutures were found. Abnormalities in the skeleton were observed too, such as posterior transformation of cervical vertebrae (C7), flatter thoracic vertebral bodies, presence of bilateral ossification centres in lumbar vertebrae, and bifid sternebrae due to incomplete sternal band fusion. In addition, homozygotes were smaller in size, as confirmed by shorter appendicular bones, and stage-specific defects in endochondral bone formation were found like the delayed onset of hypertrophy at midgestation and expanded hypertophic zone at late gestation. Furthermore, significant embryonic and neonatal lethality was observed, as merely 5% of the progeny were homozygous and all live-born pups died within 24 h after birth due to an unspecified cause. Heterozygotes were not examined in this model, alike with other organ systems, especially no data on the cardiovascular system in the homozygotes were reported.
The last published model56 was generated on a CD1 background to elucidate the effects of Smad6 loss on blood vessel development. Wylie et al. reported on embryonic and postnatal lethality of homozygotes (all died by P2–6), in addition to regions of haemorrhages in skin and brown fat pads without any sign of hyperplastic valve thickening in these animals. The observed vessel phenotype was a consequence of disrupted endothelial cell junctions, thereby compromising vessel wall integrity. No in-depth experiments were performed on heterozygous animals, nor other organ systems were examined.
Altogether, this mouse knock-out data support a role for unique genetic background-related clinical presentations. Additional gene expression or pathway analyses in the different Madh6-deficient mouse models might provide essential insights into the pathogenesis of these phenotypes. With respect to the observed cardiovascular phenotype in 129/SvEv × BALB/cBy, 129/SvEv × C57Bl/6 and inbred 129/SvEv Madh6−/− mice, a major codominant modifier gene for lethality might be present. Alike to Tgfβ1−/− mice created on different genetic backgrounds to study angiogenesis57,58,59, independent but epistatically interacting genetic loci might be found that determine the incidence of lethality depending on the model. Interesting modifying genes have already been described to alter the response of lack to TGF-β1 in mice, suggesting that proper TGF-β signalling is key for embryonic survival. Whether by analogy, improper BMP or TGF-β signalling explains the incidence of lethality in Madh6−/− mice with a cardiovascular phenotype remains to be determined.
Cellular mechanisms orchestrated by SMAD6
Epithelial-to-mesenchymal transition (EMT) is a reversible fundamental biological process for (1) the formation of the body plan, (2) the differentiation of multiple tissues and organs, and (3) to repair tissues. EMT is an extremely coordinated multifaceted process, in which cells disrupt their intercellular adhesion complexes and lose their apicobasal polarity in order to migrate60,61. Two highly conserved and critical regulators of EMT are the TGF-β and BMP signalling pathway, which either stimulates or tempers this process, respectively62. Hence, SMAD6 modulates EMT by interfering with, predominantly, BMP signalling54,63.
The mechanosensitive BMP signalling pathway (Fig. 2) regulates cellular lineage commitment, morphogenesis, differentiation, proliferation and apoptosis64,65. BMPs activate numerous pathways, of which the SMAD signalling pathway has best been studied66. BMP signalling interferes with its own signalling as SMAD6, a direct BMP target, selectively recruits SMURF1 to BMP type 1 receptors67 or competes with receptor-regulated Smads for binding to SMAD468, thereby establishing a negative feedback loop. A further level of control is achieved by cross-talk with TGF-β, FGF, MAPK, Hedgehog, PI3K/Akt, Wnt/beta-catenin, retinoic acid and Notch signalling pathways in order to regulate cellular BMP-related processes in a very tight spatial and temporal manner64,65,69.
