Abstract
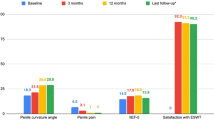
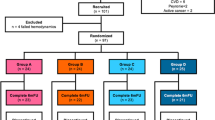
This study retrospectively examined the effects of low-intensity extracorporeal shockwave therapy and tadalafil on erectile dysfunction patients. 116 patients got low-intensity extracorporeal shockwave therapy twice weekly for 3 weeks, 5 mg of tadalafil daily for 3 weeks, and adjuvant therapy for 6 months. Group A (1 year), Group B (2 years), Group C (3 years), and Group D (4+ years) were treated patients’ follow-up groups. The patients’ International Index of Erectile Function-5 (IIEF-5) scores were gathered at the start of treatment via face-to-face interviews and at the end of follow-ups by telephone conversations. IIEF-5 scores and the minimal clinically significant differences were the main outcomes. Mean follow-up length, age, and IIEF-5 scores of the patients were 2 ± 1.16 years, 47.34 ± 12.65 years, and 12.09 ± 3.66 points, respectively. Compared to baseline, treatment increased the median IIEF-5 scores of patients in groups A, B, C, and D by 7 [3–12], 6 [0–8], 7 [1–9], and 6.5 [2.5–10] points, respectively (p = 0.001). 71%, 63%, 65.8%, and 65% of treated patients in groups A, B, C, and D met the minimal clinically significant differences criteria (n = 77). Mild disease patients were 9.14 times more likely to respond to treatment than severe illness patients (OR, 9.14; 95% CI, 1.28–65.46; P = 0.02). Low-intensity extracorporeal shockwave therapy and 5 mg of tadalafil can treat erectile dysfunction for up to 4 years with sustained outcomes. This treatment is optimal for mild illnesses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The data generated during this study can be found within the published article and its supplementary files. The additional data are available from the corresponding author upon reasonable request.
References
Consensus NIH. Conference. Impotence. NIH consensus development panel on impotence. JAMA. 1993;270:83–90.
Benet AE, Melman A. The epidemiology of erectile dysfunction. Urol Clin North Am. 1995;22:699–709.
Sullivan ME, Keoghane SR, Miller MA. Vascular risk factors and erectile dysfunction. BJU Int. 2001;87:838–45.
Salonia A, Castagna G, Saccà A, Ferrari M, Capitanio U, Castiglione F, et al. Is erectile dysfunction a reliable proxy of general male health status? The case for the international index of erectile function—erectile function domain. J Sex Med. 2012;9:2708–15.
Salonia A, Bettocchi C, Carvalho J, Corona G, Jones TH, Kadioglu A, et al. 2020 EAU guidelines on sexual and reproductive health. Arnhem: EAU Guidelines Office; 2020. https://uroweb.org/guideline/sexual-and-reproductive-health.
Yuan J, Zhang R, Yang Z, Lee J, Liu Y, Tian J, et al. Comparative effectiveness and safety of oral phosphodiesterase type 5 inhibitors for erectile dysfunction: a systematic review and network meta-analysis. Eur Urol. 2013;63:902–12.
Goldstein I, Lue TF, Padma-Nathan H, Rosen RC, Steers WD, Wicker PA. Oral sildenafil in the treatment of erectile dysfunction. Sildenafil Study Group. N. Engl J Med. 1998;338:1397–404.
Porst H, Rosen R, Padma-Nathan H, Goldstein I, Giuliano F, Ulbrich E, et al. The efficacy and tolerability of vardenafil, a new, oral, selective phosphodiesterase type 5 inhibitor, in patients with erectile dysfunction: the first at-home clinical trial. Int J Impot Res. 2001;13:192–9.
Brock GB, McMahon CG, Chen KK, Costigan T, Shen W, Watkins V, et al. Efficacy and safety of tadalafil for the treatment of erectile dysfunction: results of integrated analyses. J Urol. 2002;168:1332–6.
Porst H. The rationale for prostaglandin E1 in erectile failure: a survey of worldwide experience. J Urol. 1996;155:802–15.
Kohn TP, Rajanahally S, Hellstrom WJG, Hsieh TC, Raheem OA. Global trends in prevalence, treatments, and costs of penile prosthesis for erectile dysfunction in men. Eur Urol Focus. 2022;8:803–13.
Porst H, Giuliano F, Glina S, Ralph D, Casabé AR, Elion-Mboussa A, et al. Evaluation of the efficacy and safety of once-a-day dosing of tadalafil 5mg and 10mg in the treatment of erectile dysfunction: results of a multicenter, randomized, double-blind, placebo-controlled trial. Eur Urol. 2006;50:351–9.
Mulhall JP, Jahoda AE, Ahmed A, Parker M. Analysis of the consistency of intraurethral prostaglandin E(1) (MUSE) during at-home use. Urology. 2001;58:262–6.
