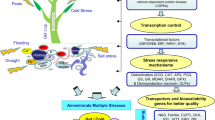
Abstract
A major problem facing humanity is that our numbers are growing but the availability of land and fresh water for agriculture is not. This problem is being exacerbated by climate change-induced increases in drought, and other abiotic stresses. Stress-resistant crops are needed to ensure yield stability under stress conditions and to minimize the environmental impacts of crop production. Evolution has created thousands of species of naturally stress-resistant plants (NSRPs), some of which have already been subjected to human domestication and are considered minor crops. Broader cultivation of these minor crops will diversify plant agriculture and the human diet, and will therefore help improve global food security and human health. More research should be directed toward understanding and utilizing NSRPs. Technologies are now available that will enable researchers to rapidly improve the genetics of NSRPs, with the goal of increasing NSRP productivity while retaining NSRP stress resistance and nutritional value.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Foley, J. A. et al. Solutions for a cultivated planet. Nature 478, 337–342 (2011).
Godfray, H. C. et al. Food security: the challenge of feeding 9 billion people. Science 327, 812–818 (2010).
Shinozaki, K., Uemura, M., Bailey-Serres, J., Bray, E. A. & Weretilnyk, E. in Biochemistry and Molecular Biology of Plants (eds Buchanan, B. B., Gruissem, W. & Jones, R. L.) Ch. 22, 1051–1100 (Wiley, Chichester, 2015).
Cai, W. J. et al. Increasing frequency of extreme El Nino events due to greenhouse warming. Nat. Clim. Change 4, 111–116 (2014).
Dai, A. G. Increasing drought under global warming in observations and models. Nat. Clim. Change 3, 52–58 (2013).
Battisti, D. S. & Naylor, R. L. Historical warnings of future food insecurity with unprecedented seasonal heat. Science 323, 240–244 (2009).
Pingali, P. L. Green revolution: impacts, limits, and the path ahead. Proc. Natl Acad. Sci. USA 109, 12302–12308 (2012).
FAOSTAT (FAO, 2017); http://www.fao.org/faostat/en/#data/QC
Ray, D. K., Ramankutty, N., Mueller, N. D., West, P. C. & Foley, J. A. Recent patterns of crop yield growth and stagnation. Nat. Commun. 3, 1293 (2012).
Alston, J. M., Beddow, J. M. & Pardey, P. G. Agricultural research, productivity, and food prices in the long run. Science 325, 1209–1210 (2009).
Lobell, D. B. et al. Greater sensitivity to drought accompanies maize yield increase in the U. S. Midwest. Science 344, 516–519 (2014).
Li, H., Rasheed, A., Hickey, L. T. & He, Z. Fast-forwarding genetic gain. Trends Plant Sci. 23, 184–186 (2018).
Zhu, Y. et al. Genetic diversity and disease control in rice. Nature 406, 718–722 (2000).
Gilliham, M., Able, J. A. & Roy, S. J. Translating knowledge about abiotic stress tolerance to breeding programmes. Plant J. 90, 898–917 (2017).
Zheng, P. et al. A phenylalanine in DGAT is a key determinant of oil content and composition in maize. Nat. Genet. 40, 367–372 (2008).
Dwivedi, S. L. et al. Landrace germplasm for improving yield and abiotic stress adaptation. Trends Plant Sci. 21, 31–42 (2016).
Munns, R. et al. Wheat grain yield on saline soils is improved by an ancestral Na+ transporter gene. Nat. Biotechnol. 30, 360–364 (2012).
Tardieu, F. Any trait or trait-related allele can confer drought tolerance: just design the right drought scenario. J. Exp. Bot. 63, 25–31 (2012).
Mittler, R. & Blumwald, E. Genetic engineering for modern agriculture: challenges and perspectives. Annu. Rev. Plant Biol. 61, 443–462 (2010).
Cooper, M., van Eeuwijk, F. A., Hammer, G. L., Podlich, D. W. & Messina, C. Modeling QTL for complex traits: detection and context for plant breeding. Curr. Opin. Plant Biol. 12, 231–240 (2009).
Tardieu, F. & Tuberosa, R. Dissection and modelling of abiotic stress tolerance in plants. Curr. Opin. Plant Biol. 13, 206–212 (2010).
Zhu, J. K. Abiotic stress signaling and responses in plants. Cell 167, 313–324 (2016).
