Abstract
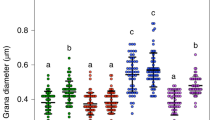
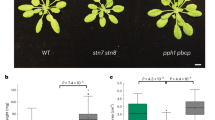
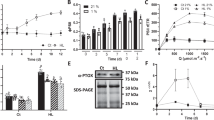
Upon transition of plants from darkness to light the initiation of photosynthetic linear electron transfer (LET) from H2O to NADP+ precedes the activation of CO2 fixation, creating a lag period where cyclic electron transfer (CET) around photosystem I (PSI) has an important protective role. CET generates ΔpH without net reduced NADPH formation, preventing overreduction of PSI via regulation of the cytochrome b 6 f (cytb 6 f) complex and protecting PSII from overexcitation by inducing non-photochemical quenching. The dark-to-light transition also provokes increased phosphorylation of light-harvesting complex II (LHCII). However, the relationship between LHCII phosphorylation and regulation of the LET/CET balance is not understood. Here, we show that the dark-to-light changes in LHCII phosphorylation profoundly alter thylakoid membrane architecture and the macromolecular organization of the photosynthetic complexes, without significantly affecting the antenna size of either photosystem. The grana diameter and number of membrane layers per grana are decreased in the light while the number of grana per chloroplast is increased, creating a larger contact area between grana and stromal lamellae. We show that these changes in thylakoid stacking regulate the balance between LET and CET pathways. Smaller grana promote more efficient LET by reducing the diffusion distance for the mobile electron carriers plastoquinone and plastocyanin, whereas larger grana enhance the partition of the granal and stromal lamellae plastoquinone pools, enhancing the efficiency of CET and thus photoprotection by non-photochemical quenching.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
29 May 2018
In the version of this Article originally published, the authors incorrectly labelled the timescale in Fig. 6b as milliseconds (ms) on the x axis and the indicated half-life values; the correct units are microseconds (μs). The figure has now been amended in all versions of the Article.
References
Horton, P. in Photosynthetic Mechanisms and the Environment (eds Barber, J. & Baker, N. R.) 135–187 (Elsevier, Amsterdam, 1985).
Robinson, S. P., Walker, D. A. The control of 3-phosphoglycerate reduction in isolated chloroplasts by the concentrations of ATP, ADP and 3-phosphoglycerate. Biochim. Biophys. Acta 545, 528–536 (1979).
Slovacek, R. E., Crowther, D. & Hind, G. Relative activities of linear and cyclic electron flows during chloroplast CO2 fixation. Biochim. Biophys. Acta 592, 495–505 (1980).
Joliot, P. & Joliot, A. Quantification of cyclic and linear flows in plants. Proc. Natl Acad. Sci. USA 102, 4913–4918 (2005).
Yamori, W. & Shikanai, T. Physiological functions of cyclic electron transport around photosystem I in sustaining photosynthesis and plant growth. Annu. Rev. Plant Biol. 67, 81–106 (2016).
Johnson, G. N. Physiology of PSI cyclic electron transport in higher plants. Biochim. Biophys. Acta 1807, 384–389 (2011).
Soursa, M. et al. Proton gradient regulation 5 is essential for proper acclimation of Arabidopsis photosystem I to naturally and artificially fluctuating light conditions. Plant Cell 24, 2394–2948 (2012).
Munekage, Y. et al. Cyclic electron flow around photosystem I is essential for photosynthesis. Nature 429, 579–582 (2004).
Herte, A. P. et al. PGRL1 is the elusive ferredoxin–plastoquinone reductase in photosynthetic cyclic electron flow. Mol. Cell. 49, 511–523 (2013).
Avenson, T. J., Cruz, J. A., Kanazawa, A. & Kramer, D. M. Regulating the proton budget of plant photosynthesis. Proc. Natl Acad. Sci. USA 102, 9709–9713 (2005).
Strand, D., Fisher, N. & Kramer, D. M. The higher plant plastid NAD(P)H dehydrogenase-like complex (NDH) is a high efficiency proton pump that increases ATP production by cyclic electron flow. J. Biol. Chem. 292, 11850–11860 (2017).
Yamori, W., Shikanai, T. & Makino, A. Photosystem I cyclic electron ow via chloroplast NADH dehydrogenase-like complex performs a physiological role for photosynthesis at low light. Sci. Rep. 5, 13908 (2015).
Iwai, M. et al. Isolation of the elusive supercomplex that drives cyclic electron flow in photosynthesis. Nature 464, 1210–1213 (2010).
