Abstract
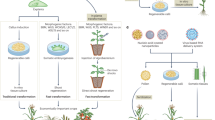
Mixing maternal and paternal genomes in embryos is not only responsible for the evolutionary success of sexual reproduction, but is also a cornerstone of plant breeding. However, once an interesting gene combination is obtained, further genetic mixing is problematic. To rapidly fix genetic information, doubled haploid plants can be produced: haploid embryos having solely the genetic information from one parent are allowed to develop, and chromosome doubling generates fully homozygous plants. A powerful path to the production of doubled haploids is based on haploid inducer lines. A simple cross between a haploid inducer line and the line with gene combinations to be fixed will trigger haploid embryo development. However, the exact mechanism behind in planta haploid induction remains an enduring mystery. The recent discoveries of molecular actors triggering haploid induction in the maize crop and the model Arabidopsis thaliana pinpoint an essential role of processes related to gamete development, gamete interactions and genome stability. These findings enabled translation of haploid induction capacity to other crops as well as the use of haploid inducer lines to deliver genome editing machinery into various crop varieties. These recent advances not only hold promise for the next generations of plant breeding strategies, but they also provide a deeper insight into the fundamental bases of sexual reproduction in plants.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Forster, B. P. & Thomas, W. T. B. in Plant Breeding Reviews (Ed. Janick, J.) 57–88 (John Wiley & Sons, 2005).
Wędzony, M. et al. in Advances in Haploid Production in Higher Plants (eds Touraev, A. et al.) 1–33 (Springer, 2009).
Gilles, L. M., Martinant, J.-P., Rogowsky, P. M. & Widiez, T. Haploid induction in plants. Curr. Biol. 27, R1095–R1097 (2017).
Doubled Haploid Production in Crop Plants: a Manual (eds Maluszynski, M. et al.) (Springer, 2003).
Dunwell, J. M. Haploids in flowering plants: origins and exploitation. Plant Biotechnol. J 8, 377–424 (2010).
Forster, B. P., Heberle-Bors, E., Kasha, K. J. & Touraev, A. The resurgence of haploids in higher plants. Trends Plant Sci. 12, 368–375 (2007).
Guha, S. & Maheshwari, S. C. In vitro production of embryos from anthers of Datura. Nature 204, 497–497 (1964).
Advances in Haploid Production in Higher Plants (eds Touraev, A. et al.) (Springer, 2009).
Haploids in Crop Improvement II (eds Palmer, C. E. D. et al.) (Springer, 2005).
Bajaj, Y. P. S. Haploids in Crop Improvement I Vol. 12 (Springer-Verlag, 1990).
In Vitro Haploid Production in Higher Plants (eds Jain, S. et al.) Vol. 25 (Springer, 1996).
Ishii, T., Karimi-Ashtiyani, R. & Houben, A. Haploidization via chromosome elimination: means and mechanisms. Annu. Rev. Plant Biol. 67, 421–438 (2016).
Kalinowska, K. et al. State-of-the-art and novel developments of in vivo haploid technologies. Theor. Appl. Genet. 132, 593–605 (2019).
Kermicle, J. L. Androgenesis conditioned by a mutation in maize. Science 166, 1422–1424 (1969).
Coe, E. H. A line of maize with high haploid frequency. The American Naturalist 93, 381–382 (1959).
Rober, F. K., Gordillo, G. A. & Geiger, H. H. In vivo haploid induction in maize-performance of new inducers and significance of doubled haploid lines in hybrid breeding. Maydica 50, 275 (2005).
Geiger, H. H. & Gordillo, G. A. Doubled haploids in hybrid maize breeding. Maydica 54, 485–499 (2009).
Sarkar, K. R. & Coe, E. H. A genetic analysis of the origin of maternal haploids in maize. Genetics 54, 453–464 (1966).
Barret, P., Brinkmann, M. & Beckert, M. A major locus expressed in the male gametophyte with incomplete penetrance is responsible for in situ gynogenesis in maize. Theor. Appl. Genet. 117, 581–594 (2008).
Hu, H. et al. The genetic basis of haploid induction in maize identified with a novel genome-wide association method. Genetics 202, 1267–1276 (2016).
