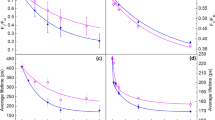
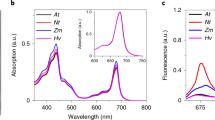
Abstract
Photosystem II of higher plants is protected against light damage by thermal dissipation of excess excitation energy, a process that can be monitored through non-photochemical quenching of chlorophyll fluorescence. When the light intensity is lowered, non-photochemical quenching largely disappears on a time scale ranging from tens of seconds to many minutes. With the use of picosecond fluorescence spectroscopy, we demonstrate that one of the underlying mechanisms is only functional when the reaction centre of photosystem II is closed, that is when electron transfer is blocked and the risk of photodamage is high. This is accompanied by the appearance of a long-wavelength fluorescence band. As soon as the reaction centre reopens, this quenching, together with the long-wavelength fluorescence, disappears instantaneously. This allows plants to maintain a high level of photosynthetic efficiency even in dangerous high-light conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Horton, P., Ruban, A. V. & Walters, R. G. Regulation of light harvesting in green plants. Annu. Rev. Plant Phys. 47, 655–684 (1996).
Demmig-Adams, B. & Adams, W. W. The role of xanthophyll cycle carotenoids in the protection of photosynthesis. Trends Plant Sci. 1, 21–26 (1996).
de Bianchi, S., Ballottari, M., Dall’Osto, L. & Bassi, R. Regulation of plant light harvesting by thermal dissipation of excess energy. Biochem. Soc. Trans. 38, 651–660 (2010).
Ruban, A. V., Johnson, M. P. & Duffy, C. D. The photoprotective molecular switch in the photosystem II antenna. Biochim. Biophys. Acta 1817, 167–181 (2012).
Li, X. P., Muller-Moule, P., Gilmore, A. M. & Niyogi, K. K. PsbS-dependent enhancement of feedback de-excitation protects photosystem II from photoinhibition. Proc. Natl Acad. Sci. USA 99, 15222–15227 (2002).
Belgio, E. et al. Economic photoprotection in photosystem II that retains a complete light-harvesting system with slow energy traps. Nat. Commun. 5, 4433 (2014).
Müller, P., Li, X.-P. & Niyogi, K. K. Non-photochemical quenching. A response to excess light energy. Plant Physiol. 125, 1558–1566 (2001).
Li, X. P. et al. A pigment-binding protein essential for regulation of photosynthetic light harvesting. Nature 403, 391–395 (2000).
Li, X. P. et al. Regulation of photosynthetic light harvesting involves intrathylakoid lumen pH sensing by the PsbS protein. J. Biol. Chem. 279, 22866–22874 (2004).
Demmig-Adams, B. Carotenoids and photoprotection in plants: A role for the xanthophyll zeaxanthin. Biochim. Biophys. Acta Bioenerg. 1020, 1–24 (1990).
Niyogi, K. K., Grossman, A. R. & Bjorkman, O. Arabidopsis mutants define a central role for the xanthophyll cycle in the regulation of photosynthetic energy conversion. Plant Cell 10, 1121–1134 (1998).
Belgio, E., Johnson, M. P., Juric, S. & Ruban, A. V. Higher plant photosystem II light-harvesting antenna, not the reaction center, determines the excited-state lifetime-both the maximum and the nonphotochemically quenched. Biophys. J. 102, 2761–2771 (2012).
Ruban, A. V. & Horton, P. Spectroscopy of non-photochemical and photochemical quenching of chlorophyll fluorescence in leaves; evidence for a role of the light harvesting complex of photosystem II in the regulation of energy dissipation. Photosynth. Res. 40, 181–190 (1994).
Horton, P., Johnson, M. P., Perez-Bueno, M. L., Kiss, A. Z. & Ruban, A. V. Photosynthetic acclimation: does the dynamic structure and macro-organisation of photosystem II in higher plant grana membranes regulate light harvesting states? FEBS J. 275, 1069–1079 (2008).
