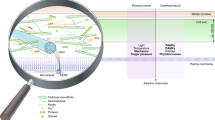
Abstract
The plant cell wall has many roles: structure, hydraulics, signalling and immunity. Monitoring its status is therefore essential for plant life. Among many candidate cell wall sensors, FERONIA, a member of the Catharanthus roseus receptor-like kinase-1-like kinase (CrRLK1L) family, has received considerable attention, notably because of its numerous interactors and its implication in many biological pathways. Conversely, such an analytical dissection may blur its core function. Here we revisit the array of feronia phenotypes as an attempt to identify a unifying feature behind the plethora of biological and biochemical functions. We propose that the contribution of FERONIA in monitoring turgor-dependent cell wall tension may explain its pleiotropy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Wolf, S. Cell wall signaling in plant development and defense. Annu. Rev. Plant Biol. https://doi.org/10.1146/annurev-arplant-102820-095312 (2022).
Ortiz-Morea, F. A., Liu, J., Shan, L. & He, P. Malectin-like receptor kinases as protector deities in plant immunity. Nat. Plants 8, 27–37 (2021).
Escobar-Restrepo, J.-M. et al. The FERONIA receptor-like kinase mediates male–female interactions during pollen tube reception. Science 317, 656–660 (2007).
Duan, Q., Kita, D., Li, C., Cheung, A. Y. & Wu, H.-M. FERONIA receptor-like kinase regulates RHO GTPase signaling of root hair development. Proc. Natl Acad. Sci. USA 107, 17821–17826 (2010).
Guo, H. et al. Three related receptor-like kinases are required for optimal cell elongation in Arabidopsis thaliana. Proc. Natl Acad. Sci. USA 106, 7648–7653 (2009).
Duan, Q. et al. Reactive oxygen species mediate pollen tube rupture to release sperm for fertilization in Arabidopsis. Nat. Commun. 5, 3129 (2014).
Huck, N., Moore, J. M., Federer, M. & Grossniklaus, U. The Arabidopsis mutant feronia disrupts the female gametophytic control of pollen tube reception. Development 130, 2149–2159 (2003).
Yeats, T. H., Sorek, H., Wemmer, D. E. & Somerville, C. R. Cellulose deficiency is enhanced on hyper accumulation of sucrose by a H+-coupled sucrose symporter. Plant Physiol. 171, 110–124 (2016).
Malivert, A. et al. FERONIA and microtubules independently contribute to mechanical integrity in the Arabidopsis shoot. PLoS Biol. 19, e3001454 (2021).
Li, C. et al. Glycosylphosphatidylinositol-anchored proteins as chaperones and co-receptors for FERONIA receptor kinase signaling in Arabidopsis. eLife 4, e06587 (2015).
Lin, W. et al. Arabidopsis pavement cell morphogenesis requires FERONIA binding to pectin for activation of ROP GTPase signaling. Curr. Biol. https://doi.org/10.1016/j.cub.2021.11.030 (2021).
Haruta, M., Sabat, G., Stecker, K., Minkoff, B. B. & Sussman, M. R. A peptide hormone and its receptor protein kinase regulate plant cell expansion. Science 343, 408–411 (2014).
Dong, Q., Zhang, Z., Liu, Y., Tao, L.-Z. & Liu, H. FERONIA regulates auxin-mediated lateral root development and primary root gravitropism. FEBS Lett. 593, 97–106 (2019).
Keinath, N. F. et al. PAMP (pathogen-associated molecular pattern)-induced changes in plasma membrane compartmentalization reveal novel components of plant immunity. J. Biol. Chem. 285, 39140–39149 (2010).
Stegmann, M. et al. The receptor kinase FER is a RALF-regulated scaffold controlling plant immune signaling. Science 355, 287–289 (2017).
Zhang, X., Yang, Z., Wu, D. & Yu, F. RALF–FERONIA signaling: linking plant immune response with cell growth. Plant Commun. 1, 100084 (2020).
Tang, J. et al. Plant immunity suppression via PHR1–RALF–FERONIA shapes the root microbiome to alleviate phosphate starvation. EMBO J. 41, e109102 (2022).
Gronnier, J. et al. Regulation of immune receptor kinase plasma membrane nanoscale organization by a plant peptide hormone and its receptors. eLife 11, e74162 (2022).
