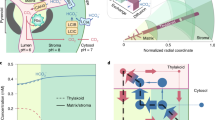
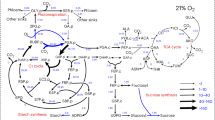
Abstract
Photorespiration is a major bioengineering target for increasing crop yields as it is often considered a wasteful process. Photorespiratory metabolism is integrated into leaf metabolism and thus may have certain benefits. Here, we show that plants can increase their rate of photosynthetic CO2 uptake when assimilating nitrogen de novo via the photorespiratory pathway by fixing carbon as amino acids in addition to carbohydrates. Plants fed NO3 − had higher rates of CO2 assimilation under photorespiratory than low-photorespiratory conditions, while plants lacking NO3 − nutrition exhibited lower stimulation of CO2 uptake. We modified the widely used Farquhar, von Caemmerer and Berry photosynthesis model to include the carbon and electron requirements for nitrogen assimilation via the photorespiratory pathway. Our modified model improves predictions of photosynthetic CO2 uptake and of rates of photosynthetic electron transport. The results highlight how photorespiration can improve photosynthetic performance despite reducing the efficiency of Rubisco carboxylation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Farquhar, G. D., von Caemmerer, S. & Berry, J. A. A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta 149, 78–90 (1980).
Busch, F. A., Sage, T. L., Cousins, A. B. & Sage, R. F. C3 plants enhance rates of photosynthesis by reassimilating photorespired and respired CO2. Plant Cell Environ. 36, 200–212 (2013).
Busch, F. A. Current methods for estimating the rate of photorespiration in leaves. Plant Biology 15, 648–655 (2013).
Sage, R. F., Sage, T. L. & Kocacinar, F. Photorespiration and the evolution of C4 photosynthesis. Annu. Rev. Plant Biol. 63, 19–47 (2012).
Wingler, A., Lea, P. J., Quick, W. P. & Leegood, R. C. Photorespiration: metabolic pathways and their role in stress protection. Philos. T. Roy. Soc. B 355, 1517–1529 (2000).
Bloom, A. J. Photorespiration and nitrate assimilation: a major intersection between plant carbon and nitrogen. Photosynth. Res. 123, 117–128 (2015).
Ros, R., Muñoz-Bertomeu, J. & Krueger, S. Serine in plants: biosynthesis, metabolism, and functions. Trends. Plant Sci. 19, 564–569 (2014).
Rachmilevitch, S., Cousins, A. B. & Bloom, A. J. Nitrate assimilation in plant shoots depends on photorespiration. Proc. Natl Acad. Sci. USA 101, 11506–11510 (2004).
Benstein, R. M. et al. Arabidopsis phosphoglycerate dehydrogenase1 of the phosphoserine pathway is essential for development and required for ammonium assimilation and tryptophan biosynthesis. Plant Cell 25, 5011–5029 (2013).
Andrews, M. The partitioning of nitrate assimilation between root and shoot of higher plants. Plant Cell Environ. 9, 511–519 (1986).
Scheurwater, I., Koren, M., Lambers, H. & Atkin, O. K. The contribution of roots and shoots to whole plant nitrate reduction in fast‐ and slow‐growing grass species. J. Exp. Bot. 53, 1635–1642 (2002).
Bloom, A. J., Caldwell, R. M., Finazzo, J., Warner, R. L. & Weissbart, J. Oxygen and carbon dioxide fluxes from barley shoots depend on nitrate assimilation. Plant Physiol. 91, 352–356 (1989).
Stitt, M. et al. Steps towards an integrated view of nitrogen metabolism. J. Exp. Bot. 53, 959–970 (2002).
von Caemmerer, S. & Farquhar, G. D. Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves. Planta 153, 376–387 (1981).
Sharkey, T. D. O2-insensitive photosynthesis in C3 plants: Its occurrence and a possible explanation. Plant Physiol. 78, 71–75 (1985).
Busch, F. A. & Sage, R. F. The sensitivity of photosynthesis to O2 and CO2 concentration identifies strong Rubisco control above the thermal optimum. New. Phytol. 213, 1036–1051 (2017).
