Abstract
To evaluate whether mammographic texture features were associated with second primary contralateral breast cancer (CBC) risk, we created a “texture risk score” using pre-treatment mammograms in a case–control study of 212 women with CBC and 223 controls with unilateral breast cancer. The texture risk score was associated with CBC (odds per adjusted standard deviation = 1.25, 95% CI 1.01–1.56) after adjustment for mammographic percent density and confounders. These results support the potential of texture features for CBC risk assessment of breast cancer survivors.
Similar content being viewed by others
Manuscript text
Breast cancer survivors have a high risk of developing a second primary contralateral breast cancer (CBC), between 2 and 6 times greater than the risk of first primary breast cancer in the general population1. CBC risk factors include younger age and an estrogen receptor (ER)-negative first breast cancer, family history of breast cancer, and stray radiation dose received by the contralateral breast during treatment for the first breast cancer1,2,3,4,5,6. Mammographic percent density (MPD), among the stronger known predictors for first primary breast cancer, also predicts CBC risk, but the associations are generally weaker than for first primary breast cancer7,8. Other quantitative features from mammograms (namely, “texture” features) are possibly associated with first primary breast cancer risk9,10, but it is not known whether these features may also identify breast cancer survivors at increased risk of subsequent CBC. In this study, we developed and tested a “texture risk score” for CBC using the Women’s Environment, Cancer, and Radiation Epidemiology (WECARE) Study.
The feature reduction model used to select the most predictive texture features was the LASSO (λ = 0.033), which identified three features with non-zero coefficients: gray level run length matrix (GLRLM) short run low gray level emphasis (weight = −0.032); gray level size zone matrix (GLSZM) small area emphasis (weight = −0.093); and GLSZM small area low gray level emphasis (weight = −0.066). The Pearson correlation between the texture risk score and MPD was 0.37 (standard error = 0.04). The score was approximately normally distributed with mean = 0, standard deviation = 0.14, and skewness = −0.98 (see Fig. 1) and was not further transformed. The characteristics of participants with a lower (<median) vs higher (≥median) texture risk score differed by BMI but no other clinical or epidemiological characteristics (Supplementary Table 2).
Table 1 shows that, in multivariable-adjusted models, CBC was statistically significantly associated with the texture risk score for age and BMI (odds ratio per adjusted standard deviation (OPERA) = 1.31, 95% CI 1.06–1.62), and borderline significantly associated with MPD for age and BMI (OPERA 1.21, 95% CI 1.00–1.48). When fitting the texture risk score and MPD together, the association of CBC with the texture risk score remained statistically significant with minimal attenuation while the association with MPD was attenuated by 31% and was no longer statistically significant.
From stratified model fits, the association of CBC with the texture risk score was statistically significant for premenopausal women (OPERA 1.39, 95% CI 1.08–1.81), but not for postmenopausal women (OPERA 0.95, 95% CI 0.60–1.48); the OPERA estimates did not differ statistically from one another (P-heterogeneity = 0.1). The association of CBC with the texture risk score was greater for women with body mass index (BMI) ≥ 25 kg/m2 (1.86, 95% CI 1.23–2.94) compared with women with BMI < 25 kg/m2 (1.07, 95% CI 0.82–1.40; P-heterogeneity = 0.03) (Table 2).
This study evaluated the association of quantitative mammographic features with CBC risk for breast cancer survivors, whose absolute risk of CBC is high1. We found that the combination of three mammographic texture features was associated with CBC after adjusting for known CBC risk factors. Importantly, fitting the texture risk score in a model with MPD strongly attenuated the association between CBC and MPD. This finding is consistent with previous studies of first primary breast cancer, where inclusion of new mammogram-based risk measures, such as texture features, resulted in the associations with conventional MPD attenuating to the null11,12. While increased MPD is consistently one of the strongest known risk factors (ORs > 2.0 per SD) for first primary breast cancer, the association with CBC is generally weaker, as reported in the WECARE Study and elsewhere7,8,13.