Kessler, S. A. et al. Conserved molecular components for pollen tube reception and fungal invasion. Science 330, 968–971 (2010).
Masachis, S. et al. A fungal pathogen secretes plant alkalinizing peptides to increase infection. Nat. Microbiol. 1, 16043 (2016).
Huang, Y.-Y. et al. Identification of FERONIA-like receptor genes involved in rice–Magnaporthe oryzae interaction. Phytopathol. Res. 2, 14 (2020).
Chen, J. et al. FERONIA interacts with ABI2-type phosphatases to facilitate signaling cross-talk between abscisic acid and RALF peptide in Arabidopsis. Proc. Natl Acad. Sci. USA 113, E5519–E5527 (2016).
Feng, W. et al. The FERONIA receptor kinase maintains cell-wall integrity during salt stress through Ca2+ signaling. Curr. Biol. 28, 666–675.e5 (2018).
Richter, J., Ploderer, M., Mongelard, G., Gutierrez, L. & Hauser, M.-T. Role of CrRLK1L cell wall sensors HERCULES1 and 2, THESEUS1, and FERONIA in growth adaptation triggered by heavy metals and trace elements. Front Plant Sci. 8, 1554 (2017).
Wang, L. et al. Receptor kinase FERONIA regulates flowering time in Arabidopsis. BMC Plant Biol. 20, 26 (2020).
Yu, F. et al. FERONIA receptor kinase controls seed size in Arabidopsis thaliana. Mol. Plant 7, 920–922 (2014).
Yang, T. et al. Receptor protein kinase FERONIA controls leaf starch accumulation by interacting with glyceraldehyde-3-phosphate dehydrogenase. Biochem. Biophys. Res. Commun. 465, 77–82 (2015).
Du, S., Qu, L.-J. & Xiao, J. Crystal structures of the extracellular domains of the CrRLK1L receptor-like kinases ANXUR1 and ANXUR2. Protein Sci. 27, 886–892 (2018).
Moussu, S., Augustin, S., Roman, A. O., Broyart, C. & Santiago, J. Crystal structures of two tandem malectin-like receptor kinases involved in plant reproduction. Acta Crystallogr. D Struct. Biol. 74, 671–680 (2018).
Dünser, K. et al. Extracellular matrix sensing by FERONIA and Leucine-Rich Repeat Extensins controls vacuolar expansion during cellular elongation in Arabidopsis thaliana. EMBO J. 38, e100353 (2019).
Xiao, Y. et al. Mechanisms of RALF peptide perception by a heterotypic receptor complex. Nature 572, 270–274 (2019).
Deslauriers, S. D. & Larsen, P. B. FERONIA is a key modulator of brassinosteroid and ethylene responsiveness in Arabidopsis hypocotyls. Mol. Plant 3, 626–640 (2010).
Xie, Y. et al. FERONIA receptor kinase integrates with hormone signaling to regulate plant growth, development, and responses to environmental stimuli. Int. J. Mol. Sci. 23, 3730 (2022).
Zhao, C. et al. The LRXs–RALFs–FER module controls plant growth and salt stress responses by modulating multiple plant hormones. Natl Sci. Rev. 8, nwaa149 (2021).
Wang, G., Zhao, Z., Zheng, X., Shan, W. & Fan, J. How a single receptor-like kinase exerts diverse roles: lessons from FERONIA. Mol. Hortic. 2, 25 (2022).
Verger, S., Long, Y., Boudaoud, A. & Hamant, O. A tension–adhesion feedback loop in plant epidermis. eLife 7, e34460 (2018).
Gigli-Bisceglia, N., van Zelm, E., Huo, W., Lamers, J. & Testerink, C. Arabidopsis root responses to salinity depend on pectin modification and cell wall sensing. Development 149, dev200363 (2022).
Borowska-Wykręt, D., Elsner, J., De Veylder, L. & Kwiatkowska, D. Defects in leaf epidermis of Arabidopsis thaliana plants with CDKA;1 activity reduced in the shoot apical meristem. Protoplasma 250, 955–961 (2013).
Gu, Y. et al. Identification of a cellulose synthase-associated protein required for cellulose biosynthesis. Proc. Natl Acad. Sci. USA 107, 12866–12871 (2010).