Takahashi, H., Clowez, S., Wollman, F. A., Vallon, O. & Rappaport, F. Cyclic electron flow is redox-controlled but independent of state transition. Nat. Commun. 4, 1954 (2013).
Joliot, P., Lavergne, J. & Beal, D. Plastoquinone compartmentation in chloroplasts. I. Evidence for domains with different rates of photo-reduction. Biochim. Biophys. Acta 1101, 1–12 (1992).
Dumas, L., Chazaux, M., Peltier, G., Johnson, X. & Alric, J. Cytochrome b 6 f function and localization, phosphorylation state of thylakoid membrane proteins and consequences on cyclic electron flow. Photosynt. Res. 29, 307–320 (2016).
Puthiyaveeti, S., van Oort, B. & Kirchhoff, H. Surface charge dynamics in photosynthetic membranes and the structural consequences. Nat. Plants 3, 17020 (2017).
Bellafiore, S., Barneche, F., Peltier, G. & Rochaix, J. D. State transitions and light adaptation require chloroplast thylakoid protein kinase STN7. Nature 433, 892–895 (2005).
Bonardi, V. et al. Photosystem II core phosphorylation and photosynthetic acclimation require two different protein kinases. Nature 437, 1179–1182 (2005).
Pribil, M., Pesaresi, P., Hertle, A., Barbato, R. & Leister, D. Role of plastid protein phosphatase TAP38 in LHCII dephosphorylation and thylakoid electron flow. PLoS Biol. 8, e1000288 (2010).
Samol, I. et al. Identification of a photosystem II phosphatase involved in light acclimation in Arabidopsis. Plant Cell 24, 2596–2609 (2012).
Fristedt, R. et al. Phosphorylation of photosystem II controls functional macroscopic folding of photosynthetic membranes in Arabidopsis. Plant Cell 21, 3950–3964 (2009).
Armbruster, U. et al. Arabidopsis CURVATURE THYLAKOID1 proteins modify thylakoid architecture by inducing membrane curvature. Plant Cell 25, 2661–2678 (2013).
Rozak, P. R., Seiser, R. M., Wacholtz, W. F. & Wise, R. R. Rapid, reversible alterations in spinach thylakoid appression upon changes in light intensity. Plant Cell Environ. 25, 421–429 (2002).
Anderson, J. M., Horton, P., Kim, E.-H. & Chow, W. S. Towards elucidation of dynamic structural changes of plant thylakoid architecture. Phil. Trans. R. Soc. Lond. B 367, 3515–3524 (2012).
Rintamaki, E. et al. Phosphorylation of light-harvesting complex II and photosystem II core proteins shows different irradiance-dependent regulation in vivo. J. Biol. Chem. 272, 30476–30482 (1997).
Ruban, A. V. & Johnson, M. P. Dynamics of higher plant photosystem cross-section associated with state transitions. Photosynth. Res. 99, 173–183 (2009).
Fristedt, R., Granath, P. & Vener, A. V. A protein phosphorylation threshold for functional stacking of plant photosynthetic membranes. PLoS ONE 5, e10963 (2010).
Benson, S. L. et al. An intact light harvesting complex I antenna system is required for complete state transitions in Arabidopsis. Nat. Plants 1, 15176 (2015).
Mekala, N. R., Soursa, M., Rantala, M., Aro, E. M. & Tikkanen, M. Plants actively avoid state transitions upon changes in light intensity: role of light-harvesting complex II protein dephosphorylation in high light. Plant Phys. 168, 721–734 (2015).
Wientjes, E., van Amerongen, H. & Croce, R. LHCII is an antenna of both photosystems after long-term acclimation. Biochim. Biophys. Acta 1827, 420–426 (2013).
Kyle, D. J., Staehelin, L. A. & Arntzen, C. J. Lateral mobility of the light-harvesting complex in chloroplast membranes controls excitation energy distribution in higher plants. Arch. Biochem. Biophys. 222, 527–541 (1983).
Grieco, M., Suorsa, M., Jajoo, A., Tikkanen, M. & Aro, E. M. Light-harvesting II antenna trimers connect energetically the entire photosynthetic machinery—including both photosystems II and I. Biochim. Biophys. Acta 1847, 607–619 (2015).
Sznee, K. et al. Jumping mode atomic force microscopy on grana membranes from spinach. J. Biol. Chem. 286, 39164–39171 (2011).