Upon BMP ligand binding, specific type I and type II receptors form a heterotetrameric complex. The type II receptor phosphorylates the type I receptor, which, in turn, phosphorylates Smad1, Smad5, and Smad8 (canonical BMP signalling). Phosphorylated Smads propagate the signal via complex formation with Smad4 and translocates into the nucleus, where it regulates the expression of BMP-responsive target genes. In addition to Smad activation, activated BMP receptor complexes initiates several intracellular pathways to modulate BMP-dependent cellular responses like PI3-kinase, ERK, RhoA, and MAPK/JNK. Canonical BMP signalling is intracellularly inhibited by inhibitory Smads (Smad6, Smad7) and E3 ubiquitin ligases like Smurf1 and Smurf2. Created with BioRender.com. The figure was exported under a paid subscription.
SMAD6 signalling in cardiovascular development
Dysregulation of BMP signalling has extensively been investigated in numerous cardiovascular diseases1,70. Interestingly, SMAD6-deficient patients mainly exhibit defects related to two discrete cell lineages, namely second heart field and neural crest cells. Second heart field cells are multipotent progenitors originating from cardiac progenitor cells and contribute to distinct regions of the myocardium, cardiac endothelial cells and smooth muscle cells71, while neural crest cells are derived from the dorsal aorta and migrate as multipotent cells into the developing outflow tract to coordinate outflow tract septation72. During cardiac cushion development, SMAD6 is specifically expressed in endothelial cells where it functions in (1) maintaining endothelial to mesenchymal transition (EndMT)54,63, (2) stimulating cardiac cushions to grow73 and (3) interacting with cardiac neural crest cells74, cells required for aorticopulmonary septum formation. As such, this might explain the marked clinical variability of SMAD6-deficient patient with BAV-related aortopathy as predominant phenotype, and, emphasises the complexity of CHD, in which gene dosage, timing, haemodynamic flow, and its interplay with other signalling pathways like Notch and TGF-β are important too. For example, endothelial cells can undergo EndMT to become either myofibroblast-like75 or chrondrocyte- and osteoblast-like cells76, depending on their cellular context.
SMAD6 signalling in cranial suture development
Gene discovery studies, and their subsequent characterisation in mice, have determined highly conserved molecular pathways and specific biological processes at different stages in cranial suture development45. Initially, the strongest implication of BMP signalling involvement was shown by BMP type 1 receptor (BMPR1A)77, and by its convergence at key transcriptional factors downstream of BMP, i.e., Msx278 and Twist179,80, to regulate cell proliferation, mesenchyme condensation, osteoblast differentiation, and osteogenesis. Subsequent work further supported a role of SMAD-dependent signalling by the identification of causal mutations in SKI81 and SMAD382 in Shprintzen–Goldberg and Loeys–Dietz patients, both conditions associated with CRS. Additional evidence has emerged as SMAD6-deficiency increases the risk for CRS, and in particular for metopic synostosis. In literature, metopic synostosis has already been hypothesised to be the consequence of abnormal maturation of neural crest-derived mesenchymal stem cells via disturbed dynamics of cell identity or migration as a common predisposing factor, and this can now be further investigated83,84. Alternatively, processes not involved in cranial suture development but affecting osteogenesis such as osteoblast and osteoclast activity could be impaired too, and lead to CRS.
SMAD6 signalling in radioulnar development
Studies on BMP signalling in radioulnar development are very scarce. So far, published data on RUS is limited to genetic studies9,13 and clinical descriptive reports lacking in-depth functional analyses. Our current knowledge is inferred from studies in axial skeletal development, with molecular pathways like Wnt, Hedgehog, Notch, and FGF signalling pathways, to be highly involved85. As RUS is believed to be the result of anomalous differentiation and/or segmentation of the adjacent radius and ulna, it could be true that BMPs lead to impaired mesenchymal stem cell differentiation via Runx2 to promote osteoblast differentiation from mesenchymal precursor cells86,87.