Prigge, V. et al. New insights into the genetics of in vivo induction of maternal haploids, the backbone of doubled haploid technology in maize. Genetics 190, 781–793 (2012).
Gilles, L. M. et al. Loss of pollen‐specific phospholipase NOT LIKE DAD triggers gynogenesis in maize. EMBO J. 36, 707–717 (2017).
Kelliher, T. et al. MATRILINEAL, a sperm-specific phospholipase, triggers maize haploid induction. Nature 542, 105–109 (2017).
Liu, C. et al. A 4-bp insertion at ZmPLA1 encoding a putative phospholipase a generates haploid induction in maize. Mol. Plant 10, 520–522 (2017).
Zhong, Y. et al. Mutation of ZmDMP enhances haploid induction in maize. Nat. Plants 5, 575–580 (2019).
Ravi, M. & Chan, S. W. L. Haploid plants produced by centromere-mediated genome elimination. Nature 464, 615–618 (2010).
Tan, E. H. et al. Catastrophic chromosomal restructuring during genome elimination in plants. eLife 4, e06516 (2015).
Kelliher, T. et al. Maternal haploids are preferentially induced by CENH3-tailswap transgenic complementation in maize. Front. Plant Sci. 7, 414 (2016).
Maheshwari, S. et al. Naturally occurring differences in CENH3 affect chromosome segregation in zygotic mitosis of hybrids. PLoS Genet. 11, e1004970 (2015).
Karimi-Ashtiyani, R. et al. Point mutation impairs centromeric CENH3 loading and induces haploid plants. Proc. Natl Acad. Sci. USA 112, 11211–11216 (2015).
Kuppu, S. et al. Point mutations in centromeric histone induce post-zygotic incompatibility and uniparental inheritance. PLoS Genet. 11, e1005494 (2015).
Britt, A. B. & Kuppu, S. Cenh3: an emerging player in haploid induction technology. Front. Plant Sci. 7, 357 (2016).
Kuppu, S. et al. A variety of changes, including CRISPR/Cas9 mediated deletions, in CENH3 lead to haploid induction on outcrossing. Plant Biotech. J. (in the press).
van Dun, C., Lelivelt, C. & Movahedi, S. Non-transgenic haploid inducer lines in cucurbits. Worldwide patent WO/2017/081009A1 (2017).
Op Den Camp, R. H. M., Van Dijk, P. J. & Gallard, A. Method for the production of haploid and subsequent doubled haploid plants. Worldwide patent WO/2017/200386 (2017).
Bohra, A., Jha, U. C., Adhimoolam, P., Bisht, D. & Singh, N. P. Cytoplasmic male sterility (CMS) in hybrid breeding in field crops. Plant Cell Rep. 35, 967–993 (2016).
Li, L., Xu, X., Jin, W. & Chen, S. Morphological and molecular evidences for DNA introgression in haploid induction via a high oil inducer CAUHOI in maize. Planta 230, 367–376 (2009).
Zhang, Z. et al. Chromosome elimination and in vivo haploid production induced by Stock 6-derived inducer line in maize (Zea mays L.). Plant Cell Rep. 27, 1851–1860 (2008).
Zhao, X., Xu, X., Xie, H., Chen, S. & Jin, W. Fertilization and uniparental chromosome elimination during crosses with maize haploid inducers. Plant Physiol. 163, 721–731 (2013).
Qiu, F. et al. Morphological, cellular and molecular evidences of chromosome random elimination in vivo upon haploid induction in maize. Curr. Plant Biol. 1, 83–90 (2014).
Wedzony, M., Röber, F. & Geiger, H. Chromosome elimination observed in selfed progenies of maize inducer line RWS. in XVIIth International Congress on Sex Plant Reproduction 173 (2002).
Liu, L. et al. In vivo haploid induction leads to increased frequency of twin-embryo and abnormal fertilization in maize. BMC Plant Biol. 18, 313 (2018).
Bylich, V. & Chalyk, S. Existence of pollen grains with a pair of morphologically different sperm nuclei as a possible cause of the haploid-inducing capacity in ZMS line. Maize Genetics Cooperation Newsletter 70, 33 (1996).