Niyogi, K. K., Li, X.-P., Rosenberg, V. & Jung, H.-S. Is PsbS the site of non-photochemical quenching in photosynthesis? J. Exp. Bot. 56, 375–382 (2005).
Avenson, T. J. et al. Zeaxanthin radical cation formation in minor light-harvesting complexes of higher plant antenna. J. Biol. Chem. 283, 3550–3558 (2008).
Holt, N. E. et al. Carotenoid cation formation and the regulation of photosynthetic light harvesting. Science 307, 433–436 (2005).
Miloslavina, Y. et al. Far-red fluorescence: a direct spectroscopic marker for LHCII oligomer formation in non-photochemical quenching. FEBS Lett. 582, 3625–3631 (2008).
Ruban, A. V. et al. Identification of a mechanism of photoprotective energy dissipation in higher plants. Nature 450, 575–578 (2007).
Bode, S. et al. On the regulation of photosynthesis by excitonic interactions between carotenoids and chlorophylls. Proc. Natl Acad. Sci. USA 106, 12311–12316 (2009).
Ahn, T. K. et al. Architecture of a charge-transfer state regulating light harvesting in a plant antenna protein. Science 320, 794–797 (2008).
Pascal, A. A. et al. Molecular basis of photoprotection and control of photosynthetic light-harvesting. Nature 436, 134–137 (2005).
Johnson, M. P. et al. Photoprotective energy dissipation involves the reorganization of photosystem II light-harvesting complexes in the grana membranes of spinach chloroplasts. Plant Cell 23, 1468–1479 (2011).
Xu, P., Tian, L., Kloz, M. & Croce, R. Molecular insights into zeaxanthin-dependent quenching in higher plants. Sci. Rep. 5, 13679 (2015).
Lambrev, P. H., Miloslavina, Y., Jahns, P. & Holzwarth, A. R. On the relationship between non-photochemical quenching and photoprotection of photosystem II. Biochim. Biophys. Acta 1817, 760–769 (2012).
Lambrev, P. H., Nilkens, M., Miloslavina, Y., Jahns, P. & Holzwarth, A. R. Kinetic and spectral resolution of multiple nonphotochemical quenching components in Arabidopsis leaves. Plant Physiol. 152, 1611–1624 (2010).
Kromdijk, J. et al. Improving photosynthesis and crop productivity by accelerating recovery from photoprotection. Science 354, 857–861 (2016).
Zhu, X. G., Ort, D. R., Whitmarsh, J. & Long, S. P. The slow reversibility of photosystem II thermal energy dissipation on transfer from high to low light may cause large losses in carbon gain by crop canopies: a theoretical analysis. J. Exp. Bot. 55, 1167–1175 (2004).
Holzwarth, A. R. & Jahns, P. in Non-Photochemical Quenching and Energy Dissipation in Plants, Algae and Cyanobacteria (eds Barbara Demmig-Adams, Gyozo Garab, William Adams III, & Govindjee) 129–156 (Springer, Dordrecht, the Netherlands, 2014).
Sylak-Glassman, E. J., Zaks, J., Amarnath, K., Leuenberger, M. & Fleming, G. R. Characterizing non-photochemical quenching in leaves through fluorescence lifetime snapshots. Photosynth. Res. 127, 69–76 (2016).
Holzwarth, A. R., Miloslavina, Y., Nilkens, M. & Jahns, P. Identification of two quenching sites active in the regulation of photosynthetic light-harvesting studied by time-resolved fluorescence. Chem. Phys. Lett. 483, 262–267 (2009).
Miloslavina, Y. et al. Ultrafast fluorescence study on the location and mechanism of non-photochemical quenching in diatoms. Biochim. Biophys. Acta Bioenerg. 1787, 1189–1197 (2009).
Wientjes, E., van Amerongen, H. & Croce, R. Quantum yield of charge separation in photosystem II: functional effect of changes in the antenna size upon light acclimation. J. Phys. Chem. B 117, 11200–11208 (2013).
Wientjes, E., van Stokkum, Ivo, H., van Amerongen, H. & Croce, R. The role of the individual Lhcas in photosystem I excitation energy trapping. Biophys. J. 101, 745–754 (2011).