Hu, H. & Xiong, L. Genetic engineering and breeding of drought-resistant crops. Annu. Rev. Plant Biol. 65, 715–741 (2014).
Roy, S. J., Negrao, S. & Tester, M. Salt resistant crop plants. Curr. Opin. Biotechnol. 26, 115–124 (2014).
Rothstein, S. J., Bi, Y. M., Coneva, V., Han, M. & Good, A. The challenges of commercializing second-generation transgenic crop traits necessitate the development of international public sector research infrastructure. J. Exp. Bot. 65, 5673–5682 (2014).
GM crops: a story in numbers. Nature 497, 22–23 (2013).
Petitions for Determination of Nonregulated Status (USDA, 2018); https://www.aphis.usda.gov/aphis/ourfocus/biotechnology/permits-notifications-petitions/petitions/petition-status
Amtmann, A. Learning from evolution: Thellungiella generates new knowledge on essential and critical components of abiotic stress tolerance in plants. Mol. Plant 2, 3–12 (2009).
Reef, R. & Lovelock, C. E. Regulation of water balance in mangroves. Ann. Bot. 115, 385–395 (2015).
Munns, R. & Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 59, 651–681 (2008).
Brown, G. W. Desert Biology Special Topics on the Physical and Biological Aspects of Arid Regions (Elsevier, Burlington, 2013).
Gechev, T. S., Dinakar, C., Benina, M., Toneva, V. & Bartels, D. Molecular mechanisms of desiccation tolerance in resurrection plants. Cell. Mol. Life Sci. 69, 3175–3186 (2012).
Gaff, D. F. & Oliver, M. The evolution of desiccation tolerance in angiosperm plants: a rare yet common phenomenon. Funct. Plant Biol. 40, 315–328 (2013).
Porembski, S. in Plant Desiccation Tolerance (eds Luttge, U. Beck, E. & Bartels, D.) 139–156 (Blackwell Publishing, Oxford, 2011).
Mitra, J., Xu, G., Wang, B., Li, M. & Deng, X. Understanding desiccation tolerance using the resurrection plant Boea hygrometrica as a model system. Front. Plant Sci. 4, 446 (2013).
Griffiths, C. A., Gaff, D. F. & Neale, A. D. Drying without senescence in resurrection plants. Front. Plant Sci. 5, 36 (2014).
Williams, B. et al. Trehalose accumulation triggers autophagy during plant desiccation. PLoS Genet. 11, e1005705 (2015).
Asami, P., Mundree, S. & Williams, B. Saving for a rainy day: control of energy needs in resurrection plants. Plant Sci. 271, 62–66 (2018).
VanBuren, R. et al. Single-molecule sequencing of the desiccation-tolerant grass Oropetium thomaeum. Nature 527, 508–511 (2015).
Xiao, L. et al. The resurrection genome of Boea hygrometrica: a blueprint for survival of dehydration. Proc. Natl Acad. Sci. USA 112, 5833–5837 (2015).
Costa, M. D. et al. A footprint of desiccation tolerance in the genome of Xerophyta viscosa. Nat. Plants 3, 17038 (2017).
VanBuren, R. et al. Seed desiccation mechanisms co-opted for vegetative desiccation in the resurrection grass Oropetium thomaeum. Plant Cell Environ. 40, 2292–2306 (2017).
Farrant, J. M. & Moore, J. P. Programming desiccation-tolerance: from plants to seeds to resurrection plants. Curr. Opin. Plant Biol. 14, 340–345 (2011).
Land and Water (FAO, 2018); http://www.fao.org/land-water/en/
Flowers, T. J., Galal, H. K. & Bromham, L. Evolution of halophytes: multiple origins of salt tolerance in land plants. Funct. Plant Biol. 37, 604–612 (2010).
Flowers, T. J., Santos, J., Jahns, M., Warburton, B. & Reed, P. eHALOPH - Halophytes Database (Univ. Sussex, 2018); https://www.sussex.ac.uk/affiliates/halophytes/
Bromham, L. Macroevolutionary patterns of salt tolerance in angiosperms. Ann. Bot. 115, 333–341 (2015).
Bromham, L. & Bennett, T. H. Salt tolerance evolves more frequently in C4 grass lineages. J. Evol. Biol. 27, 653–659 (2014).