Chalyk, S., Baumann, A., Daniel, G. & Eder, J. Aneuploidy as a possible cause of haploid-induction in maize. Maize Genetics Cooperation Newsletter 77, 29–30 (2003).
Tian, X. et al. Hetero-fertilization together with failed egg–sperm cell fusion supports single fertilization involved in in vivo haploid induction in maize. J. Exp. Bot. 69, 4689–4701 (2018).
Li, X. et al. Single nucleus sequencing reveals spermatid chromosome fragmentation as a possible cause of maize haploid induction. Nat. Commun. 8, 991 (2017).
Swapna, M. & Sarkar, K. R. Anomalous fertilization in haploidy inducer lines in maize (Zea mays L). Maydica 56, 1717 (2012).
Mahendru, A. & Sarkar, K. R. Cytological analysis of the pollen of haploidy inducer lines in maize (Zea mays L.). Indian. J. Genet. Pl. Br. 60, 37–43 (2000).
Jackson, D. No sex please, we’re (in)breeding. EMBO J. 36, 703–704 (2017).
Cyprys, P., Lindemeier, M. & Sprunck, S. Gamete fusion is facilitated by two sperm cell-expressed DUF679 membrane proteins. Nat. Plants 5, 253–257 (2019).
Takahashi, T. et al. The male gamete membrane protein DMP9/DAU2 is required for double fertilization in flowering plants. Development 145, dev170076 (2018).
Liu, C. et al. Fine mapping of qhir8 affecting in vivo haploid induction in maize. Theor. Appl. Genet. 128, 2507–2515 (2015).
Schlupp, I. The evolutionary ecology of gynogenesis. Annu. Rev. Ecol. Evol. Syst. 36, 399–417 (2005).
Watts, A., Kumar, V. & Bhat, S. R. Centromeric histone H3 protein: from basic study to plant breeding applications. J. Plant Biochem. Biotechnol. 25, 339–348 (2016).
Wang, N. & Dawe, R. K. Centromere size and its relationship to haploid formation in plants. Mol. Plant 11, 398–406 (2018).
Comai, L. & Tan, E. H. Haploid induction and genome instability. Trends Genet. 35, 791–803 (2019).
Sanei, M., Pickering, R., Kumke, K., Nasuda, S. & Houben, A. Loss of centromeric histone H3 (CENH3) from centromeres precedes uniparental chromosome elimination in interspecific barley hybrids. Proc. Natl Acad. Sci. USA 108, E498–E505 (2011).
Gao, C. The future of CRISPR technologies in agriculture. Nat. Rev. Mol. Cell Biol. 19, 275–276 (2018).
Kelliher, T. et al. One-step genome editing of elite crop germplasm during haploid induction. Nat. Biotech. 37, 287–292 (2019).
Wang, B. et al. Development of a Haploid-inducer mediated genome editing system for accelerating maize breeding. Mol. Plant 12, 597–602 (2019).
d’Erfurth, I. et al. Turning meiosis into mitosis. PLoS Biol. 7, e1000124 (2009).
Mieulet, D. et al. Turning rice meiosis into mitosis. Cell Res. 26, 1242–1254 (2016).
Wang, C. et al. Clonal seeds from hybrid rice by simultaneous genome engineering of meiosis and fertilization genes. Nat. Biotech. 37, 283 (2019).
Qu, Y. et al. Mapping of QTL for kernel abortion caused by in vivo haploid induction in maize (Zea mays L.). PLoS ONE 15, e0228411 (2020).
Yao, L. et al. OsMATL mutation induces haploid seed formation in indica rice. Nat. Plants 4, 530–533 (2018).
Liu, C. et al. Extension of the in vivo haploid induction system from diploid maize to hexaploid wheat. Plant Biotechnol. J. 18, 316–318 (2020).
Liu, H. et al. Efficient induction of haploid plants in wheat by editing of TaMTL using an optimized Agrobacterium-mediated CRISPR system. J. Exp. Bot. 71, 1337–1349 (2020).