The association of CBC with our texture risk score was essentially unchanged after adjusting for MPD, suggesting that this score, though weakly correlated with MPD, is capturing independent sources of variation in CBC risk that are potentially causal for breast cancer risk9,14. Additionally, we identified possible effect modification of the association between CBC and the texture risk score by BMI, which is consistent with BMI-modified associations between mammographic density and first primary breast cancer15.
Strengths of this study include control for epidemiological and clinical risk factors of CBC, as well as the use of mammograms taken prior to receiving systemic therapy for the first primary breast cancer, which ensured our findings were not influenced by treatment-related changes to the breast. There were several limitations, however. First, this study lacks external validation of the texture risk score for CBC risk. We attempted to reduce the probability of overfitting by selecting the feature reduction method based on a random subset of the data, as well as by developing only one risk score, and we hope to evaluate this risk score in other large imaging studies of CBC risk when feasible. Second, the WECARE Study recruited participants during the era of film mammograms, which are rarely used today. However, previous studies comparing mammogram-based measures on film and digital images have found little difference in measuring breast size, mammographic density, and risk associations using texture features16,17. Third, while the WECARE Study is the largest imaging study of CBC risk with extensive clinical and epidemiological data, we were unable to conduct well-powered subgroup analyses.
In conclusion, mammographic texture features—which are objective and can be generated automatically—are a promising approach to improve risk stratification and tailor surveillance for CBC beginning at the time of the first primary cancer diagnosis.
Methods
The WECARE Study is a multicenter, population-based, individually matched, case-control study of CBC18. Case women were diagnosed with a first primary invasive breast cancer followed ≥1 year later by a second primary invasive cancer in the contralateral breast. Control women were diagnosed with a first primary unilateral invasive breast cancer (UBC) but did not have a second primary invasive CBC and were selected such that each matched control had similar time at risk for CBC (“at-risk time”) as her matched case, with additional matching on age, year of diagnosis, race/ethnicity, and recruitment center. After providing informed consent, participants completed a structured questionnaire and study staff completed medical record abstraction. In the second recruitment phase, the WECARE II Study (2010–2012), participants identified from four population-based cancer registries (Iowa, Seattle, Northern California, and Ontario) also provided consent to access their mammogram of the unaffected breast taken prior to systemic treatment for the first breast cancer. The WECARE Study did not collect the characteristics of facilities where the mammograms were obtained. Nearly all images were film mammograms, which were digitized at two study sites (Seattle and Toronto) using two Kodak Lumisys Digital Scanners set to 12-bit gray-scale resolution with regular calibration (see Supplementary Methods for details). MPD was estimated centrally in Toronto for each film mammogram using the semi-automated thresholding software, Cumulus19. All study participants provided written informed consent and the study was approved by the institutional ethics review boards at the University of Iowa (IRB-01), Fred Hutchinson Cancer Research Center, Cancer Prevention Institute of California, Mount Sinai Hospital, and Memorial Sloan Kettering Cancer Center and by the Committee for the Protection of Human Subjects of the State of California.
A fully automated analysis pipeline was used to calculate texture features from digitized cranio-caudal view film mammograms taken prior to systemic therapy for the first breast cancer. Automated in-house software was used to identify the breast area mask (i.e. region for analysis). The range of gray levels within the mask were binned (discretized) into eight gray levels based on intensity quantiles, rather than absolute intensity, to account for possible between-image brightness differences attributable to image acquisition and digitization9. From the discretized masks, we calculated five texture matrices: the gray level co-occurrence matrix (GLCM), the gray level run-length matrix (GLRLM), the gray level size zone matrix (GLSZM), the neighborhood gray tone difference matrix (NGTDM), and the neighborhood gray level dependence matrix (NGLDM), with detailed extraction parameters provided in Supplementary Table 1. Seventy-seven unique features, as defined by the Image Biomarker Standardization Initiative20, were calculated for the entire breast area from these matrices. Features were centered prior to analysis. Calculation of matrices and features was conducted using the open-source MATLAB package, CERR21.