Johnson, M. P., Vasilev, C., Olsen, J. D. & Hunter, C. N. Nanodomains of cytochrome b 6 f and photosystem II complexes in spinach grana thylakoid membranes. Plant Cell 26, 3051–3061 (2014).
Phuthong, W. et al. The use of contact mode atomic force microscopy in aqueous medium for structural analysis of spinach photosynthetic complexes. Plant Physiol. 169, 1318–1332 (2015).
Su, X. et al. Structure and assembly mechanism of plant C2S2M2-type PSII-LHCII supercomplex. Science 357, 815–820 (2017).
Stroebel, D., Choquet, Y., Popot, J.-L. & Picot, D. An atypical haem in the cytochrome b 6 f complex. Nature 426, 413–418 (2003).
Qin, X., Suga, M., Kuang, T. & Shen, J. R. Structural basis for energy transfer pathways in the plant PSI-LHCI supercomplex. Science 348, 989–995 (2015).
MacGregor-Chatwin, C. et al. Lateral segregation of photosystem I in cyanobacterial thylakoids. Plant Cell 29, 1119–1136 (2017).
Fotiadis, D. et al. Surface analysis of the photosystem I complex by electron and atomic force microscopy. J. Mol. Biol. 283, 83–94 (1998).
Zhou, A. et al. Structure and conformational states of the bovine mitochondrial ATP synthase by cryo-EM. Elife 4, e10180 (2015).
Semchonok, D. A., Li, M., Bruce, B. D., Oostergetel, G. T. & Boekema, E. J. Cryo-EM structure of a tetrameric cyanobacterial photosystem I complex reveals novel subunit interactions. Biochim. Biophys. Acta 1857, 1619–1626 (2016).
Sathish Yadav, K. N. et al. Supercomplexes of plant photosystem I with cytochrome b 6 f, light-harvesting complex II and NDH. Biochim. Biophys. Acta 1858, 12–20 (2016).
Gustafsson, M. G. et al. Three-dimensional resolution doubling in wide-field fluorescence microscopy by structured illumination. Biophys. J. 9, 4957–4970 (2008).
Kirchhoff, H. et al. Dynamic control of protein diffusion within the granal thylakoid lumen. Proc. Natl Acad. Sci. USA 108, 20248–20253 (2011).
Haehnel, W. Photosynthetic electron transport in higher plants. Annu. Rev. Plant Physiol. 35, 659–693 (1984).
Joliot, P. & Johnson, G. Regulation of cyclic and linear electron flow in higher plants. Proc. Natl Acad. Sci. USA 108, 13317–13322 (2011).
Ruban, A. V., & Murchie, E. H. Assessing the photoprotective effectiveness of non-photochemical chlorophyll fluorescence quenching: a new approach. Biochim. Biophys. Acta 1817, 977–982 (2012).
Tikkanen, M., Grieco, M., Kangasjärvi, S. & Aro, E. M. Thylakoid protein phosphorylation in higher plant chloroplasts optimizes electron transfer under fluctuating light. Plant Physiol. 152, 723–735 (2010).
Kouril, R. et al. Structural characterization of a complex of photosystem I and light-harvesting complex II of Arabidopsis thaliana. Biochemistry 44, 10935–10940 (2005).
Bell, A. J., Frankel, L. K. & Bricker, T. M. High yield non-detergent isolation of photosystem I-light harvesting chlorophyll II membranes from spinach thylakoids. J. Biol. Chem. 290, 18429–18437 (2015).
Bos, I. et al. Multiple LHCII antennae can transfer energy efficiently to a single Photosystem I. Biochim. Biophys. Acta 1858, 371–378 (2017).
Belgio, E. et al. Economic photoprotection in photosystem II that retains a complete light-harvesting system with slow energy traps. Nat. Commun. 5, 4433 (2014).
Johnson, M. P. et al. Photoprotective energy dissipation involves the reorganization of photosystem II light harvesting complexes in the grana membranes of spinach chloroplasts. Plant Cell 23, 1468–1479 (2011).
Belgio, E., Ungerer, P. & Ruban, A. V. Light harvesting superstructures of green plant chloroplasts lacking photosystems. Plant Cell Environ. 38, 2035–2047 (2015).
Furbank, R. T., & Horton, P. Regulation of photosynthesis in isolated barley protoplasts: the contribution of cyclic photophosphorylation. Biochim. Biophys. Acta 894, 332–338 (1987).
