Abstract
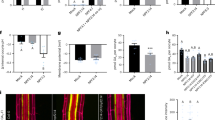
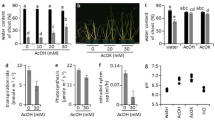
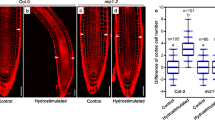
Plants are able to sense a rise in temperature of several degrees, and appropriately adapt their metabolic and growth processes. To this end, plants produce various signalling molecules that act throughout the plant body. Here, we report that root-derived GA12, a precursor of the bioactive gibberellins, mediates thermo-responsive shoot growth in Arabidopsis. Our data suggest that root-to-shoot translocation of GA12 enables a flexible growth response to ambient temperature changes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in the published article and its Supplementary Information.
References
Quint, M. et al. Molecular and genetic control of plant thermomorphogenesis. Nat. Plants 2, 15190 (2016).
Martins, S. et al. Brassinosteroid signaling-dependent root responses to prolonged elevated ambient temperature. Nat. Commun. 8, 309 (2017).
Kumar, S. V. et al. Transcription factor PIF4 controls the thermosensory activation of flowering. Nature 484, 242–245 (2012).
Davière, J.-M. & Achard, P. Gibberellin signaling in plants. Development 140, 1147–1151 (2013).
Achard, P. et al. Integration of plant responses to environmentally activated phytohormonal signals. Science 311, 91–94 (2006).
Colebrook, E. H., Thomas, S. G., Phillips, A. L. & Hedden, P. The role of gibberellin signaling in plant responses to abiotic stress. J. Exp. Bot. 217, 67–75 (2014).
Stavang, J. A. et al. Hormonal regulation of temperature-induced growth in Arabidopsis. Plant J. 60, 589–601 (2009).
Bai, L., Deng, H., Zhang, X., Yu, X. & Li, Y. Gibberellin is involved in inhibition of cucumber growth and nitrogen uptake at suboptimal root-zone temperature. PLoS ONE 11, e0156188 (2016).
Regnault, T. et al. The gibberellin precursor GA12 acts as a long-distance growth signal in Arabidopsis. Nat. Plants 1, 15073 (2015).
Binenbaum, J., Weinstain, R. & Shani, E. Gibberellin localization and transport in plants. Trends Plant Sci. 23, 410–421 (2018).
Hedden, P. & Thomas, S. G. Gibberellin biosynthesis and its regulation. Biochem. J. 444, 11–25 (2012).
Gaymard, F. et al. Identification and disruption of a plant shaker-like outward channel involved in K+ release into the xylem sap. Cell 94, 647–655 (1998).
de Lucas, M. et al. A molecular framework for light and gibberellin control of cell elongation. Nature 451, 480–484 (2008).
Regnault, T., Davière, J.-M., Heintz, D., Lange, T. & Achard, P. The gibberellin biosynthetic genes AtKAO1 and AtKAO2 have overlapping roles throughout Arabidopsis development. Plant J. 80, 462–474 (2014).
Hwang, I. & Goodman, H. M. An Arabidopsis thaliana root-specific kinase homolog is induced by dehydration, ABA, and NaCl. Plant J. 8, 37–43 (1995).
Osugi, A. et al. Systemic transport of trans-zeatin and its precursor have differing roles in Arabidopsis shoots. Nat. Plants 3, 17112 (2017).
Saito, H. et al. The jasmonate-responsive GTR1 transporter is required for gibberellin-mediated stamen development in Arabidopsis. Nat. Commun. 6, 6095 (2015).
Tal, I. et al. The Arabidopsis NPF3 protein is a GA transporter. Nat. Commun. 7, 11486 (2016).
Reid, D. M., Crozier, A. & Harvey, B. M. The effects of flooding on the export of gibberellins from the root to the shoot. Planta 89, 346–379 (1969).
Lavender, D. P., Sweet, G. B., Zaerr, J. B. & Hermann, R. K. Spring shoot growth in Douglas fir may be initiated by gibberellins exported from the roots. Science 182, 838–839 (1973).
Nakamura, S. et al. Gateway binary vectors with the bialaphos resistance gene, bar, as a selection marker for plant transformation. Biosci. Biotechnol. Biochem. 74, 1315–1319 (2010).
Karimi, M., Inzé, D. & Depicker, A. GATEWAY vectors for Agrobacterium-mediated plant transformation. Trends Plant Sci. 7, 193–195 (2002).
Turnbull, C. G. N., Booker, J. P. & Leyser, O. Micrografting techniques for testing long-distance signalling in Arabidopsis. Plant J. 32, 255–262 (2002).
Lange, T. et al. Gibberellin biosynthesis in developing pumpkin seedlings. Plant Physiol. 139, 213–223 (2005).
Acknowledgements
We thank T.P. Sun for providing seeds of ga1-3 (Col-0 background) and P. Hedden for providing ga20ox1-2-3. This work was supported by the Centre National de la Recherche Scientifique and the French Ministry of Research and Higher Education.
Author information
Authors and Affiliations
Contributions
L.C., T.R., L.S.-A., E.C., J.Z., D.H., N.L., M.J.P.L., T.L., J.-M.D. and P.A. performed experimental work. L.C., T.R., D.H., N.L., M.J.P.L., T.L., J.-M.D. and P.A. designed the experiments. M.S., M.J.P.L., T.L., J.-M.D. and P.A. realised the figures and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Discussion and Supplementary Figures 1–5.
Supplementary Tables
Supplementary Tables 1–5.
Supplementary Dataset 1
Statistics (ANOVA and t-test), P values.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Supplementary Fig. 1
Statistical source data.
Source Data Supplementary Fig. 2
Statistical source data.
Source Data Supplementary Fig. 3
Statistical source data.
Source Data Supplementary Fig. 4
Statistical source data.
Source Data Fig. 2d
Unprocessed blots.
Source Data Supplementary Fig. 4b
Unprocessed blots.
Rights and permissions
About this article
Cite this article
Camut, L., Regnault, T., Sirlin-Josserand, M. et al. Root-derived GA12 contributes to temperature-induced shoot growth in Arabidopsis. Nat. Plants 5, 1216–1221 (2019). https://doi.org/10.1038/s41477-019-0568-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-019-0568-8
This article is cited by
-
Gibberellin and abscisic acid transporters facilitate endodermal suberin formation in Arabidopsis
Nature Plants (2023)
-
McMYB4 improves temperature adaptation by regulating phenylpropanoid metabolism and hormone signaling in apple
Horticulture Research (2021)
-
Cut and paste: temperature-enhanced cotyledon micrografting for Arabidopsis thaliana seedlings
Plant Methods (2020)