Harley, P. C. & Sharkey, T. D. An improved model of C3 photosynthesis at high CO2: Reversed O2 sensitivity explained by lack of glycerate reentry into the chloroplast. Photosynth. Res. 27, 169–178 (1991).
von Caemmerer, S. Biochemical Models of Leaf Photosynthesis (CSIRO Publishing, Collingwood, 2000).
Sage, R., Sharkey, T. & Pearcy, R. The effect of leaf nitrogen and temperature on the CO2 response of photosynthesis in the C3 dicot Chenopodium album L. Aust. J. Plant. Physiol. 17, 135–148 (1990).
Sage, R. F. & Sharkey, T. D. The effect of temperature on the occurrence of O2 and CO2 insensitive photosynthesis in field-grown plants. Plant Physiol. 84, 658–664 (1987).
Woo, K. & Wong, S. Inhibition of CO2 assimilation by supraoptimal CO2: Effect of light and temperature. Funct. Plant Biol. 10, 75–85 (1983).
Sharkey, T. D. & Vassey, T. L. Low oxygen inhibition of photosynthesis is caused by inhibition of starch synthesis. Plant Physiol. 90, 385–387 (1989).
Bauwe, H., Hagemann, M. & Fernie, A. R. Photorespiration: Players, partners and origin. Trends. Plant Sci. 15, 330–336 (2010).
Dirks, R. C., Singh, M., Potter, G. S., Sobotka, L. G. & Schaefer, J. Carbon partitioning in soybean (Glycine max) leaves by combined 11C and 13C labeling. New Phytol. 196, 1109–1121 (2012).
Misra, J. B. Integrated operation of the photorespiratory cycle and cytosolic metabolism in the modulation of primary nitrogen assimilation and export of organic N-transport compounds from leaves: A hypothesis. J. Plant Physiol. 171, 319–328 (2014).
Xu, G., Fan, X. & Miller, A. J. Plant nitrogen assimilation and use efficiency. Annu. Rev. Plant Biol. 63, 153–182 (2012).
Tischner, R. Nitrate uptake and reduction in higher and lower plants. Plant Cell Environ. 23, 1005–1024 (2000).
Taniguchi, M. & Miyake, H. Redox-shuttling between chloroplast and cytosol: Integration of intra-chloroplast and extra-chloroplast metabolism. Curr. Opin. Plant Biol. 15, 252–260 (2012).
Rathnam, C. K. M. Malate and dihydroxyacetone phosphate-dependent nitrate reduction in spinach leaf protoplasts. Plant Physiol. 62, 220–223 (1978).
Holfgrefe, S., Backhausen, J. E., Kitzmann, C. & Scheibe, R. Regulation of steady-state photosynthesis in isolated intact chloroplasts under constant light: Responses of carbon fluxes, metabolite pools and enzyme-activation states to changes of electron pressure. Plant Cell Physiol. 38, 1207–1216 (1997).
Kopriva, S. et al. Interaction of sulfate assimilation with carbon and nitrogen metabolism in Lemna minor. Plant Physiol. 130, 1406–1413 (2002).
Van Beusichem, M. L., Kirkby, E. A. & Baas, R. Influence of nitrate and ammonium nutrition on the uptake, assimilation, and distribution of nutrients in Ricinus communis. Plant Physiol. 86, 914–921 (1988).
Bloom, A. J. et al. CO2 enrichment inhibits shoot nitrate assimilation in C3 but not C4 plants and slows growth under nitrate in C3 plants. Ecology 93, 355–367 (2012).
Lewis, O. A. M. & Chadwick, S. An 15N investigation into nitrogen assimilation in hydroponically-grown barley (Hordeum vulgare L. cv. Clipper) in response to nitrate, ammonium and mixed nitrate and ammonium nutrition. New Phytol. 95, 635–646 (1983).
Murphy, A. T. & Lewis, O. A. M. Effect of nitrogen feeding source on the supply of nitrogen from root to shoot and the site of nitrogen assimilation in maize (Zea mays L. cv. R201). New Phytol. 107, 327–333 (1987).