Miyake, C., Miyata, M., Shinzaki, Y. & Tomizawa, K. CO2 response of cyclic electron flow around PSI (CEF-PSI) in tobacco leaves—relative electron fluxes through PSI and PSII determine the magnitude of non-photochemical quenching (NPQ) of Chl fluorescence. Plant Cell Physiol. 46, 629–637 (2005).
Albertsson, P. A., Andreasson, E., Stefansson, H. & Wollenberger, L. Fractionation of the thylakoid membrane. Methods Enzymol. 228, 469–482 (1994).
Melis, A. Kinetic analysis of P700 photoconversion: effect of secondary electron donation and plastocyanin inhibition. Arch. Biochem. Biophys. 217, 536–545 (1982).
Sanderson, D., Anderson, L. & Gross, E. Determination of the redox potential and diffusion coefficient of the protein plastocyanin using optically transparent filar electrodes. Biochim. Biophys. Acta 852, 269–278 (1987).
Blackwell, M. F., Gibas, C., Gygax, S., Roman, D. & Wagner, B. The plastoquinone diffusion coefficient in chloroplasts and its mechanistic implications. Biochim. Biophys. Acta 1183, 533–543 (1994).
Danielsson, R., Albertsson, P. A., Mamedov, F. & Styring, S. Quantification of photosystem I and II in different parts of the thylakoid membrane from spinach. Biochim. Biophys. Acta 1608, 53–61 (2004).
Pribil, M., Labs, M. & Leister, D. Structure and dynamics of thylakoids in land plants. J. Exp. Bot. 5, 1955–1972 (2014).
Acknowledgements
We wish to thank P. Horton (University of Sheffield) and F.-A. Wollman (CNRS, Paris) for fruitful discussions on the manuscript. We also thank J. Walker (University of Cambridge) for providing samples of the bovine ATP synthase complex, E. Murchie (University of Nottingham) for loan of the Dual-PAM and C. Hill (University of Sheffield) for assistance with the electron microscopy. M.P.J. acknowledges funding from the Biotechnology and Biological Sciences Research Council (UK) grant BB/M000265/1, the Leverhulme Trust grant RPG-2016-161, the Krebs Institute, the Grantham Centre for Sustainable Futures and the Kirkwood Memorial Fund. C.N.H. acknowledges the Photosynthetic Antenna Research Center (PARC), an Energy Frontier Research Center funded by the US Department of Energy, Office of Science, and Office of Basic Energy Sciences under Award Number DE-SC0001035. PARC’s role was to partially fund the Multimode VIII AFM system and to provide partial support for C.N.H. The SIM imaging was performed at the University of Sheffield Wolfson Light Microscopy Facility and was partly funded by MRC Grant MR/K015753/1.
Author information
Authors and Affiliations
Contributions
W.W., C.M.C. and M.J. performed the purification of membranes and characterized them by AFM. W.W. and M.J. performed the spectroscopy experiments. W.W. performed the electron microscopy experiments. W.W. performed the AFM, electron microscopy and spectroscopy data analysis, figure preparation and Monte Carlo simulations. G.E.M. assisted with the modelling. S.F.H.B. performed the three-dimensional SIM experiments and data analysis. X.H. performed the AFM experiments on purified ATP synthase. J.H. provided advice and support to the AFM experiments. The work was conceived and written by M.P.J. and C.N.H. All authors discussed the results and commented upon the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–5 and Supplementary Tables 1–3
Rights and permissions
About this article
Cite this article
Wood, W.H.J., MacGregor-Chatwin, C., Barnett, S.F.H. et al. Dynamic thylakoid stacking regulates the balance between linear and cyclic photosynthetic electron transfer. Nature Plants 4, 116–127 (2018). https://doi.org/10.1038/s41477-017-0092-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-017-0092-7
This article is cited by
-
Thylakoid membrane stacking controls electron transport mode during the dark-to-light transition by adjusting the distances between PSI and PSII
Nature Plants (2024)
-
The cytochrome b6f complex: plastoquinol oxidation and regulation of electron transport in chloroplasts
Photosynthesis Research (2024)
-
Salinity-induced changes in plastoquinone pool redox state in halophytic Mesembryanthemum crystallinum L.
Scientific Reports (2023)
-
Flavodiiron-mediated O2 photoreduction at photosystem I acceptor-side provides photoprotection to conifer thylakoids in early spring
Nature Communications (2023)
-
Solar energy conversion by photosystem II: principles and structures
Photosynthesis Research (2023)