Vardi Y, Appel B, Jacob G, Massarwi O, Gruenwald I. Can low-intensity extracorporeal shockwave therapy improve erectile function? A 6-month follow-up pilot study in patients with organic erectile dysfunction. Eur Urol. 2010;58:243–8.
Vardi Y, Appel B, Kilchevsky A, Gruenwald I. Does low intensity extracorporeal shock wave therapy have a physiological effect on erectile function? Short-term results of a randomized, double-blind, sham controlled study. J Urol. 2012;187:1769–75.
Aicher A, Heeschen C, Sasaki K, Urbich C, Zeiher AM, Dimmeler S. Low-energy shock wave for enhancing recruitment of endothelial progenitor cells: a new modality to increase efficacy of cell therapy in chronic hind limb ischemia. Circulation. 2006;114:2823–30.
Clavijo RI, Kohn TP, Kohn JR, Ramasamy R. Effects of low-intensity extracorporeal shockwave therapy on erectile dysfunction: a systematic review and meta-analysis. J Sex Med. 2017;14:27–35.
Lu Z, Lin G, Reed-Maldonado A, Wang C, Lee YC, Lue TF. Low-intensity extracorporeal shock wave treatment improves erectile function: a systematic review and meta-analysis. Eur Urol. 2017;71:223–33.
Rosen RC, Cappelleri JC, Smith MD, Lipsky J, Peña BM. Development and evaluation of an abridged, 5-item version of the International Index of Erectile Function (IIEF-5) as a diagnostic tool for erectile dysfunction. Int J Impot Res. 1999;11:319–26.
Turunc T, Deveci S, Güvel S, Peşkircioğlu L. The assessment of Turkish validation with 5 question version of International Index of Erectile Function (IIEF-5). Turk J Urol. 2007;33:45–49.
Rosen RC, Allen KR, Ni X, Araujo AB. Minimal clinically important differences in the erectile function domain of the International Index of Erectile Function scale. Eur Urol. 2011;60:1010–6.
Porst H, Buvat J, Meuleman E, Michal V, Wagner G. Intracavernous Alprostadil Alfadex—an effective and well tolerated treatment for erectile dysfunction. Results of a long-term European study. Int J Impot Res. 1998;10:225–31.
Zucchi A, Costantini E, Scroppo FI, Silvani M, Kopa Z, Illiano E, et al. The first-generation phosphodiesterase 5 inhibitors and their pharmacokinetic issue. Andrology. 2019;7:804–17.
Shah T, Wang R. A review of factors affecting patient satisfaction with inflatable penile prosthesis. Sex Med Rev. 2021;9:350–7.
Kitrey ND, Vardi Y, Appel B, Shechter A, Massarwi O, Abu-Ghanem Y, et al. Low intensity shock wave treatment for erectile dysfunction-how long does the effect last. J Urol. 2018;200:167–70.
Sokolakis I, Dimitriadis F, Teo P, Hatzichristodoulou G, Hatzichristou D, Giuliano F. The basic science behind low-intensity extracorporeal shockwave therapy for erectile dysfunction: a systematic scoping review of pre-clinical studies. J Sex Med. 2019;16:168–94.
Gallo L, Pecoraro S, Sarnacchiaro P. Adjuvant daily therapy with L-arginine 2,500 mg and tadalafil 5 mg increases efficacy and duration of benefits of low-intensity extracorporeal shock wave therapy for erectile dysfunction: a prospective, randomized, single-blinded study with 1-year follow-up. Investig Clin Urol. 2022;63:83–91.
Verze P, Capece M, Creta M, La Rocca R, Persico F, Spirito L, et al. Efficacy and safety of low-intensity shockwave therapy plus tadalafil 5 mg once daily in men with type 2 diabetes mellitus and erectile dysfunction: a matched-pair comparison study. Asian J Androl. 2020;22:379–82.
Palmieri A, Arcaniolo D, Palumbo F, Verze P, Liguori G, Mondaini N, et al. Low intensity shockwave therapy in combination with phosphodiesterase-5 inhibitors is an effective and safe treatment option in patients with vasculogenic ED who are PDE5i non-responders: a multicenter single-arm clinical trial. Int J Impot Res. 2021;33:634–40.
Author information
Authors and Affiliations
Contributions
Yurdaer Kaynak designed the study and contributed to data extraction. The study was written by Yurdaer Kaynak. Ilan Gruenwald contributed to the writing of the paper and editing. Both of them approved the final version and submitted the paper to the journal.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study was done under ethical approval (approval number: ESH/GOEK2022/1).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaynak, Y., Gruenwald, I. Long-term effects of combination treatment comprising low-intensity extracorporeal shockwave therapy and tadalafil for patients with erectile dysfunction: a retrospective study. Int J Impot Res (2023). https://doi.org/10.1038/s41443-023-00757-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41443-023-00757-7