Ruban, A. V., Dekker, J. P., Horton, P. & Grondelle, R. V. Temperature dependence of chlorophyll fluorescence from the light harvesting complex II of higher plants. Photochem. Photobiol. 61, 216–221 (1995).
Magdaong, N. M., Enriquez, M. M., LaFountain, A. M., Rafka, L. & Frank, H. A. Effect of protein aggregation on the spectroscopic properties and excited state kinetics of the LHCII pigment–protein complex from green plants. Photosynth. Res. 118, 259–276 (2013).
Chmeliov, J. et al. The nature of self-regulation in photosynthetic light-harvesting antenna. Nat. Plants 2, 16045 (2016).
Natali, A. et al. Light-harvesting complexes (LHCs) cluster spontaneously in membrane environment leading to shortening of their excited state lifetimes. J. Biol. Chem. 291, 16730–16739 (2016).
Lawton, W. H. & Sylvestre, E. A. Self modeling curve resolution. Technometrics 13, 617–633 (1971).
Trinkunas, G. et al. Exciton band structure in bacterial peripheral light-harvesting complexes. J. Phys. Chem. B 116, 5192–5198 (2012).
Chmeliov, J. et al. Excitons in the LH3 complexes from purple bacteria. J. Phys. Chem. B 117, 11058–11068 (2013).
van Oort, B. et al. Picosecond fluorescence of intact and dissolved PSI-LHCI crystals. Biophys. J. 95, 5851–5861 (2008).
Tian, L., Xu, P., Chukhutsina, V. U., Holzwarth, A. R. & Croce, R. Zeaxanthin-dependent nonphotochemical quenching does not occur in photosystem I in the higher plant Arabidopsis thaliana. Proc. Natl Acad. Sci. USA 114, 4828–4832 (2017).
Savikhin, S., Xu, W., Chitnis, P. R. & Struve, W. S. Ultrafast primary processes in PS I from Synechocystis sp. PCC 6803: roles of P700 and A(0). Biophys. J. 79, 1573–1586 (2000).
Wientjes, E. & Croce, R. PMS: photosystem I electron donor or fluorescence quencher. Photosynth. Res. 111, 185–191 (2012).
Wientjes, E., Philippi, J., Borst, J. W. & van Amerongen, H. Imaging the photosystem I/photosystem II chlorophyll ratio inside the leaf. Biochim. Biophys. Acta 1858, 259–265 (2017).
Croce, R., Zucchelli, G., Garlaschi, F. M., Bassi, R. & Jennings, R. C. Excited state equilibration in the photosystem I−light-harvesting I complex: P700 is almost isoenergetic with its antenna. Biochemistry 35, 8572–8579 (1996).
Bos, I. et al. Multiple LHCII antennae can transfer energy efficiently to a single photosystem I. Biochim. Biophys. Acta 1858, 371–378 (2017).
Chmeliov, J., Trinkunas, G., van Amerongen, H. & Valkunas, L. Light harvesting in a fluctuating antenna. J. Am. Chem. Soc. 136, 8963–8972 (2014).
Chmeliov, J., Trinkunas, G., van Amerongen, H. & Valkunas, L. Excitation migration in fluctuating light-harvesting antenna systems. Photosynth. Res. 127, 49–60 (2016).
Farooq, S., Chmeliov, J., Trinkunas, G., Valkunas, L. & van Amerongen, H. Is there excitation energy transfer between different layers of stacked photosystem-II-containing thylakoid membranes? J. Phys. Chem. Lett. 7, 1406–1410 (2016).
Mller, P. The internal conversion rate of the primary donor in reaction centers of Rhodobacter sphaeroides. Ber. Bunsen. Phys. Chem. 100, 1967–1973 (1996).
Vass, I. Role of charge recombination processes in photodamage and photoprotection of the photosystem II complex. Physiol. Plant. 142, 6–16 (2011).
Mozzo, M., Dall’Osto, L., Hienerwadel, R., Bassi, R. & Croce, R. Photoprotection in the antenna complexes of photosystem II: role of individual xanthophylls in chlorophyll triplet quenching. J. Biol. Chem. 283, 6184–6192 (2008).