Summary and future outlook
In summary, three distinctive human genetic disorders are caused by SMAD6 deficiency without domain-specific or mutation-type genotype–phenotype correlation making proper patient management difficult. Patients with cardiovascular disease or craniosynostosis do not show any manifestations in the other organ system within relatives of a single family, suggesting that, (an)other factor(s) co-segregating with the primary SMAD6 variant might explain the resultant phenotype. To further explore this hypothesis, in-depth investigation into the identification of the responsible cell type(s) and their identity, as well as defining the predominant affected signalling cascade(s) driving these disorders, will be fundamental for our knowledge. Cell lineage tracing and spatial gene expression analyses in Madh6-deficient mouse models might unravel important clues to discriminate the afflicted processes leading to cardiovascular disease, craniosynostosis and radioulnar synostosis. Furthermore, a detailed clinical and genetic assessment of additional SMAD6 variant-positive patients will be needed, and, in particular, ascertain the complete phenotypic picture of families with RUS, in which some clinical overlap with CHD-, skull-, and skeletal-related anomalies might exist.
Other (additional) genetic factor(s) might explain incomplete penetrance and extreme variability in phenotypical expressivity in a patient with SMAD6 deficiency. For example, rare (or common) variants located in a regulatory element of the trans-wild-type SMAD6 allele, or variants in genes (e.g., SMAD7) afflicting expression and/or activity of the BMP and/or the closely related TGF-β signalling activity are interesting avenues for further exploration. It is worthwhile to consider genome-wide association approaches that look into rare “second-hit” variants with large effect size in SMAD6-deficient patients in order to add novel information to the puzzle. Although this would aid to understand the molecular basis of disease, the current available number of SMAD6 mutant patients might not be sufficient to detect (a) signal(s) even when only extreme phenotypes would be selected. Nonetheless, in the upcoming years we will confidently identify the SMAD6-related molecular patterns associated with these three distinctive genetic disorders. This will allow us to detect early at-risk individuals and empower new therapies.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
All data generated or analysed during this study are included in this published article (and its supplementary information file).
References
Goumans, M. J., Zwijsen, A., Ten Dijke, P. & Bailly, S. Bone morphogenetic proteins in vascular homeostasis and disease. Cold Spring Harb. Perspect. Biol. 10, a031989 (2018).
Wang, R. N. et al. Bone morphogenetic protein (BMP) signaling in development and human diseases. Genes Dis. 1, 87–105 (2014).
Calpena, E. et al. SMAD6 variants in craniosynostosis: genotype and phenotype evaluation. Genet Med. 22, 1498–1506 (2020).
Gillis, E. et al. Candidate gene resequencing in a large bicuspid aortic valve-associated thoracic aortic aneurysm cohort: SMAD6 as an important contributor. Front. Physiol. 8, 400 (2017).
Jin, S. C. et al. Contribution of rare inherited and de novo variants in 2,871 congenital heart disease probands. Nat. Genet. 49, 1593–1601 (2017).
Kloth, K. et al. Biallelic variants in SMAD6 are associated with a complex cardiovascular phenotype. Hum. Genet. 138, 625–634 (2019).
Luyckx, I. et al. Confirmation of the role of pathogenic SMAD6 variants in bicuspid aortic valve-related aortopathy. Eur. J. Hum. Genet. 27, 1044–1053 (2019).
Park, J. E. et al. A novel SMAD6 variant in a patient with severely calcified bicuspid aortic valve and thoracic aortic aneurysm. Mol. Genet. Genom. Med. 7, e620 (2019).
Shen, F. et al. A genotype and phenotype analysis of SMAD6 mutant patients with radioulnar synostosis. Mol. Genet. Genom. Med. 10, e1850 (2022).
Tan, H. L. et al. Nonsynonymous variants in the SMAD6 gene predispose to congenital cardiovascular malformation. Hum. Mutat. 33, 720–727 (2012).
Timberlake, A. T. et al. Two locus inheritance of non-syndromic midline craniosynostosis via rare SMAD6 and common BMP2 alleles. eLife 5, e20125 (2016).