Flowers, T. J. & Colmer, T. D. Salinity tolerance in halophytes. New. Phytol. 179, 945–963 (2008).
Inan, G. et al. Salt cress. A halophyte and cryophyte Arabidopsis relative model system and its applicability to molecular genetic analyses of growth and development of extremophiles. Plant Physiol. 135, 1718–1737 (2004).
Amtmann, A., Bohnert, H. J. & Bressan, R. A. Abiotic stress and plant genome evolution. Search for new models. Plant Physiol. 138, 127–130 (2005).
Oh, D. H. et al. Loss of halophytism by interference with SOS1 expression. Plant Physiol. 151, 210–222 (2009).
Yang, R. et al. The reference genome of the halophytic plant Eutrema salsugineum. Front. Plant Sci. 4, 46 (2013).
Wu, H. J. et al. Insights into salt tolerance from the genome of Thellungiella salsuginea. Proc. Natl Acad. Sci. USA 109, 12219–12224 (2012).
Dassanayake, M. et al. The genome of the extremophile crucifer Thellungiella parvula. Nat. Genet. 43, 913–918 (2011).
Plant Uses / Edible (PFAF); https://pfaf.org/User/edibleuses.aspx
Proches, S., Wilson, J. R. U., Vamosi, J. C. & Richardson, D. M. Plant diversity in the human diet: weak phylogenetic signal indicates breadth. Bioscience 58, 151–159 (2008).
Biodiversity: Plants (FAO); http://www.fao.org/biodiversity/components/plants/en/
Miller, N. F., Spengler, R. N. & Frachetti, M. Millet cultivation across Eurasia: Origins, spread, and the influence of seasonal climate. Holocene 26, 1566–1575 (2016).
Goron, T. L. & Raizada, M. N. Genetic diversity and genomic resources available for the small millet crops to accelerate a New Green Revolution. Front. Plant Sci. 6, 157 (2015).
Bazile, D., Jacobsen, S. E. & Verniau, A. The global expansion of quinoa: trends and limits. Front. Plant Sci. 7, 622 (2016).
Habiyaremye, C. et al. Proso Millet (Panicum miliaceum L.) and its potential for cultivation in the Pacific Northwest, U. S.: a review. Front. Plant Sci. 7, 1961 (2016).
Theisen, A. A., Knox, E. G., Mann, F. L., Sprague, H. B. (eds). Feasibility of Introducing Food Crops Better Adapted to Environmental Stress 2 (National Science Foundation, Directorate for Applied Science and Research Applications, Division of Applied Research, Washington DC, 1978; 168–172.
Miao, Z. Z. et al. Principal component analysis on traits related to yield and quality of hybrid millet. J. Shanxi. Agr. Sci. 41, 785–788 (2013).
Bennetzen, J. L. et al. Reference genome sequence of the model plant Setaria. Nat. Biotechnol. 30, 555–561 (2012).
Zhang, G. et al. Genome sequence of foxtail millet (Setaria italica) provides insights into grass evolution and biofuel potential. Nat. Biotechnol. 30, 549–554 (2012).
Jia, G. et al. A haplotype map of genomic variations and genome-wide association studies of agronomic traits in foxtail millet (Setaria italica). Nat. Genet. 45, 957–961 (2013).
Varshney, R. K. et al. Pearl millet genome sequence provides a resource to improve agronomic traits in arid environments. Nat. Biotechnol. 35, 969–976 (2017).
Hariadi, Y., Marandon, K., Tian, Y., Jacobsen, S. E. & Shabala, S. Ionic and osmotic relations in quinoa (Chenopodium quinoa Willd.) plants grown at various salinity levels. J. Exp. Bot. 62, 185–193 (2011).
Zou, C. et al. A high-quality genome assembly of quinoa provides insights into the molecular basis of salt bladder-based salinity tolerance and the exceptional nutritional value. Cell Res. 27, 1327–1340 (2017).
Yasui, Y. et al. Draft genome sequence of an inbred line of Chenopodium quinoa, an allotetraploid crop with great environmental adaptability and outstanding nutritional properties. DNA Res. 23, 535–546 (2016).
Jarvis, D. E. et al. The genome of Chenopodium quinoa. Nature 542, 307–312 (2017).