Peterman, E. J., Dukker, F. M., van Grondelle, R. & van Amerongen, H. Chlorophyll a and carotenoid triplet states in light-harvesting complex II of higher plants. Biophys. J. 69, 2670–2678 (1995).
Peterman, E. J. G. et al. Xanthophylls in light-harvesting complex II of higher plants: light harvesting and triplet quenching. Biochemistry 36, 12208–12215 (1997).
Gruber, J. M., Chmeliov, J., Krger, T. P., Valkunas, L. & van Grondelle, R. Singlet-triplet annihilation in single LHCII complexes. Phys. Chem. Chem. Phys. 17, 19844–19853 (2015).
Su, X. et al. Structure and assembly mechanism of plant C2S2M2-type PSII-LHCII supercomplex. Science 357, 815–820 (2017).
van Oort, B. et al. Ultrafast resonance energy transfer from a site-specifically attached fluorescent chromophore reveals the folding of the N-terminal domain of CP29. Chem. Phys. 357, 113–119 (2009).
van Stokkum, I. H. M. et al. (Sub)-picosecond spectral evolution of fluorescence in photoactive proteins studied with a synchroscan streak camera system. Photochem. Photobiol. 82, 380–388 (2006).
Barzda, V. et al. Singlet-singlet annihilation kinetics in aggregates and trimers of LHCII. Biophys. J. 80, 2409–2421 (2001).
Krüger, T. P. J., Novoderezhkin, V. I., Ilioaia, C. & van Grondelle, R. Fluorescence spectral dynamics of single LHCII trimers. Biophys. J. 98, 3093–3101 (2010).
Kulheim, C., Agren, J. & Jansson, S. Rapid regulation of light harvesting and plant fitness in the field. Science 297, 91–93 (2002).
Berry, M. W., Murray, B., Langville, A. N., Pauca, V. P. & Robert, J. P. Algorithms and applications for approximate nonnegative matrix factorization. Comput. Stat. Data Anal. 52, 155–173 (2006).
Acknowledgements
The authors thank J. Philippi for the design and construction of the moving cuvette and actinic lighting system and R. Croce for critically reading the manuscript. H.v.A. and S.F. received financial support from the Foundation for Fundamental Research on Matter (FOM), which is part of the Netherlands Organization for Scientific Research (NWO). E.W. acknowledges funding from a Marie Sklodowska-Curie fellowship (655542; E.W.) and a Veni grant (016.161.038; E.W.) from the NWO, Earth and Life Sciences (ALW). J.C. and L.V. were supported by the Research Council of Lithuania (LMT grant no. MIP-080/2015).
Author information
Authors and Affiliations
Contributions
H.v.A. devised the project. S.F. (with assistance of R.K., E.W. and A.B.) performed the measurements. J.C., H.v.A., S.F. and E.W. analysed the data (with significant input from L.V., G.T. and R.K.). H.v.A., S.F. and J.C. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Table 1 and 2, and Supplementary Figures 1–4
Rights and permissions
About this article
Cite this article
Farooq, S., Chmeliov, J., Wientjes, E. et al. Dynamic feedback of the photosystem II reaction centre on photoprotection in plants. Nature Plants 4, 225–231 (2018). https://doi.org/10.1038/s41477-018-0127-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-018-0127-8
This article is cited by
-
Computational dissection of genetic variation modulating the response of multiple photosynthetic phenotypes to the light environment
BMC Genomics (2024)
-
Excitation transfer and quenching in photosystem II, enlightened by carotenoid triplet state in leaves
Photosynthesis Research (2024)
-
Identification of multiple nonphotochemical quenching processes in the extremophilic red alga Cyanidioschyzon merolae
Photosynthesis Research (2022)
-
The PsbS protein and low pH are necessary and sufficient to induce quenching in the light-harvesting complex of plants LHCII
Scientific Reports (2021)
-
Variable fluorescence of closed photochemical reaction centers
Photosynthesis Research (2020)