Timberlake, A. T. et al. De novo mutations in inhibitors of Wnt, BMP, and Ras/ERK signaling pathways in non-syndromic midline craniosynostosis. Proc. Natl Acad. Sci. USA 114, E7341–E7347 (2017).
Yang, Y. et al. SMAD6 is frequently mutated in nonsyndromic radioulnar synostosis. Genet Med. 21, 2577–2585 (2019).
Bakker, M. K. et al. Prenatal diagnosis and prevalence of critical congenital heart defects: an international retrospective cohort study. BMJ Open 9, e028139 (2019).
Leirgul, E. et al. Birth prevalence of congenital heart defects in Norway 1994–2009-a nationwide study. Am. Heart J. 168, 956–964 (2014).
Liu, Y. et al. Global birth prevalence of congenital heart defects 1970–2017: updated systematic review and meta-analysis of 260 studies. Int. J. Epidemiol. 48, 455–463 (2019).
McCracken, C. et al. Mortality following pediatric congenital heart surgery: an analysis of the causes of death derived from the national death index. J. Am. Heart Assoc. 7, e010624 (2018).
Verma, S. & Siu, S. C. Aortic dilatation in patients with bicuspid aortic valve. N. Engl. J. Med. 370, 1920–1929 (2014).
Verstraeten, A., Luyckx, I. & Loeys, B. Aetiology and management of hereditary aortopathy. Nat. Rev. 14, 197–208 (2017).
Criado, F. J. Aortic dissection: a 250-year perspective. Tex. Heart Inst. J. 38, 694–700 (2011).
Senser, E. M., Misra, S. & Henkin, S. Thoracic aortic aneurysm: a clinical review. Cardiol. Clin. 39, 505–515 (2021).
Boulet, S. L., Rasmussen, S. A. & Honein, M. A. A population-based study of craniosynostosis in metropolitan Atlanta, 1989-2003. Am. J. Med. Genet. A 146A, 984–991 (2008).
Lajeunie, E., Le Merrer, M., Bonaiti-Pellie, C., Marchac, D. & Renier, D. Genetic study of nonsyndromic coronal craniosynostosis. Am. J. Med. Genet. 55, 500–504 (1995).
Utria, A. F. et al. The importance of timing in optimizing cranial vault remodeling in syndromic craniosynostosis. Plast. Reconstr. Surg. 135, 1077–1084 (2015).
Timberlake, A. T. & Persing, J. A. Genetics of nonsyndromic craniosynostosis. Plast. Reconstr. Surg. 141, 1508–1516 (2018).
Tsai, J. Congenital radioulnar synostosis. Radio. Case Rep. 12, 552–554 (2017).
Mathijssen, I. M. J. Introduction to updated guideline on treatment and management of craniosynostosis. J. Craniofac Surg. 32, 370 (2021).
Pei, X. & Han, J. Efficacy and feasibility of proximal radioulnar derotational osteotomy and internal fixation for the treatment of congenital radioulnar synostosis. J. Orthop. Surg. Res. 14, 81 (2019).
Rao, P. S. Management of congenital heart disease: state of the art-part II-cyanotic heart defects. Children 6, 54 (2019).
Rao, P. S. Management of congenital heart disease: state of the art; part I-ACYANOTIC heart defects. Children 6, 54 (2019).
Braverman, A. C. et al. The bicuspid aortic valve. Curr. Probl. Cardiol. 30, 470–522 (2005).
Blue, G. M., Kirk, E. P., Sholler, G. F., Harvey, R. P. & Winlaw, D. S. Congenital heart disease: current knowledge about causes and inheritance. Med. J. Aust. 197, 155–159 (2012).
McBride, K. L. et al. Inheritance analysis of congenital left ventricular outflow tract obstruction malformations: Segregation, multiplex relative risk, and heritability. Am. J. Med Genet. A 134A, 180–186 (2005).
Morton, S. U., Quiat, D., Seidman, J. G. & Seidman, C. E. Genomic frontiers in congenital heart disease. Nat. Rev. 19, 26–42 (2022).