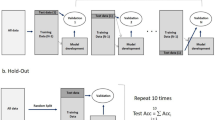
To identify combinations of features associated with CBC, we considered three different penalized regression models: ridge (L2 regularization), elastic net (L1 and/or L2 regularization), and lLASSO (L1 regularization). To identify the tuning parameters for each model without overfitting, we selected 70% of CBC cases and UBC controls at random, and for each model type used 10-fold cross-validation repeated 20 times to select a value of λ between 10−3 and 103 (and, for elastic net, α between 0 and 1). After identifying the best tuning parameters for the ridge, elastic net, and LASSO models, we selected one model from the three that produced the lowest unadjusted root mean square error for CBC prediction in the 30% holdout data. The chosen model was used to develop a “texture risk score” calculated for each WECARE Study participant as a linear combination of the scaled feature values multiplied by the coefficients from the selected penalized regression model.
The association between CBC and the texture risk score was examined by fitting a logistic regression model. To enable direct comparison of the texture risk score and MPD, both measures were parameterized as odds ratio per adjusted standard deviation (OPERA) by dividing the age- and body mass index (BMI)-adjusted residuals of each measure for the controls by the standard deviation of the residuals22. We evaluated the association between each OPERA measure and CBC risk, and then further adjusted the models for menopausal status at time of mammogram; stage (localized or regional) and ER status at the time of the first diagnosis; receipt of chemotherapy, hormonal therapy, and radiation therapy for the first breast cancer; first-degree family history of breast cancer; age at menarche; and matching factors. Exploratory stratified models were estimated within subgroups defined by menopausal status at time of mammogram and BMI (<25 kg/m2 vs ≥25 kg/m2) prior to the first diagnosis.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Access to analytic code repository will be provided upon request.
References
Chen, Y., Thompson, W., Semenciw, R. & Mao, Y. Epidemiology of contralateral breast cancer. Cancer Epidemiol. Biomark. Prev. 8, 855–861 (1999).
Bernstein, J., Thompson, W., Risch, N. & Holford, T. Risk factors predicting the incidence of second primary breast cancer among women diagnosed with a first primary breast cancer. Am. J. Epidemiol. 136, 925–936 (1992).
Brooks, J. D. et al. Body mass index, weight change, and risk of second primary breast cancer in the WECARE study: influence of estrogen receptor status of the first breast cancer. Cancer Med. 5, 3282–3291 (2016).
Langballe, R. et al. Systemic therapy for breast cancer and risk of subsequent contralateral breast cancer in the WECARE Study. Breast Cancer Res. 18, 65 (2016).
Reiner, A. S. et al. Hormone receptor status of a first primary breast cancer predicts contralateral breast cancer risk in the WECARE study population. Breast Cancer Res. 19, 83 (2017).
Stovall, M. et al. Dose to the contralateral breast from radiotherapy and risk of second primary breast cancer in the WECARE study. Int J. Radiat. Oncol. Biol. Phys. 72, 1021–1030 (2008).
Raghavendra, A. et al. Mammographic breast density is associated with the development of contralateral breast. Cancer 123, 1935–1940 (2017).
Knight, J. A. et al. The association of mammographic density with risk of contralateral breast cancer and change in density with treatment in the WECARE study. Breast Cancer Res. 20, 23 (2018).
Schmidt, D. F. et al. Cirrus: an automated mammography-based measure of breast cancer risk based on textural features. JNCI Cancer Spectr. 2 2, pky057 (2018).
Kontos, D. et al. Radiomic phenotypes of mammographic parenchymal complexity: toward augmenting breast density in breast cancer risk assessment. Radiology 290, 41–49 (2019).
Nguyen, T. L. et al. Predicting interval and screen-detected breast cancers from mammographic density defined by different brightness thresholds. Breast Cancer Res. 20, 152 (2018).