Clouse, J. W. et al. The Amaranth Genome: genome, transcriptome, and physical map assembly. Plant Genome. https://doi.org/10.3835/plantgenome2015.07.0062 (2016).
Yasui, Y. et al. Assembly of the draft genome of buckwheat and its applications in identifying agronomically useful genes. DNA Res. 23, 215–224 (2016).
Zhang, L. et al. The tartary buckwheat genome provides insights into rutin biosynthesis and abiotic stress tolerance. Mol. Plant 10, 1224–1237 (2017).
Ho, W. K. et al. Use of microsatellite markers for the assessment of bambara groundnut breeding system and varietal purity before genome sequencing. Genome 59, 427–431 (2016).
Castaneda-Alvarez, N. P. et al. Global conservation priorities for crop wild relatives. Nat. Plants 2, 16022 (2016).
Cardinale, B. J. et al. Biodiversity loss and its impact on humanity. Nature 486, 59–67 (2012).
Altieri, M. A., Nicholls, C. I., Henao, A. & Lana, M. A. Agroecology and the design of climate change-resilient farming systems. Agron. Sustain. Dev. 35, 869–890 (2015).
Garibaldi, L. A. et al. Farming approaches for greater biodiversity, livelihoods, and food security. Trends Ecol. Evol. 32, 68–80 (2017).
Rasmussen, C., Lagnaoui, A. & Esbjerg, P. Advances in the knowledge of quinoa pests. Food Rev. Intl. 19, 61–75 (2003).
Khoury, C. K. et al. Increasing homogeneity in global food supplies and the implications for food security. Proc. Natl Acad. Sci. USA 111, 4001–4006 (2014).
Cordain, L. et al. Origins and evolution of the Western diet: health implications for the 21st century. Am. J. Clin. Nutr. 81, 341–354 (2005).
Pilbeam, D. J. Breeding crops for improved mineral nutrition under climate change conditions. J. Exp. Bot. 66, 3511–3521 (2015).
Saleh, A. S. M., Zhang, Q., Chen, J. & Shen, Q. Millet grains: nutritional quality, processing, and potential health benefits. Compr. Rev. Food. Sci. F. 12, 281–295 (2013).
Nowak, V., Du, J. & Charrondiere, U. R. Assessment of the nutritional composition of quinoa (Chenopodium quinoa Willd.). Food Chem. 193, 47–54 (2016).
Martinez-Outschoorn, U. E., Peiris-Pages, M., Pestell, R. G., Sotgia, F. & Lisanti, M. P. Cancer metabolism: a therapeutic perspective. Nat. Rev. Clin. Oncol. 14, 11–31 (2017).
Maddocks, O. D. K. et al. Modulating the therapeutic response of tumours to dietary serine and glycine starvation. Nature 544, 372–376 (2017).
Knott, S. R. V. et al. Asparagine bioavailability governs metastasis in a model of breast cancer. Nature 554, 378–381 (2018).
Popkin, B. M., Adair, L. S. & Ng, S. W. Global nutrition transition and the pandemic of obesity in developing countries. Nutr. Rev. 70, 3–21 (2012).
Meyer, R. S. & Purugganan, M. D. Evolution of crop species: genetics of domestication and diversification. Nat. Rev. Genet. 14, 840–852 (2013).
Lenser, T. & Theissen, G. Molecular mechanisms involved in convergent crop domestication. Trends Plant Sci. 18, 704–714 (2013).
Hedden, P. The genes of the Green Revolution. Trends Genet. 19, 5–9 (2003).
The cost of sequencing a human genome. NIH https://www.genome.gov/sequencingcosts/ (2016).
Goodwin, S., McPherson, J. D. & McCombie, W. R. Coming of age: ten years of next-generation sequencing technologies. Nat. Rev. Genet. 17, 333–351 (2016).
Carvalho, A. B., Dupim, E. G. & Goldstein, G. Improved assembly of noisy long reads by k-mer validation. Genome Res. 26, 1710–1720 (2016).
IWGSC et al. Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 361, eaar7191 (2018).
Yin, K., Gao, C. & Qiu, J. L. Progress and prospects in plant genome editing. Nat. Plants 3, 17107 (2017).
Zhang, H., Zhang, J. S., Lang, Z. B., Botella, J. R. & Zhu, J. K. Genome editing-principles and applications for functional genomics research and crop improvement. Crit. Rev. Plant. Sci. 36, 291–309 (2017).