Bonachea, E. M. et al. Rare GATA5 sequence variants identified in individuals with bicuspid aortic valve. Pediatr. Res. 76, 211–216 (2014).
Shi, L. M. et al. GATA5 loss-of-function mutations associated with congenital bicuspid aortic valve. Int. J. Mol. Med. 33, 1219–1226 (2014).
Guo, D. C. et al. LOX mutations predispose to thoracic aortic aneurysms and dissections. Circ. Res. 118, 928–934 (2016).
Garg, V. et al. Mutations in NOTCH1 cause aortic valve disease. Nature 437, 270–274 (2005).
Guo, D. C. et al. Mutations in smooth muscle alpha-actin (ACTA2) cause coronary artery disease, stroke, and Moyamoya disease, along with thoracic aortic disease. Am. J. Hum. Genet. 84, 617–627 (2009).
Gould, R. A. et al. ROBO4 variants predispose individuals to bicuspid aortic valve and thoracic aortic aneurysm. Nat. Genet. 51, 42–50 (2019).
Parker, L. E. & Landstrom, A. P. Genetic etiology of left-sided obstructive heart lesions: a story in development. J. Am. Heart Assoc. 10, e019006 (2021).
Silberbach, M. et al. Cardiovascular health in turner syndrome: a scientific statement from the American Heart Association. Circ. Genom. Precis. Med. 11, e000048 (2018).
Hinton, R. B. et al. Hypoplastic left heart syndrome links to chromosomes 10q and 6q and is genetically related to bicuspid aortic valve. J. Am. Coll. Cardiol. 53, 1065–1071 (2009).
Mu, T. S., McAdams, R. M. & Bush, D. M. A case of hypoplastic left heart syndrome and bicuspid aortic valve in monochorionic twins. Pediatr. Cardiol. 26, 884–885 (2005).
Twigg, S. R. & Wilkie, A. O. A genetic-pathophysiological framework for craniosynostosis. Am. J. Hum. Genet. 97, 359–377 (2015).
Goos, J. A. C. & Mathijssen, I. M. J. Genetic causes of craniosynostosis: an update. Mol. Syndromol. 10, 6–23 (2019).
Justice, C. M. et al. A genome-wide association study identifies susceptibility loci for nonsyndromic sagittal craniosynostosis near BMP2 and within BBS9. Nat. Genet. 44, 1360–1364 (2012).
Justice, C. M. et al. A genome-wide association study implicates the BMP7 locus as a risk factor for nonsyndromic metopic craniosynostosis. Hum. Genet. 139, 1077–1090 (2020).
Rizzo, R. et al. Autosomal dominant and sporadic radio-ulnar synostosis. Am. J. Med. Genet. 68, 127–134 (1997).
Spritz, R. A. Familial radioulnar synostosis. J. Med. Genet. 15, 160–162 (1978).
Mazauric-Stuker, M., Kordt, G. & Brodersen, D. Y aneuploidy: a further case of a male patient with a 48,XYYY karyotype and literature review. Annales de. genetique 35, 237–240 (1992).
Murgai, A., Altmeyer, S., Wiegand, S., Tylzanowski, P. & Stricker, S. Cooperation of BMP and IHH signaling in interdigital cell fate determination. PLoS ONE 13, e0197535 (2018).
Stafford, D. A., Brunet, L. J., Khokha, M. K., Economides, A. N. & Harland, R. M. Cooperative activity of noggin and gremlin 1 in axial skeleton development. Development 138, 1005–1014 (2011).
Galvin, K. M. et al. A role for smad6 in development and homeostasis of the cardiovascular system. Nat. Genet. 24, 171–174 (2000).
Estrada, K. D., Retting, K. N., Chin, A. M. & Lyons, K. M. Smad6 is essential to limit BMP signaling during cartilage development. J. Bone Miner. Res. 26, 2498–2510 (2011).