Nguyen, T. L. et al. Novel mammogram-based measures improve breast cancer risk prediction beyond an established mammographic density measure. Int. J. Cancer 148, 2193–2202 (2021).
Sandberg, M. E. et al. Change of mammographic density predicts the risk of contralateral breast cancer–a case-control study. Breast Cancer Res. 15, R57 (2013).
Hopper, J. L. et al. Going beyond conventional mammographic density to discover novel mammogram-based predictors of breast cancer risk. J. Clin. Med. 9, 627 (2020).
Chen, H. et al. Association of interactions between mammographic density phenotypes and established risk factors with breast cancer risk, by tumor subtype and menopausal status. Am. J. Epidemiol. 190, 44–58 (2020).
Burton, A. et al. Mammographic density assessed on paired raw and processed digital images and on paired screen-film and digital images across three mammography systems. Breast Cancer Res. 18, 130–130 (2016).
Li, H. et al. Computerized analysis of mammographic parenchymal patterns on a large clinical dataset of full-field digital mammograms: robustness study with two high-risk datasets. J. Digital Imaging 25, 591–598 (2012).
Bernstein, J. L. et al. Study design: evaluating gene-environment interactions in the etiology of breast cancer - the WECARE study. Breast Cancer Res. 6, R199–R214 (2004).
Byng, J. W., Boyd, N. F., Fishell, E., Jong, R. A. & Yaffe, M. J. The quantitative analysis of mammographic densities. Phys. Med. Biol. 39, 1629–1638 (1994).
Zwanenburg, A. et al. The image biomarker standardization initiative: standardized quantitative radiomics for high-throughput image-based phenotyping. Radiology 295, 328–338 (2020).
Apte, A. P. et al. Technical note: extension of CERR for computational radiomics: a comprehensive MATLAB platform for reproducible radiomics research. Med. Phys. https://doi.org/10.1002/mp.13046 (2018).
Hopper, J. L. Odds per adjusted standard deviation: comparing strengths of associations for risk factors measured on different scales and across diseases and populations. Am. J. Epidemiol. 182, 863–867 (2015).
Acknowledgements
The authors would like to thank the participants of the WECARE Study for making this work possible. This work was supported by the United States National Cancer Institute award numbers R01 CA168339, R01 CA97397, P30 CA008748, U01 CA83178, R01 CA129649, R01 CA129639, R01 CA114236, and P30CA086862; by the Victoria Cancer Council award numbers 1139144 and TP900783; and by the Cure Cancer Australia Foundation award number 1159399.
Author information
Authors and Affiliations
Contributions
G.P.W. conceived the study, completed the analysis, and drafted the manuscript; J.A.K. supervised data collection and digitization of mammograms, completed CUMULUS measurements, guided analysis, and critically revised the manuscript; C.L. developed the automatic breast segmentation algorithm; C.F.L. supervised data collection, guided analysis, and critically revised the manuscript; K.E.M. supervised data collection and digitization of mammograms, advised analysis and critically revised the manuscript; E.M.J. supervised data collection, advised analysis, and critically revised the manuscript; L.B. advised analysis and critically revised the manuscript; J.D.B. contributed analysis and critically revised the manuscript; A.S.R. advised analysis and critically revised the manuscript. X.L. developed analytic files and critically revised the manuscript; M.W. managed clinical and imaging data and critically revised the manuscript. T.L.N. advised the statistical approach and critically revised the manuscript; J.L.H. advised the ‘OPERA’ approach and critically revised the manuscript; M.C.P. supervised the statistical analysis and critically revised the manuscript; J.L.B. is the P.I. of the WECARE Study, supervised the analysis, and critically revised the manuscript. All authors approved the final submitted manuscript and take responsibility for the accuracy and integrity of the findings.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Watt, G.P., Knight, J.A., Lin, C. et al. Mammographic texture features associated with contralateral breast cancer in the WECARE Study. npj Breast Cancer 7, 146 (2021). https://doi.org/10.1038/s41523-021-00354-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41523-021-00354-1