Duan, Q. et al. FERONIA controls pectin- and nitric oxide-mediated male–female interaction. Nature https://doi.org/10.1038/s41586-020-2106-2 (2020).
Shih, H.-W., Miller, N. D., Dai, C., Spalding, E. P. & Monshausen, G. B. The receptor-like kinase FERONIA is required for mechanical signal transduction in Arabidopsis seedlings. Curr. Biol. 24, 1887–1892 (2014).
Beauzamy, L., Nakayama, N. & Boudaoud, A. Flowers under pressure: ins and outs of turgor regulation in development. Ann. Bot. 114, 1517–1533 (2014).
Tang, W. et al. Mechano-transduction via the pectin–FERONIA complex activates ROP6 GTPase signaling in Arabidopsis pavement cell morphogenesis. Curr. Biol. https://doi.org/10.1016/j.cub.2021.11.031 (2021).
Bellati, J. et al. Novel aquaporin regulatory mechanisms revealed by interactomics. Mol. Cell. Proteom. 15, 3473–3487 (2016).
Kapilan, R., Vaziri, M. & Zwiazek, J. J. Regulation of aquaporins in plants under stress. Biol. Res. 51, 4 (2018).
Rodrigues, O. et al. Aquaporins facilitate hydrogen peroxide entry into guard cells to mediate ABA- and pathogen-triggered stomatal closure. Proc. Natl Acad. Sci. USA 114, 9200–9205 (2017).
Xu, G. et al. FERONIA phosphorylates E3 ubiquitin ligase ATL6 to modulate the stability of 14-3-3 proteins in response to the carbon/nitrogen ratio. J. Exp. Bot. 70, 6375–6388 (2019).
Prado, K. et al. Oscillating aquaporin phosphorylation and 14-3-3 proteins mediate the circadian regulation of leaf hydraulics. Plant Cell 31, 417–429 (2019).
Choquet, D., Felsenfeld, D. P. & Sheetz, M. P. Extracellular matrix rigidity causes strengthening of integrin-cytoskeleton linkages. Cell 88, 39–48 (1997).
Haas, K. T., Wightman, R., Meyerowitz, E. M. & Peaucelle, A. Pectin homogalacturonan nanofilament expansion drives morphogenesis in plant epidermal cells. Science 367, 1003–1007 (2020).
Sapala, A. et al. Why plants make puzzle cells, and how their shape emerges. eLife 7, e32794 (2018).
Ferrari, S. et al. Oligogalacturonides: plant damage-associated molecular patterns and regulators of growth and development. Front. Plant Sci. 4, 49 (2013).
Zhao, C. et al. Leucine-rich repeat extensin proteins regulate plant salt tolerance in Arabidopsis. Proc. Natl Acad. Sci. USA 115, 13123–13128 (2018).
Hamilton, E. S. et al. Mechanosensitive channel MSL8 regulates osmotic forces during pollen hydration and germination. Science 350, 438–441 (2015).
Guichard, M., Thomine, S. & Frachisse, J.-M. Mechanotransduction in the spotlight of mechano-sensitive channels. Curr. Opin. Plant Biol. 68, 102252 (2022).
Yuan, F. et al. OSCA1 mediates osmotic-stress-evoked Ca2+ increases vital for osmosensing in Arabidopsis. Nature 514, 367–371 (2014).
Radin, I. et al. Plant PIEZO homologs modulate vacuole morphology during tip growth. Science 373, 586–590 (2021).
Mielke, S. et al. Jasmonate biosynthesis arising from altered cell walls is prompted by turgor-driven mechanical compression. Sci. Adv. 7, eabf0356 (2021).
Popko, J., Hänsch, R., Mendel, R.-R., Polle, A. & Teichmann, T. The role of abscisic acid and auxin in the response of poplar to abiotic stress. Plant Biol. 12, 242–258 (2010).
Author information
Authors and Affiliations
Contributions
A.M. wrote the initial draft of the manuscript. A.M. and O.H. revised and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Plants thanks José Dinneny, Sebastian Wolf and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Malivert, A., Hamant, O. Why is FERONIA pleiotropic?. Nat. Plants 9, 1018–1025 (2023). https://doi.org/10.1038/s41477-023-01434-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-023-01434-9
This article is cited by
-
Structure and growth of plant cell walls
Nature Reviews Molecular Cell Biology (2024)