Wylie, L. A., Mouillesseaux, K. P., Chong, D. C. & Bautch, V. L. Developmental SMAD6 loss leads to blood vessel hemorrhage and disrupted endothelial cell junctions. Dev. Biol. 442, 199–209 (2018).
Bonyadi, M. et al. Mapping of a major genetic modifier of embryonic lethality in TGF beta 1 knockout mice. Nat. Genet. 15, 207–211 (1997).
Tang, Y. et al. Epistatic interactions between modifier genes confer strain-specific redundancy for Tgfb1 in developmental angiogenesis. Genomics 85, 60–70 (2005).
Tang, Y. et al. Genetic modifiers interact with maternal determinants in vascular development of Tgfb1(-/-) mice. Hum. Mol. Genet. 12, 1579–1589 (2003).
Kalluri, R. & Weinberg, R. A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 119, 1420–1428 (2009).
Thiery, J. P., Acloque, H., Huang, R. Y. & Nieto, M. A. Epithelial-mesenchymal transitions in development and disease. Cell 139, 871–890 (2009).
Kahata, K., Dadras, M. S. & Moustakas, A. TGF-beta family signaling in epithelial differentiation and epithelial-mesenchymal transition. Cold Spring Harb. Perspect. Biol. 10, a022194 (2018).
Desgrosellier, J. S., Mundell, N. A., McDonnell, M. A., Moses, H. L. & Barnett, J. V. Activin receptor-like kinase 2 and Smad6 regulate epithelial-mesenchymal transformation during cardiac valve formation. Dev. Biol. 280, 201–210 (2005).
Garside, V. C., Chang, A. C., Karsan, A. & Hoodless, P. A. Co-ordinating Notch, BMP, and TGF-beta signaling during heart valve development. Cell. Mol. life Sci.: CMLS 70, 2899–2917 (2013).
Gonzalez, D. M. & Medici, D. Signaling mechanisms of the epithelial-mesenchymal transition. Sci. Signal 7, re8 (2014).
Nishimura, R. et al. The role of Smads in BMP signaling. Front. Biosci.: a J. virtual Libr. 8, s275–s284 (2003).
Goto, K., Kamiya, Y., Imamura, T., Miyazono, K. & Miyazawa, K. Selective inhibitory effects of Smad6 on bone morphogenetic protein type I receptors. J. Biol. Chem. 282, 20603–20611 (2007).
Hata, A., Lagna, G., Massague, J. & Hemmati-Brivanlou, A. Smad6 inhibits BMP/Smad1 signaling by specifically competing with the Smad4 tumor suppressor. Genes Dev. 12, 186–197 (1998).
Derynck, R. & Zhang, Y. E. Smad-dependent and Smad-independent pathways in TGF-beta family signalling. Nature 425, 577–584 (2003).
Wang, J., Greene, S. B. & Martin, J. F. BMP signaling in congenital heart disease: new developments and future directions. Birth Defects Res. A Clin. Mol. Teratol. 91, 441–448 (2011).
Kelly, R. G. The second heart field. Curr. Top. Dev. Biol. 100, 33–65 (2012).
Plein, A., Fantin, A. & Ruhrberg, C. Neural crest cells in cardiovascular development. Curr. Top. Dev. Biol. 111, 183–200 (2015).
Yamada, M., Szendro, P. I., Prokscha, A., Schwartz, R. J. & Eichele, G. Evidence for a role of Smad6 in chick cardiac development. Dev. Biol. 215, 48–61 (1999).
Delot, E. C. Control of endocardial cushion and cardiac valve maturation by BMP signaling pathways. Mol. Genet. Metab. 80, 27–35 (2003).
Kovacic, J. C. et al. Endothelial to mesenchymal transition in cardiovascular disease: JACC state-of-the-art review. J. Am. Coll. Cardiol. 73, 190–209 (2019).