Prasanna, B., Chaikam, V. & Mahuku, G. Doubled Haploid Technology in Maize Breeding: Theory and Practice (CIMMYT, 2012).
Chaikam, V., Gowda, M., Nair, S. K., Melchinger, A. E. & Boddupalli, P. M. Genome-wide association study to identify genomic regions influencing spontaneous fertility in maize haploids. Euphytica 215, 138 (2019).
Ren, J., Wu, P., Tian, X., Lübberstedt, T. & Chen, S. QTL mapping for haploid male fertility by a segregation distortion method and fine mapping of a key QTL qhmf4 in maize. Theor. Appl. Genet. 130, 1349–1359 (2017).
Yang, J. et al. Genetic dissection of haploid male fertility in maize (Zea mays L.). Plant Breeding 138, 259–265 (2019).
Ren, J. et al. Mapping of QTL and identification of candidate genes conferring spontaneous haploid genome doubling in maize (Zea mays L.). Plant Sci. 293, 110337 (2020).
Boerman, N. A., Frei, U. K. & Lübberstedt, T. Impact of spontaneous haploid genome doubling in maize breeding. Plants 9, 369 (2020).
Begheyn, R. F., Vangsgaard, K., Roulund, N. & Studer, B. In Breeding in a World of Scarcity (eds Roldán-Ruiz, I. et al.) 151–155 (Springer, 2016).
Dong, Y.-Q. et al. Androgenesis, gynogenesis, and parthenogenesis haploids in cucurbit species. Plant Cell Rep. 35, 1991–2019 (2016).
Hussain, T. & Franks, C. in Sorghum Vol. 1931 (eds Zhao, Z.-Y. & Dahlberg, J.) 49–59 (Springer, 2019).
Kloiber-Maitz, M., Wieckhorst, S., Bolduan, C. & Ouzunova, M. Haploidisierung in sorghum. European patent EP3366778A1 (2018).
Acknowledgements
We apologize to authors whose research could not be discussed here due to space restrictions. We thank M. Bodin and M. Philippot for their help with Table 1. We also thank J. Xicluna and O. Hamant for the critical reading of the abstract. This research was supported by the ANR grant (no. ANR-19-CE20-0012) to T.W., and by pack ambition recherche from the Région Auvergne-Rhone-Alpes (HD-INNOV) to T.W. N.M.A.J. and L.M.G. were supported by CIFRE PhD fellowships from ANRT funding agency (grant nos. 2019/0771 and 2015/0777, respectively).
Author information
Authors and Affiliations
Contributions
T.W. led the writing of the manuscript. N.M.A.J., D.E.P. and T.W. contributed to the critical reading of the manuscript, provided suggestions and contributed to the writing of specific sections. L.M.G., P.M.R. and J.-P.M. contributed to the critical reading of the manuscript and provided suggestions. L.M.G. composed the figures with help from N.M.A.J. T.W. initiated and coordinated the project.
Corresponding author
Ethics declarations
Competing interests
N.M.A.J, L.M.G. and J.-P.M. are employees of LIMAGRAIN Europe. The authors also have pending patent applications (PCT/EP2016/060202 (published as WO2016177887) and EP3091076).
Additional information
Peer review information Nature Plants thanks Andreas Houben, Weicai Yang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jacquier, N.M.A., Gilles, L.M., Pyott, D.E. et al. Puzzling out plant reproduction by haploid induction for innovations in plant breeding. Nat. Plants 6, 610–619 (2020). https://doi.org/10.1038/s41477-020-0664-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-020-0664-9
This article is cited by
-
Fine mapping of major QTL qshgd1 for spontaneous haploid genome doubling in maize (Zea mays L.)
Theoretical and Applied Genetics (2024)
-
Current approaches and future potential for delivering CRISPR/Cas components in oilseeds and millets
The Nucleus (2024)
-
Protocol optimization and assessment of genotypic response for inbred line development through doubled haploid production in maize
BMC Plant Biology (2023)
-
A simple and highly efficient strategy to induce both paternal and maternal haploids through temperature manipulation
Nature Plants (2023)
-
Heat stress promotes haploid formation during CENH3-mediated genome elimination in Arabidopsis
Plant Reproduction (2023)