Miki, D., Zhang, W., Zeng, W., Feng, Z. & Zhu, J. K. CRISPR/Cas9-mediated gene targeting in Arabidopsis using sequential transformation. Nat. Commun. 9, 1967 (2018).
Hua, K., Tao, X., Yuan, F., Wang, D. & Zhu, J. K. Precise A.T to G.C base editing in the rice genome. Mol. Plant 11, 627–630 (2018).
Lowe, K. et al. Morphogenic regulators baby boom and wuschel improve monocot transformation. Plant Cell 28, 1998–2015 (2016).
Zhao, X. et al. Pollen magnetofection for genetic modification with magnetic nanoparticles as gene carriers. Nat. Plants 3, 956–964 (2017).
Cunningham, F. J., Goh, N. S., Demirer, G. S., Matos, J. L. & Landry, M. P. Nanoparticle-mediated delivery towards advancing plant genetic engineering. Trends Biotechnol. 36, 882–897 (2018).
Watson, A. et al. Speed breeding is a powerful tool to accelerate crop research and breeding. Nat. Plants 4, 23–29 (2018).
Borlaug, N. E. Nobel lecture: the Green Revolution, peace, and humanity. The Nobel Prize https://www.nobelprize.org/prizes/peace/1970/borlaug/lecture/ (1970).
Wang, W. et al. Cassava genome from a wild ancestor to cultivated varieties. Nat. Commun. 5, 5110 (2014).
Wang, M. et al. The genome sequence of African rice (Oryza glaberrima) and evidence for independent domestication. Nat. Genet. 46, 982–988 (2014).
Monat, C. et al. De novo assemblies of three Oryza glaberrima accessions provide first insights about pan-genome of African rices. Genome Biol. Evol. 9, 1–6 (2017).
Zhang, Y. et al. Genome and comparative transcriptomics of African wild rice Oryza longistaminata provide insights into molecular mechanism of rhizomatousness and self-incompatibility. Mol. Plant 8, 1683–1686 (2015).
Mondal, T. K., Rawal, H. C., Gaikwad, K., Sharma, T. R. & Singh, N. K. First de novo draft genome sequence of Oryza coarctata, the only halophytic species in the genus. Oryza. F1000Res 6, 1750 (2017).
Aversano, R. et al. The Solanum commersonii genome sequence provides insights into adaptation to stress conditions and genome evolution of wild potato relatives. Plant Cell 27, 954–968 (2015).
Ming, R. et al. The pineapple genome and the evolution of CAM photosynthesis. Nat. Genet. 47, 1435–1442 (2015).
Ma, T. et al. Genomic insights into salt adaptation in a desert poplar. Nat. Commun. 4, 2797 (2013).
Acknowledgements
We would like to thank H. Zhang and R. Bressan for careful reading and editing of the manuscript. Research funding was provided by the Chinese Academy of Sciences (CAS) to J.K.Z. and by Strategic Priority Research Program of the Chinese Academy of Sciences (XDB27040108), Shanghai Science and Technology Committee (17391900200), Youth Innovation Promotion Association of CAS (2014242), National Key R&D Program of China (2016YFA0503200) and CAS to H.Z.
Author information
Authors and Affiliations
Contributions
H.Z. and J.K.Z. wrote the paper; H.Z. and Y.L. analysed data.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, H., Li, Y. & Zhu, JK. Developing naturally stress-resistant crops for a sustainable agriculture. Nature Plants 4, 989–996 (2018). https://doi.org/10.1038/s41477-018-0309-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-018-0309-4
This article is cited by
-
Long-term organogenic callus cultivation of Ranunculus illyricus L.: a blueprint for sustainable ex situ conservation of the species in urban greenery
BMC Plant Biology (2024)
-
A review on plant-derived nanomaterials: an effective and innovative insect-resistant strategy for alternate pesticide development
International Journal of Environmental Science and Technology (2024)
-
Specific metabolic and cellular mechanisms of the vegetative desiccation tolerance in resurrection plants for adaptation to extreme dryness
Planta (2024)
-
Dynamics of chromatin accessibility and genome wide control of desiccation tolerance in the resurrection plant Haberlea rhodopensis
BMC Plant Biology (2023)
-
The impact of wind energy on plant biomass production in China
Scientific Reports (2023)