Gomez-Stallons, M. V., Wirrig-Schwendeman, E. E., Hassel, K. R., Conway, S. J. & Yutzey, K. E. Bone morphogenetic protein signaling is required for aortic valve calcification. Arteriosclerosis Thrombosis Vasc. Biol. 36, 1398–1405 (2016).
Komatsu, Y. et al. Augmentation of Smad-dependent BMP signaling in neural crest cells causes craniosynostosis in mice. J. Bone Miner. Res. 28, 1422–1433 (2013).
Jabs, E. W. et al. A mutation in the homeodomain of the human MSX2 gene in a family affected with autosomal dominant craniosynostosis. Cell 75, 443–450 (1993).
el Ghouzzi, V. et al. Mutations of the TWIST gene in the Saethre-Chotzen syndrome. Nat. Genet. 15, 42–46 (1997).
Howard, T. D. et al. Mutations in TWIST, a basic helix-loop-helix transcription factor, in Saethre-Chotzen syndrome. Nat. Genet. 15, 36–41 (1997).
Doyle, A. J. et al. Mutations in the TGF-beta repressor SKI cause Shprintzen-Goldberg syndrome with aortic aneurysm. Nat. Genet. 44, 1249–1254 (2012).
Velchev, J. D., Van Laer, L., Luyckx, I., Dietz, H. & Loeys, B. Loeys-Dietz syndrome. Adv. Exp. Med Biol. 1348, 251–264 (2021).
Piacentino, M. L., Hutchins, E. J. & Bronner, M. E. Essential function and targets of BMP signaling during midbrain neural crest delamination. Dev. Biol. 477, 251–261 (2021).
Siismets, E. M. & Hatch, N. E. Cranial neural crest cells and their role in the pathogenesis of craniofacial anomalies and coronal craniosynostosis. J. Dev. Biol. 8, 18 (2020).
Williams, S., Alkhatib, B. & Serra, R. Development of the axial skeleton and intervertebral disc. Curr. Top. Dev. Biol. 133, 49–90 (2019).
Liu, Q. et al. Recent advances of osterix transcription factor in osteoblast differentiation and bone formation. Front Cell Dev. Biol. 8, 601224 (2020).
Phimphilai, M., Zhao, Z., Boules, H., Roca, H. & Franceschi, R. T. BMP signaling is required for RUNX2-dependent induction of the osteoblast phenotype. J. Bone Miner. Res. 21, 637–646 (2006).
Miyazawa, K. & Miyazono, K. Regulation of TGF-beta family signalling by inhibitory smads. Cold Spring Harb Perspect Biol. 9, a022095 (2017).
Lo, R. S. et al. The L3 loop: a structural motif determining specific interactions between SMAD proteins and TGF-β receptors. EMBO J. 17, 996–1005 (1998).
Acknowledgements
This research was supported by funding from the University of Antwerp (Methusalem-OEC grant “Genomed” FFB190208). B.L. holds a consolidator grant from the European Research Council (Genomia – ERC-COG-2017-771945). B.L. and A.V. are members of the European Reference Network on rare multisystemic vascular disorders (VASCERN - project ID: 769036 partly co-funded by the European Union Third Health Programme). I.L. is supported by the Outreach project (Dutch Heart Foundation).
Author information
Authors and Affiliations
Contributions
I.L. and B.L. conceived the idea. I.L. drafted the initial manuscript and revised the manuscript. A.V., M.J.G. and B.L. contributed to the critical review and editing of the manuscript. All authors contributed to the review and the final approval of the completed version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Luyckx, I., Verstraeten, A., Goumans, MJ. et al. SMAD6-deficiency in human genetic disorders. npj Genom. Med. 7, 68 (2022). https://doi.org/10.1038/s41525-022-00338-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41525-022-00338-5
This article is cited by
-
Deamidation enables pathogenic SMAD6 variants to activate the BMP signaling pathway
Science China Life Sciences (2024)