Fair, P., Tew, J. & Cresswell, C. F. Enzyme activities associated with carbon dioxide exchange in illuminated leaves of Hordeum vulgare L. III. Effects of concentration and form of nitrogen supplied on carbon dioxide compensation point. Ann. Bot. London 38, 39–43 (1974).
Fridlyand, L. E., Backhausen, J. E. & Scheibe, R. Flux control of the malate valve in leaf cells. Arch. Biochem. Biophys. 349, 290–298 (1998).
Abadie, C., Boex-Fontvieille, E. R. A., Carroll, A. J. & Tcherkez, G. In vivo stoichiometry of photorespiratory metabolism. Nat. Plants 2, 15220 (2016).
Close, T. J. Dehydrins: A commonalty in the response of plants to dehydration and low temperature. Physiol. Plant. 100, 291–296 (1997).
Layton, B. E. et al. Dehydration-induced expression of a 31-kDa dehydrin in Polypodium polypodioides (Polypodiaceae) may enable large, reversible deformation of cell walls. Am. J. Bot. 97, 535–544 (2010).
Sakamoto, A. & Murata, N. The role of glycine betaine in the protection of plants from stress: Clues from transgenic plants. Plant Cell Environ. 25, 163–171 (2002).
Noctor, G. et al. Glutathione in plants: An integrated overview. Plant Cell Environ. 35, 454–484 (2012).
Laisk, A., Eichelmann, H., Oja, V., Rasulov, B. & Ramma, H. Photosystem II cycle and alternative electron flow in leaves. Plant Cell Physiol. 47, 972–983 (2006).
Warren, C. Estimating the internal conductance to CO2 movement. Funct. Plant Biol. 33, 431–442 (2006).
Feng, Z. et al. Constraints to nitrogen acquisition of terrestrial plants under elevated CO2. Glob. Change Biol. 21, 3152–3168 (2015).
Noctor, G. & Foyer, C. H. A re-evaluation of the ATP:NADPH budget during C3 photosynthesis: A contribution from nitrate assimilation and its associated respiratory activity? J. Exp. Bot. 49, 1895–1908 (1998).
Sharkey, T. D., Bernacchi, C. J., Farquhar, G. D. & Singsaas, E. L. Fitting photosynthetic carbon dioxide response curves for C3 leaves. Plant Cell Environ. 30, 1035–1040 (2007).
Acknowledgements
We thank M. Holloway-Phillips for assistance with growing the plants. This work was supported by the ARC Centre of Excellence for Translational Photosynthesis and by the Australian Science Industry and Endowment Fund (SIEF grant RP04-122). R.F.S. was supported by Discovery grants 154273-2007 and 154273-2012 from the Natural Science and Engineering Research Council (NSERC) of Canada.
Author information
Authors and Affiliations
Contributions
F.A.B. conceived the study and undertook the experimental work, with input from R.F.S. F.A.B. and G.D.F. carried out the modelling. F.A.B. wrote the manuscript with help from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
A description of the derivation of expressions for α G and α S, Supplementary Figures 1–4. Supplementary Table 1.
Rights and permissions
About this article
Cite this article
Busch, F.A., Sage, R.F. & Farquhar, G.D. Plants increase CO2 uptake by assimilating nitrogen via the photorespiratory pathway. Nature Plants 4, 46–54 (2018). https://doi.org/10.1038/s41477-017-0065-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-017-0065-x
This article is cited by
-
Nature-based Solutions can help restore degraded grasslands and increase carbon sequestration in the Tibetan Plateau
Communications Earth & Environment (2024)
-
Photon costs of shoot and root NO3−, and root NH4+, assimilation in terrestrial vascular plants considering associated pH regulation, osmotic and ontogenetic effects
Photosynthesis Research (2023)
-
Potential Impacts of Exogenous Nitrogen Enrichment on Distribution and Transfer of Nitrogen in Plant-Soil System of Suaeda salsa Marsh in the Yellow River Estuary, China
Journal of Ocean University of China (2023)
-
An integrated isotopic labeling and freeze sampling apparatus (ILSA) to support sampling leaf metabolomics at a centi-second scale
Plant Methods (2022)
-
Integrated flux and pool size analysis in plant central metabolism reveals unique roles of glycine and serine during photorespiration
Nature Plants (2022)